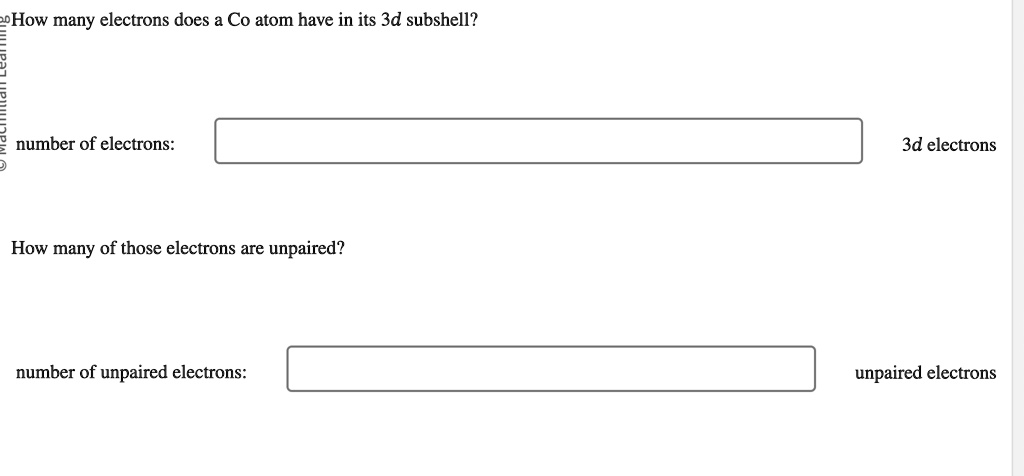
How Many Unpaired Electrons Does Vanadium Have
Hey there, science adventurers! Ever find yourself staring at the periodic table, wondering what all those squiggly lines and letters are about? Well, buckle up, because today we're going on a super-duper fun journey to meet a fascinating element: Vanadium!
Imagine a world where tiny, invisible helpers called electrons are constantly zipping around the center of an atom, like little hummingbirds around a flower. These electrons are the real stars of the show when it comes to how elements behave. And some of these electrons are a bit more… independent than others.
Unpaired Electrons: The Lone Rangers of the Atomic World!
So, what exactly are these "unpaired electrons" we're talking about? Think of electrons as being a bit like socks. Usually, they like to hang out in pairs, cozy and content. But sometimes, just sometimes, one sock gets left behind. That's our unpaired electron – a solo adventurer, ready for anything!
These lone rangers are super important because they're the ones that get to do all the exciting interacting. They're like the social butterflies of the atom, always looking for a friend to pair up with. When an atom has unpaired electrons, it's like it's waving a little flag saying, "Hey, I'm ready to mingle!"
Now, some elements are totally boring. They're like a closet full of perfectly matched pairs of socks. No surprises there! But Vanadium? Oh no, Vanadium is a different story altogether. It's got a few of those adventurous, unpaired electrons just waiting to make some chemistry happen.
Let's Meet the Mighty Vanadium!
Vanadium, with its atomic number 23, is a metal that’s pretty strong and can be found in all sorts of cool places. It’s not as famous as gold or as colorful as a rainbow, but it plays a huge role in making things better and stronger.
Think about your super-strong bicycle frame or the steel that holds up skyscrapers. Yep, chances are good that a little bit of Vanadium is happily contributing to all that toughness. It’s like the secret ingredient that makes things extra resilient and ready to take on the world.
But to understand why Vanadium is so good at making things strong, we need to peek inside its atomic structure. And that’s where our unpaired electrons come in, ready to perform their atomic ballet.
The Electron Dance: A Peek Inside Vanadium
Atoms have different "shells" or "energy levels" where electrons hang out. Imagine these like different floors in a very tall apartment building. The electrons closest to the center (the nucleus) are on the lower floors, and they're usually all paired up and happy.
But then there are the outer floors, and things get a bit more interesting. In Vanadium, the electrons on its outermost floors are not all neatly paired up. They’re a bit like a group of friends at a party, some are in pairs, but others are looking around for someone new to talk to.

The key to Vanadium's special abilities lies in its electron configuration. It’s not just one or two unpaired electrons; Vanadium can have a few! This is what makes it so versatile and useful in so many applications.
So, How Many Unpaired Electrons Does Vanadium Have? The Big Reveal!
Alright, drumroll please! The exact number of unpaired electrons an atom of Vanadium has can actually vary depending on how it's feeling, or rather, how it's bonded with other atoms. It’s like a chameleon that can change its colors based on its surroundings!
In its most common and stable form, Vanadium usually has three unpaired electrons. Three! That’s like having three extra best friends at your party, all looking for someone to do a dance with. This makes Vanadium incredibly ready to form bonds and create new, exciting compounds.
Imagine Vanadium as a talented artist with a palette of three extra brushes, ready to paint a masterpiece of a chemical bond!
How many valence electrons does vanadium have?
Sometimes, depending on the situation, Vanadium can even show off with one unpaired electron or even two unpaired electrons. It’s like it has multiple personality settings, all focused on making cool chemical connections.
This ability to have different numbers of unpaired electrons is what gives Vanadium its superpower of oxidation states. Don't let the fancy word scare you! It just means Vanadium can play nicely with many different kinds of atoms, forming a variety of compounds with different properties.
Why This Matters (Besides Being Super Cool!)
Having these unpaired electrons isn't just a neat party trick for Vanadium. It’s the reason why it’s so useful in everyday life. Those three unpaired electrons are like tiny magnets, eager to attract and hold onto other atoms.
When Vanadium forms bonds, it makes materials incredibly strong. Think of alloys, which are mixtures of metals. Adding a little Vanadium to steel, for example, makes it much harder and more resistant to wear and tear. It’s like giving your ordinary steel a superhero cape!

Even in the world of catalysts, which are substances that speed up chemical reactions, Vanadium is a superstar. Those unpaired electrons help it grab onto molecules and rearrange them, making industrial processes more efficient. It's like having a super-fast helper in a busy factory.
Vanadium: A Tiny Element with a Huge Impact
So, the next time you see something made of strong metal, or hear about a new catalytic process, give a little nod to Vanadium. It’s a humble element, often working behind the scenes, but its amazing ability to have three unpaired electrons (and sometimes other numbers!) makes it a true workhorse of chemistry.
It's a reminder that even the smallest parts of the universe have incredible stories to tell. And sometimes, those stories involve a few lone rangers, ready to make a big difference. Vanadium is definitely one of those stories, and it's a blast to learn about!
Isn't it fascinating how something so tiny can have such a massive impact? The world of atoms is full of these amazing surprises, and Vanadium is just one glowing example. Keep exploring, and you’ll find even more wonders waiting to be discovered!

