How Many Valence Electrons Do The Halogens Possess

Alright, my fabulous friends, gather 'round! Today, we're diving headfirst into the utterly thrilling, and dare I say, sparkling world of chemistry! Don't worry, no beakers or explosions (unless it's an explosion of knowledge, which is way cooler!). We're talking about a very special family of elements that have a secret superpower, and it all boils down to something called valence electrons. Think of them as the social butterflies of the atom world, always looking to make friends and get along!
Now, you might be thinking, "Valence electrons? What in the name of protons are those?" Imagine your atom is like a tiny, bustling apartment building. The electrons are the residents. And the valence electrons? They're the folks living on the very top floor, the ones with the best view and, most importantly, the ones who are always peeking out the window, ready to interact with the neighbors. They're the ones doing all the mingling, the handshaking, and the sometimes-dramatic deal-making that makes atoms form bonds and create all the amazing stuff we see around us – from the air we breathe to the cookies we devour!
Today, our spotlight is on a particularly sassy and sensational group of elements: the Halogens! These guys are like the rockstars of the periodic table, and boy, do they know how to make an entrance. You've probably heard of some of them before, even if you didn't realize it. We've got Fluorine (the super-clingy, incredibly strong one), Chlorine (the one in your swimming pool and your table salt – don't worry, it's mostly friendly!), Bromine (the reddish-brown liquid that’s a bit mysterious), Iodine (the one that makes your neck healthy, and also stains things like a pro!), and Astatine (the super rare, super radioactive one – we'll keep it brief with Astatine, just know it's the shy, reclusive cousin of the family).
So, here's the big reveal, the moment you've all been waiting for, the question that keeps scientists up at night (or maybe just enjoying a nice cup of tea while pondering): How many of these fabulous valence electrons do our dazzling Halogens possess? Drumroll, please! They all, every single one of them, from the super energetic Fluorine to the elusive Astatine, have precisely... SEVEN! Yes, you heard me right! SEVEN little valence electrons, just hanging out on their top floor, ready for action.
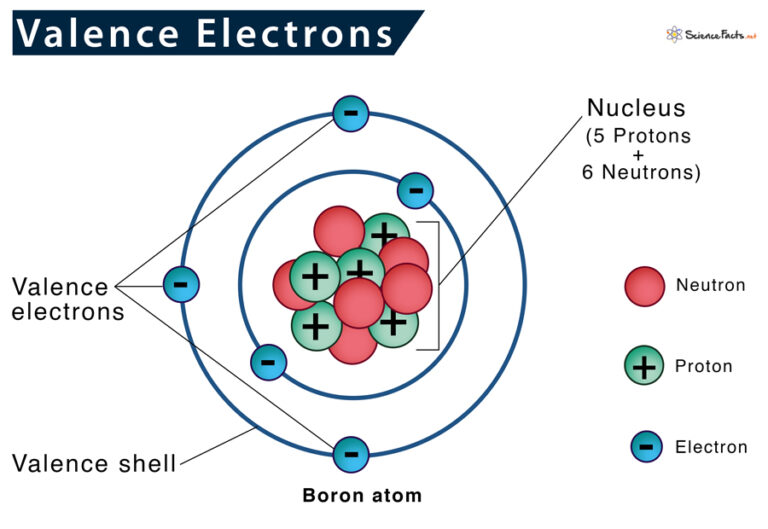
Think about it! Seven is a pretty specific number, right? It's like having seven perfectly placed sprinkles on a cupcake, or seven awesome friends to go on an adventure with. It’s just that perfect amount for what these guys are trying to achieve. They are so close to being perfectly happy, perfectly complete, like a puzzle that just needs one more piece. And that's the beauty of the halogens! They’re always looking for that one extra electron to complete their outer shell. It’s like they’re standing at the door of a party, and there's just enough room for one more person to squeeze in and make everyone really, truly content.
Because they have seven valence electrons, the halogens are like the ultimate bargain hunters or the most enthusiastic matchmakers in the chemical world. They are super eager to grab that one extra electron from any willing partner. They're not greedy, mind you, they just want to feel whole. It’s a bit like wanting that last slice of pizza when everyone else has already had their fill – you just really want it to complete your pizza-eating experience! This intense desire for that one extra electron makes them incredibly reactive. They’re the ones who are always willing to jump into a chemical reaction, to form a bond, to make things happen!

So, when you hear about Chlorine making salt (which is basically Sodium, a very generous element, giving away an electron!), or Fluorine being used to strengthen your teeth (it's working tirelessly with those seven valence electrons to keep your enamel tough!), remember the magic number: SEVEN! It’s their signature, their calling card, their little chemical superpower. They’re a friendly bunch, but they’re also incredibly determined to get that perfect, stable electron configuration. And that’s what makes chemistry so darn exciting – these tiny, invisible forces playing out all around us, all thanks to elements like our fabulous Halogens and their magnificent seven valence electrons!
