How Many Valence Electrons Does Roentgenium Have
Hey there, cosmic wanderer! Ever find yourself staring at the night sky, or maybe just the cool, metallic sheen of your everyday objects, and wondering about the tiny, invisible forces that hold everything together? It’s a pretty mind-blowing thought, right? We’re talking about the building blocks of the universe, the stuff that makes a sparkly diamond sparkle, or your phone hum with life. And today, we’re diving headfirst into the dazzling, and perhaps a little bit obscure, world of Roentgenium.
Now, I know what you’re thinking. "Roentgen-what-ium?" Don’t worry, you’re not alone. This isn't exactly the element you’ll find on your kitchen table next to the salt and pepper. Roentgenium, with the not-so-catchy symbol Rg, is a bit of a superstar in the realm of synthetic elements. It’s a heavyweight, a real rarity, and honestly, kind of a rockstar of the periodic table. We’re talking about an element that doesn’t exist in nature and is created in super-high-tech labs. Pretty cool, huh?
So, why are we even talking about this elusive element? Because understanding its valence electrons gives us a peek into its personality, its potential to bond, and ultimately, its place in the grand cosmic dance of chemistry. Think of valence electrons as the element's social butterflies, the ones who mingle and make connections. They’re the outermost electrons in an atom, and they’re the key players when it comes to chemical reactions. They’re the reason why water is so… watery, and why oxygen is so eager to pair up with things.
The Quest for Clarity: Roentgenium's Valence Electron Count
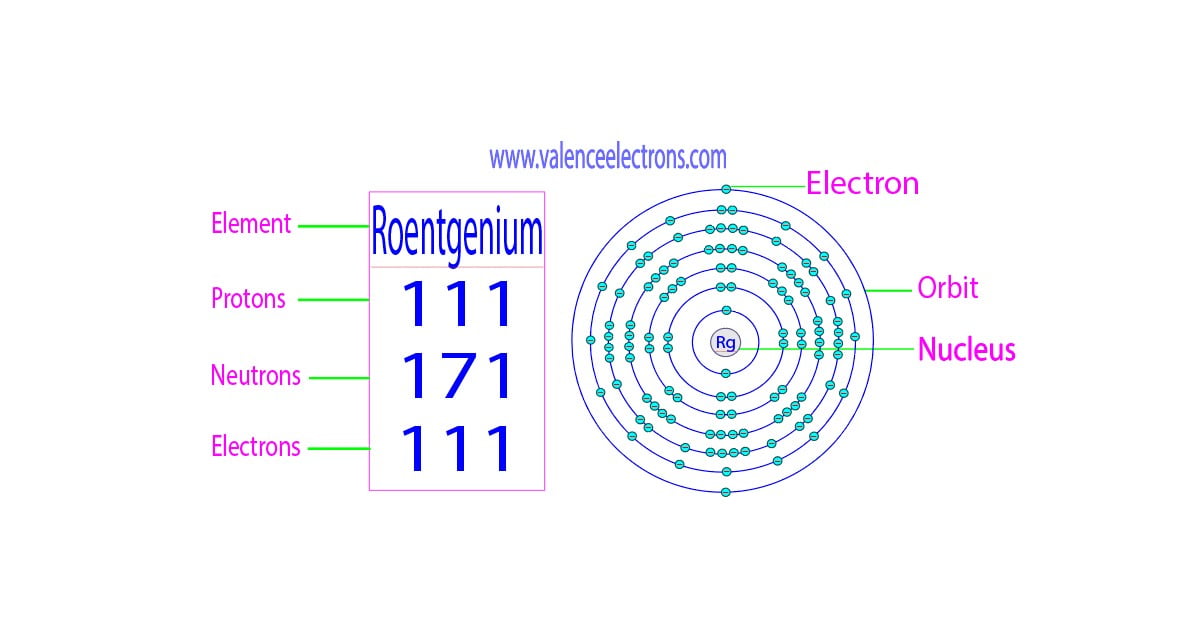
Now, for the big question, the one that might be keeping you up at night (or at least mildly curious): How many valence electrons does Roentgenium have? Drumroll, please… For Roentgenium, the answer is a tad complex, and in the typical, laid-back style of this element’s existence, it’s not as straightforward as you might imagine.
Generally, based on its position in the periodic table, specifically in Group 11 (alongside copper, silver, and gold – talk about a fancy neighborhood!), Roentgenium is expected to have one valence electron in its outermost shell. This is the conventional wisdom, the textbook answer that makes a lot of sense when you look at its electron configuration. Imagine it like this: it’s got that one super-available electron, just waiting to be part of something bigger, to share its energy and form a bond.
However, here’s where things get a little more nuanced, like trying to perfectly fold a fitted sheet. Because Roentgenium is a superheavy element, its behavior can get a little… quirky. The electrons in these heavy elements experience some pretty intense relativistic effects. Think of it like the electrons are zipping around so fast due to the massive nucleus that their mass actually increases, and their orbitals behave a bit differently than they would for lighter elements. This means that while the theoretical expectation is one valence electron, the reality might be more… fluid.

