How Many Valence Electrons Does A Tin Sn Atom Have
So, you're curious about Tin? That's awesome! You've stumbled upon a little piece of the universe that's actually pretty darn cool. And when we talk about Tin, specifically its valence electrons, we're diving into a tiny, fascinating world. It's like peeking behind the curtain of how matter works, and honestly, it’s way more exciting than you might think!
Think of atoms as tiny LEGO bricks that build everything around us. Each LEGO brick has certain "connectors" that let it stick to other bricks. In the world of atoms, these connectors are called valence electrons. They're the super important bits on the outside of an atom that get to play with other atoms. It's all about making connections, forming bonds, and creating all the amazing stuff we see and touch.
Now, let's get to the star of our show: Tin. This metal is probably more familiar to you than you realize. You might have heard of tin cans, right? That shiny coating that keeps your food fresh? Yep, that's Tin! It's been used for ages, and it’s got a special place in history. From ancient bronze swords (which is actually a mix of copper and tin!) to modern electronics, Tin has been a reliable buddy for humans.
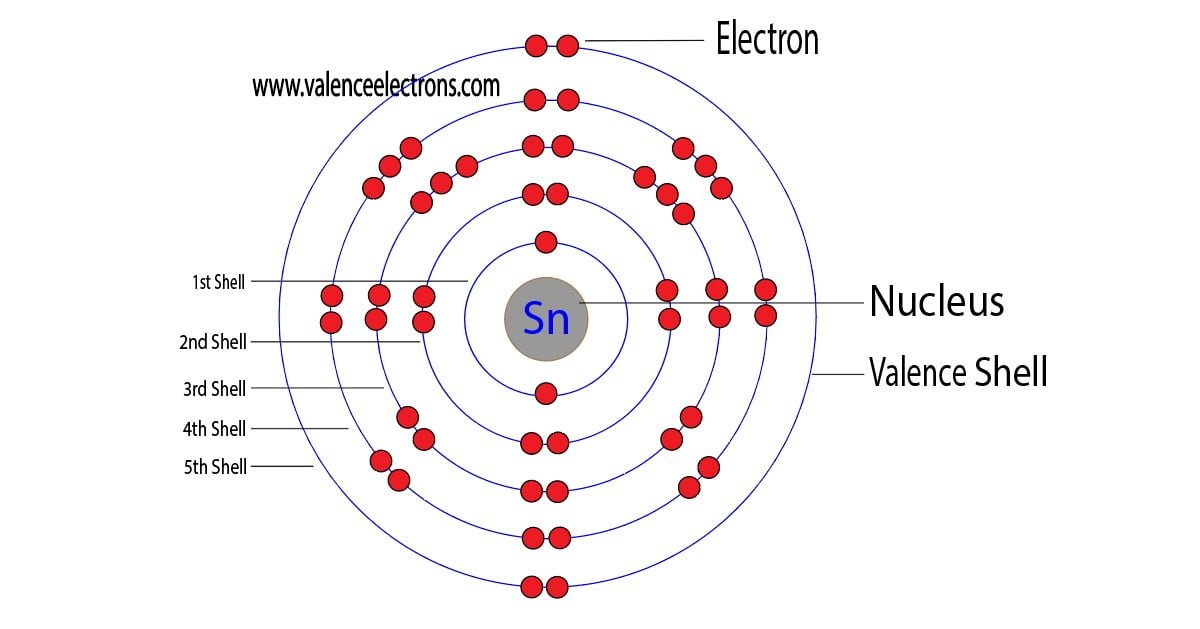
So, how many of these super-connectors, these valence electrons, does a Tin atom have? Drumroll, please… Tin (which has the cool symbol Sn, don’t ask me why, it’s a secret code from Latin!) has four valence electrons. Four! That’s like having four handy little hands ready to shake hands with other atoms. It makes Tin quite flexible and able to do some neat tricks in the world of chemistry.
Why is this so entertaining? Well, imagine a party. Every atom is invited, and they're all looking to mingle. The atoms with more valence electrons are like the super social butterflies, always ready to chat and make new friends. Atoms with fewer might be a bit more reserved. Tin, with its four valence electrons, is like that friendly person who’s happy to join any group, forming strong bonds and creating interesting combinations.
This number, four, is actually a sweet spot for Tin. It allows it to form stable compounds. Think about how strong a table with four legs is, right? It’s pretty stable. In a similar way, having four valence electrons helps Tin atoms build sturdy structures when they bond with other elements. It’s not too few, where it’s desperate to grab more, and not too many, where it’s pushing others away. It’s just right!
What makes Tin special is its versatility. Because it has those four valence electrons, it can form different kinds of connections. It can share them, give them away, or even take them (though it prefers sharing and giving!). This makes it a key player in creating all sorts of materials. For instance, when Tin teams up with Lead (another element with its own cool story!), it forms solder. Solder is that stuff that helps connect the tiny parts in your phone, your computer, and all those other amazing gadgets that make our lives easier and more fun. Without Tin and its four pals, your tech wouldn’t be so techy!

It’s also really interesting how Tin behaves. It's a relatively soft metal, which is why it was perfect for making those malleable tin cans. It’s also quite resistant to corrosion, meaning it doesn’t rust easily. That’s a huge deal! Imagine if your food cans just rusted through in a week. Yikes! So, Tin is not just about how many electrons it has; it’s about what those electrons allow it to do. It’s like having a special talent that opens up a world of possibilities.
"The magic of Tin lies in its ability to connect, to build, and to protect, all thanks to those four little powerhouses on its outer shell."
And get this – Tin can even exist in different forms! There’s the shiny, metallic tin we know and love, but there’s also a powdery, grey form. This is another testament to how those valence electrons can be arranged and interacted with, leading to different personalities for the same element. It’s like the same actor playing two completely different roles in a movie – pretty cool, right?

So, next time you see a tin can, or if you ever get curious about how your electronics are put together, remember our friend Tin. Remember its symbol, Sn. And most importantly, remember those four valence electrons. They are the unsung heroes, the tiny connectors that enable Tin to play such an important role in our world. It’s a tiny detail, but it unlocks a universe of possibilities, making Tin a truly special element. Isn't it amazing how much wonder can be found in something as small as an atom?
It’s this sort of hidden complexity, this elegant simplicity in nature, that makes learning about elements like Tin so captivating. You start with a simple question, “How many valence electrons does Tin have?”, and you end up exploring its history, its uses, and the fundamental forces that shape our reality. It’s a journey of discovery, one electron at a time. So, go ahead, let your curiosity lead you to more amazing facts about the elements. The universe is full of them!
