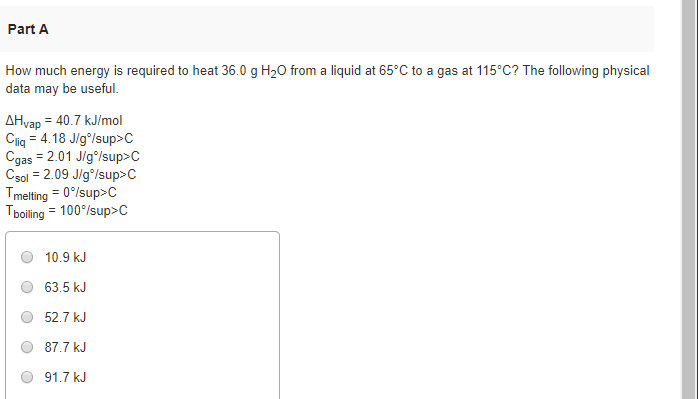
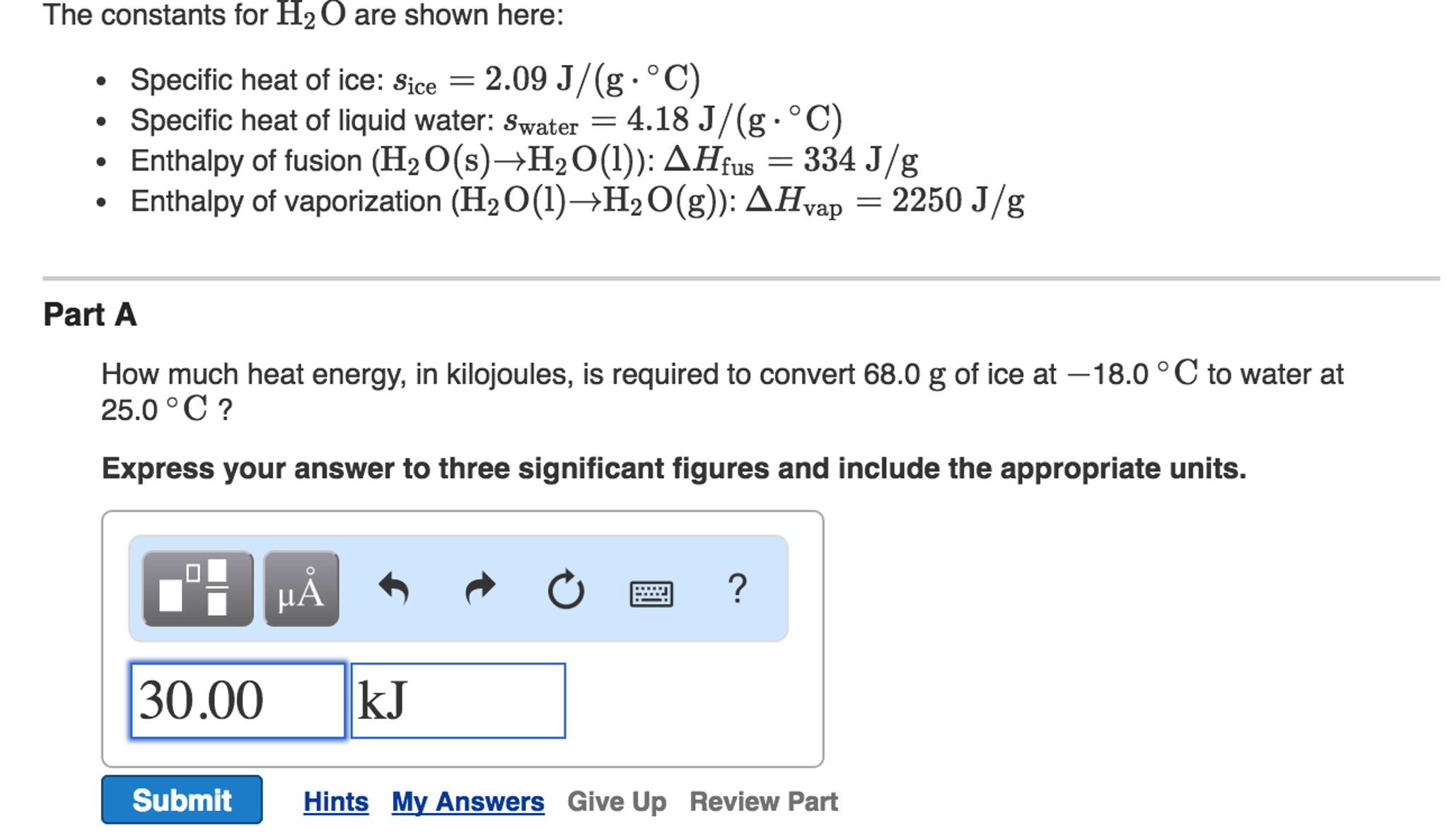
How Much Energy Is Required To Heat 36.0 G H2o

