How Many Unpaired Electrons Are In The Titanium Atom

Alright, gather ‘round, you beautiful humans! Today, we’re diving into the electrifying world of… drumroll please… titanium! Yeah, I know, sounds a bit like a superhero's secret identity, right? But this metal is way cooler than your average cape-wearer. We’re not talking about the stuff that makes your pots and pans all shiny and indestructible (though it does do that). We’re talking about the teeny-tiny, subatomic playground where electrons go to party. And the burning question, the one that keeps scientists up at night, the riddle that’s probably plastered on every coffee shop napkin in Geneva is: How many unpaired electrons does a titanium atom have?
Now, before you start picturing a bunch of electrons doing the cha-cha with no dance partners, let’s clarify. An atom is basically a miniature solar system, with a sun (the nucleus) and planets (electrons) whizzing around. Electrons usually like to hang out in pairs, like a couple on a romantic getaway. They’ve got opposite spins, which is the atomic equivalent of holding hands and sharing a tiny, electric croissant. But sometimes, a lone ranger electron decides it’s had enough of the paired life. It wants to be a solo artist, a free spirit, an… unpaired electron. And these little rebels? They’re the secret sauce behind a lot of titanium’s awesome powers. Think of them as the wildcard in a deck of cards – they can change the whole game!
So, how many of these adventurous electrons are strutting their stuff in a titanium atom? To answer this, we gotta get a little nerdy. But don’t worry, I promise no pop quizzes. We need to look at the atom’s “electron configuration.” This is basically the seating chart for our electron party. It tells us where each electron likes to crash and who they’re bunking with. Titanium, bless its metallic heart, has 22 electrons. That’s a decent number for an atom, not too shabby, not too overwhelming. Imagine a small, bustling party. Not a rave, more like a friendly get-together at your aunt’s place.
The first few electrons are pretty well-behaved. They fill up the innermost shells, like the VIP section of a club. They’re all paired up, snuggled in their little orbital apartments, probably discussing the latest electron gossip. Then, things get a little more interesting as we move to the outer shells. This is where the action happens. These outer electrons are the ones that get to mingle with other atoms, forming bonds and making things… well, stuff. They’re the social butterflies of the atomic world.
Now, for titanium, the electron configuration looks something like this: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d². I know, I know, it looks like a secret code from a spy movie. But let’s break it down. The numbers and letters tell us which "shell" (the number) and "subshell" (the letter) the electrons are in, and the superscript tells us how many electrons are in that particular subshell. So, 1s² means there are 2 electrons in the 1s subshell. All good, all paired up. 2s² same story. 2p⁶? Six electrons, all nicely paired. You get the picture.
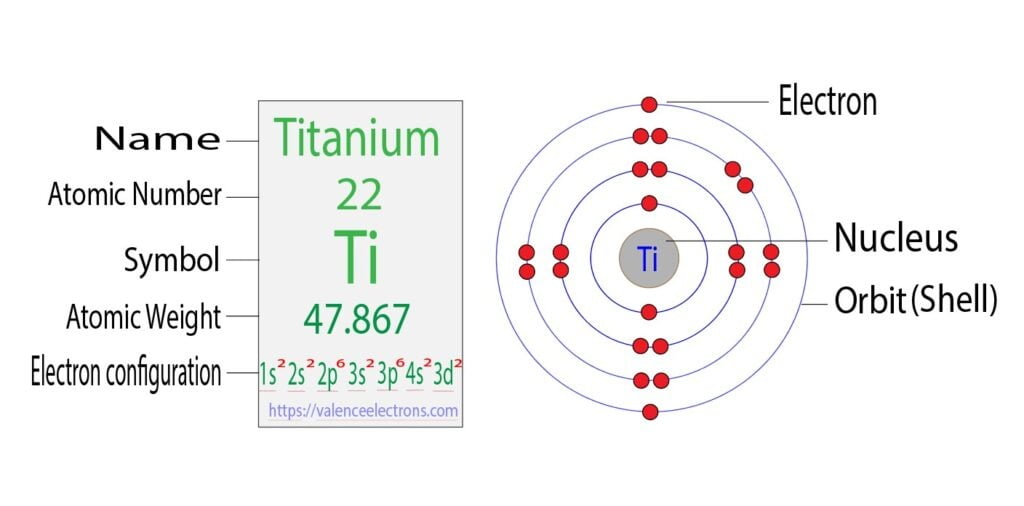
But then we hit the 3d subshell. This is where titanium starts to show its true colors, or rather, its unpaired electrons. The 3d subshell can hold up to 10 electrons, but in titanium, we only have 2 electrons hanging out there. Now, here’s the kicker: in the 3d subshell, there are five individual orbital “rooms.” Think of them as tiny studios. Electrons, bless their little quantum hearts, like to spread out before they have to pair up. It’s like when you’re at a party and you have your pick of empty chairs – you’re not going to immediately squeeze into a seat next to someone unless you have to. Electrons are a bit like that.
So, those two electrons in the 3d subshell? They’re not going to jump into the same room and become a couple right away. Nope. They’re going to take up residence in two different orbital rooms. They’ll be roommates, sure, but they’re not going to be holding hands and singing love songs. They’ll each have their own little space, their own personal bubble. And because they’re in separate rooms and have the same spin (which is a whole other mind-bending topic we won’t get into today, unless you bring donuts), they are considered… unpaired.

So, the grand finale, the answer to our burning question: a neutral titanium atom has two unpaired electrons. Ta-da! It’s not a huge number, like a dozen or a score, but these two little rebels pack a punch. These unpaired electrons are what make titanium so special. They’re the reason it can form such strong bonds with other elements, making it incredibly useful in everything from airplanes to medical implants. They’re like the energetic friends who always convince you to try something new, even if it seems a little scary at first.
Think about it! This humble metal, found in the earth’s crust, has these little electron dynamos that allow it to be super strong, lightweight, and resistant to corrosion. It’s like finding out your quiet neighbor secretly knows how to juggle chainsaws. Surprising, right? And it’s all thanks to those two lone wolves in the 3d subshell. They’re the secret ingredient, the X-factor, the reason titanium isn’t just another lump of metal.
So, next time you see something made of titanium, give it a little nod of appreciation. You’ll know that hidden within its metallic exterior is a tiny, subatomic party with a couple of daring electrons on the dance floor, rocking their unpaired status. And that, my friends, is pretty darn cool. Now, who’s up for another round of existential electron pondering… with coffee, of course?
