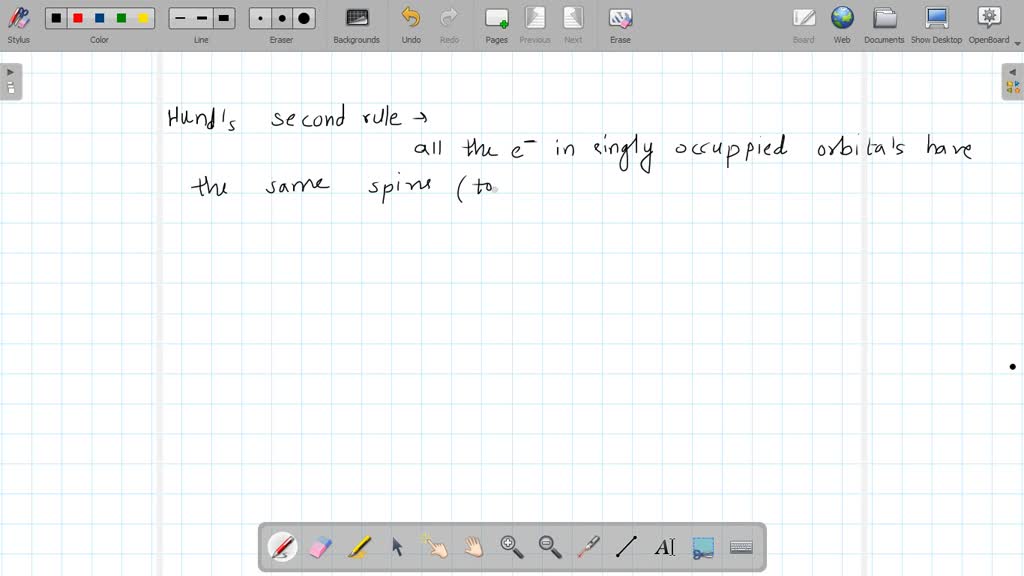
How Many Unpaired Electrons Does Nitrogen Have

Let’s talk about electrons. Specifically, the ones that are a little bit lonely. You know, the unpaired ones. They’re the rebels of the atomic world. And today, we’re zeroing in on a rather fascinating element: Nitrogen.
Now, some people might tell you that Nitrogen is pretty straightforward. A standard atom, doing its atom thing. But I have an unpopular opinion. I think Nitrogen is secretly a bit of a show-off.
Why a show-off, you ask? Well, it’s all about those elusive unpaired electrons. They’re like the guest who shows up without an invitation but brings the best snacks. And Nitrogen? It’s got a few of these uninvited guests hanging around.
So, how many unpaired electrons does this nitrogen wonder have? Drumroll, please! It’s three.
Yes, three! Think of it like this: Nitrogen is at a party. It’s got a few friends, but three of them are just… on their own. Maybe they’re deep in conversation, or maybe they’re just really enjoying their own space. We can only speculate.
It’s not exactly a crowd, is it? Just three. It’s like having three single socks in your laundry. You know they should have a partner, but for now, they’re just out there, existing independently.
And that, my friends, is the charm of Nitrogen. It’s not overly complicated. It’s not a massive electron party. It’s just a select few rebels.
The Grand Unveiling
Now, I know some of you might be thinking, "But why three?" You might be picturing some complex diagram with spinning electrons and energy levels. And while that’s all scientifically accurate, it’s also a bit of a snooze-fest for us regular folks.

Let’s stick with the relatable. Nitrogen, in its most common form, has a certain electron arrangement. Imagine a cozy little apartment for these electrons. Most of the rooms are shared, with two electrons happily cohabiting.
But then there are these three special rooms. Each of these rooms has only one electron. They’re like the single occupancy suites. And that’s where our unpaired electrons reside.
It’s a neat little number, isn’t it? Three. Not two, not four. Exactly three. It’s a number that suggests a certain… purpose. A readiness for something new.
These unpaired electrons are like little hands reaching out. They're looking for something to hold onto. They're eager to form new bonds and create something bigger and better.
Think of it as Nitrogen saying, "Hey, I've got these three amazing electrons, ready to mingle! Who wants to team up?" It’s the atomic equivalent of a confident wave.
And because of these three unpaired electrons, Nitrogen is incredibly important. It’s a building block for so many things we rely on. From the air we breathe to the DNA in our bodies, Nitrogen is there, thanks to its electron arrangement.
The Mystery of the Missing Partner
It’s this very state of having unpaired electrons that makes Nitrogen so reactive. It’s not content to just sit there. It’s actively looking to complete its electron shells.
So, while other atoms might be perfectly happy in their paired-up existence, Nitrogen is out there, actively pursuing connections. It’s the extrovert of the periodic table, in a way.
And let’s be honest, who doesn’t admire a little bit of initiative? Those three unpaired electrons are its secret weapon. Its invitation to the chemical dance.
It’s a beautiful simplicity, really. Not a chaotic free-for-all of electrons, but a distinct set of three ready for action. It's the perfect amount of "available."
Imagine you’re setting up a board game. You have a few pieces that are just waiting for their turn. They’re not part of a pair yet, but they’re essential for the game to proceed. That’s our Nitrogen.

And the fact that it’s always three just adds to its enigmatic appeal. It's a consistent number, a reliable characteristic. It's the little black dress of atomic properties – always in style, always ready.
So, the next time you think about Nitrogen, don't just think of it as a gas. Think of it as an atom with three strategically placed, unpaired electrons, just waiting for their moment to shine. It's a little bit of atomic individuality.
It’s not a flaw; it’s a feature. Those three unpaired electrons are what make Nitrogen so dynamic and essential. They are the reason it can form so many different and vital compounds.
They are the little sparks of reactivity that drive chemical processes. They are the reason why Nitrogen is a star player in the grand symphony of chemistry.
So, there you have it. The wonderfully simple, yet profoundly important answer: Nitrogen has three unpaired electrons. It’s a fact that’s both scientifically accurate and, dare I say, a little bit charmingly rebellious.
Unpaired electrons: they’re not just a scientific concept; they’re a metaphor for readiness, for seeking connection, for being just the right amount of available. And Nitrogen, with its solid three, embodies this perfectly. It's not trying too hard; it's just naturally, wonderfully, open for business.
Nitrogen, the element of three: a testament to the power of just enough. Just enough electrons, just enough excitement, just enough to make the world go 'round (or at least, build the molecules that do).
It's a beautiful thing when science can be this straightforward and, frankly, a little bit cute. Three unpaired electrons. It’s a number you can’t help but smile at.
And that’s why, in my book, Nitrogen is a quiet superstar. Not because it’s flashy, but because it has that perfectly balanced, slightly quirky electron arrangement. Those three lonely hearts are its superpower.
So, next time you encounter Nitrogen, give it a nod. A silent acknowledgement of its three unpaired electrons. They’re the unsung heroes of so many molecular stories. They are the reason Nitrogen is so darn good at what it does.
It’s like a perfectly portioned appetizer. Just enough to whet your appetite, just enough to be intriguing, just enough to make you want more. That’s the magic of Nitrogen’s three unpaired electrons.
And that, my friends, is how many unpaired electrons Nitrogen has: three. A number that’s as fundamental as it is fabulous.