Have you ever wondered about the little things that make up our world? Sometimes, the most ordinary things hold surprising secrets. Take water, for instance. We use it every day, but do you know how much oomph it takes to get it nice and warm?
Let's talk about 36.0 grams of H2O. That’s not a whole lot, is it? It’s like a couple of big gulps from your water bottle. But even this tiny amount of liquid has an energy story waiting to be told. It's a story of molecules dancing and energy zipping around.
Imagine you have a tiny pot, just big enough for our 36.0 grams of water. You want to make it a little warmer. Not boiling, just… comfortably warm. How much effort does that really take? It's like asking how much energy it takes to give a tiny ant a warm hug.
The secret sauce here is something called the specific heat capacity. It's a fancy term for how much energy it takes to raise the temperature of something by a certain amount. For water, this number is pretty special. It means water is a bit of a diva when it comes to getting heated up.
Think of it like this: some things are super eager to get hot, like a piece of metal in the sun. Others are more laid-back, needing a bit more coaxing. Water, with its specific heat capacity, is more on the laid-back side. It likes to take its time warming up.
So, for our 36.0 grams of H2O, we're looking at a specific amount of energy. It's not a giant number, but it's certainly not zero! It's the energy needed to make those water molecules jiggle a little faster. More jiggling means warmer water. Simple, right?
The unit we usually use for this energy is Joules. It's like the standard measurement for how much "heat energy" is transferred. So, when we talk about warming our water, we're talking about adding a certain number of Joules. It’s like counting spoonfuls of energy!

Now, the exact number depends on how much warmer we want our water to get. Are we talking about a tiny nudge in temperature, or are we aiming for a noticeable change? This is where things get really interesting. We can choose our adventure!
Let's say we want to warm our 36.0 grams of H2O by just 1 degree Celsius. That's a small change, like the difference between a cool morning and a slightly less cool morning. Even this small change requires a specific amount of energy. It's the minimum energy needed to get things moving a little.
For 36.0 grams of water, raising its temperature by 1 degree Celsius takes about 150.6 Joules. See? It's not a huge number! It's like giving that ant a very gentle, warm pat. Enough to feel it, but not enough to make it jump.
Isn't that neat? A tiny bit of water, a small temperature change, and a calculable amount of energy. It’s like a mini-science experiment happening right there. You could, in theory, measure this yourself with the right tools!
But what if we wanted to warm it up more? What if we wanted it to feel delightfully warm, say, a change of 10 degrees Celsius? Now we're talking about a more significant temperature hike. It’s like going from that cool morning to a pleasantly warm afternoon.

To achieve this, we’d need ten times the energy. So, instead of 150.6 Joules, we’d be looking at around 1506 Joules. That’s still a pretty manageable amount of energy. It’s like giving our ant a series of warm pats, getting it to do a little dance.
The magic formula for this is actually quite simple: Energy = mass × specific heat capacity × change in temperature. That's it! You plug in the numbers, and out pops the energy required. It’s like a secret code for understanding how heat works.
The specific heat capacity of water is approximately 4.18 Joules per gram per degree Celsius. This is the key ingredient that makes water so special. It's why oceans can absorb so much heat without getting drastically hotter, and why your coffee stays warm for a while.
So, for our 36.0 grams of H2O:
Mass = 36.0 g Specific Heat Capacity = 4.18 J/g°C Change in Temperature = let’s say 10°C
Energy = 36.0 g × 4.18 J/g°C × 10°C = 1504.8 Joules. (Slightly different from our earlier estimate due to rounding, but you get the idea!)
This number, 1504.8 Joules, is the energy story of our small sample of water. It’s the energy that makes those molecules shimmy and shake a little faster, transforming from cool to cozy. It’s a tiny victory for thermodynamics!
Why is this so entertaining? Because it takes the invisible force of energy and makes it tangible. We can put a number on it. We can understand how much "work" is being done to change the state of something so common.
It’s like looking at a tiny engine and figuring out exactly how much fuel it needs to run. Except, in this case, the "engine" is water, and the "fuel" is heat energy. And our little engine only needs a modest amount of fuel to do its job.
What makes this special is that it applies to all water. Whether it’s in your teacup, a puddle on the sidewalk, or a vast ocean, the principles are the same. Water's unique ability to hold onto heat is a fundamental part of our planet’s climate and our daily lives.
Think about cooking. When you boil water for pasta, you're adding a significant amount of energy. But the initial warming of that water? That's just a gentle introduction. It’s the start of a beautiful relationship between energy and matter.
This simple calculation allows us to predict and understand thermal processes. It’s the foundation for everything from designing efficient heating systems to understanding how our bodies regulate temperature. It’s science made practical and, dare I say, a little bit magical.
So, next time you're warming up your drink, or even just looking at a glass of water, remember the energy involved. Remember those 36.0 grams of H2O and the Joules that make them warm. It’s a tiny detail, but it’s a part of a much bigger, fascinating picture.
It's a reminder that even the simplest things have complex and beautiful scientific stories. And figuring out how much energy it takes to warm a little bit of water is a fun way to start exploring those stories. It’s an invitation to be curious about the world around you.
Perhaps it will inspire you to experiment (safely, of course!). Maybe you’ll wonder about other substances. What about heating up some oil? Or melting some ice? The energy questions just keep on coming!
The universe is full of these little scientific puzzles, and the energy needed to heat 36.0 grams of H2O is just one of them. It's an accessible mystery, a gateway into the wonderful world of physics and chemistry. And who knows what other amazing discoveries await your curiosity?