Some theoretical models and calculations suggest that due to these relativistic effects, the distinction between the valence shell and the inner electron shells can become blurred. This means that electrons from inner shells might be more readily available to participate in bonding than we’d initially predict. So, while one is the most commonly cited and generally accepted number for its valence electron count, it’s not entirely out of the question for other electrons to get in on the action under certain, very specific circumstances. It's like the universe saying, "Sure, one is the main star, but sometimes the supporting cast gets a cameo!"
Why Does This Even Matter, Dude?
Okay, so we’ve got this super-rare, lab-created element, and we’re dissecting its electron count. Why should we, as everyday folks, care? Well, it’s all about the bigger picture, the interconnectedness of everything. Even though you’re unlikely to be making Roentgenium jewelry anytime soon (or ever, really!), understanding these fundamental building blocks helps us appreciate the world around us in a deeper, more meaningful way.
Think about it: the way atoms interact, the way they form bonds, dictates the properties of every single substance we encounter. The single valence electron (or the potential for more to join the party) in Roentgenium’s case hints at its potential to behave like other Group 11 elements – the precious metals. Copper conducts electricity beautifully, silver is iconic for its luster and antimicrobial properties, and gold… well, gold is gold. It’s a symbol of wealth, beauty, and incredible durability. Roentgenium, being in the same family, could theoretically share some of these characteristics.

This is where the fun begins! Even though we can’t hold Roentgenium in our hands, scientists use their understanding of its electron configuration to predict its properties. This is crucial for advancing our knowledge of nuclear physics, creating new isotopes, and even, in the long run, developing technologies we can’t even dream of yet. It’s like a treasure hunt for scientific understanding!
Practical Tips for Embracing Your Inner Chemist (No Lab Coat Required!)
Alright, so maybe you’re not about to synthesize your own elements in your garage. But the principles of chemistry are everywhere! Here are a few ways to connect with the idea of atoms and their electrons in your daily life:
- Appreciate Your Sparkly Things: That necklace you love? That beautiful silverware? They’re likely made of or plated with elements like silver or gold, which share that characteristic single valence electron with Roentgenium. Next time you admire their shine, give a little nod to the electrons doing their thang!
- Observe Electrical Conductivity: Ever noticed how certain materials conduct electricity better than others? Think about the copper wires in your appliances. That conductivity is a direct result of the electrons in copper being free to move. It’s a visible manifestation of atomic structure!
- Think About Bonding in Everyday Life: When you bake a cake, you’re creating new chemical bonds! The heat causes ingredients to react, forming new compounds with different properties. It’s all about how atoms, and their valence electrons, decide to pair up.
- Dive into Documentaries: There are tons of amazing documentaries about the periodic table, the universe, and the elements. They’re a fantastic way to learn complex concepts in an engaging and visually appealing way. Think of it as Netflix for your brain!
A Little Pop Culture Vibe
Roentgenium, being so rare and synthetic, feels a bit like a character from a sci-fi movie. Imagine it as a super-rare, glowing artifact discovered on an alien planet, or a highly sought-after element in a futuristic quest. Its very existence pushes the boundaries of what we thought was possible.
And speaking of boundaries, the element’s name itself is a tribute to Wilhelm Conrad Röntgen, the physicist who discovered X-rays. So, it's not just a random string of letters; it’s a nod to a groundbreaking scientific discovery! It’s like an element wearing a tiny, invisible cape in honor of a scientific hero.
The ongoing research into superheavy elements like Roentgenium is like a real-life science fiction novel. Scientists are constantly pushing the limits, trying to create even heavier elements and understand their stability. It’s a thrilling frontier, and who knows what discoveries await us? Maybe one day, an element named after a Marvel superhero will grace the periodic table!
The Nuance is Where the Magic Happens
The fact that Roentgenium’s valence electron count might have a little bit of wiggle room, thanks to relativistic effects, is actually pretty darn cool. It reminds us that science isn't always black and white. It’s full of fascinating complexities and unexpected twists. It’s like discovering that your favorite artist sometimes uses a brushstroke you wouldn’t expect, and it makes the masterpiece even more intriguing.
This complexity is what makes the universe so endlessly fascinating. It’s not just about memorizing facts; it’s about understanding the underlying principles and appreciating the subtle nuances that make things work the way they do. It’s the difference between just looking at a pretty picture and understanding the artist’s technique and inspiration.
A Daily Dose of Wonder
So, what’s the takeaway from our little chat about Roentgenium and its valence electrons? It’s that even in the most obscure corners of science, there are fundamental principles at play that connect to our everyday lives. The forces that govern how Roentgenium might bond are the same forces that hold your coffee mug together, that allow electricity to flow through your phone, and that create the vibrant colors of a sunset.
The universe is a vast, intricate, and utterly beautiful tapestry, woven from the interactions of countless atoms. And while Roentgenium might be a bit of a cosmic recluse, understanding its electron configuration is like deciphering a tiny, but crucial, thread in that grand design. It’s a reminder that there’s always more to discover, more to learn, and more to be amazed by, right in our own backyards – and in the heart of the atom!
So, next time you’re feeling a little disconnected, a little unsure of your place in the grand scheme of things, take a moment to appreciate the invisible forces that bind us all. They're the silent orchestrators of our reality, and even a superheavy, synthetic element like Roentgenium has a story to tell about them. And that, my friends, is pretty darn awesome.
