How Many Unpaired Electrons Are In The Oxygen Atom
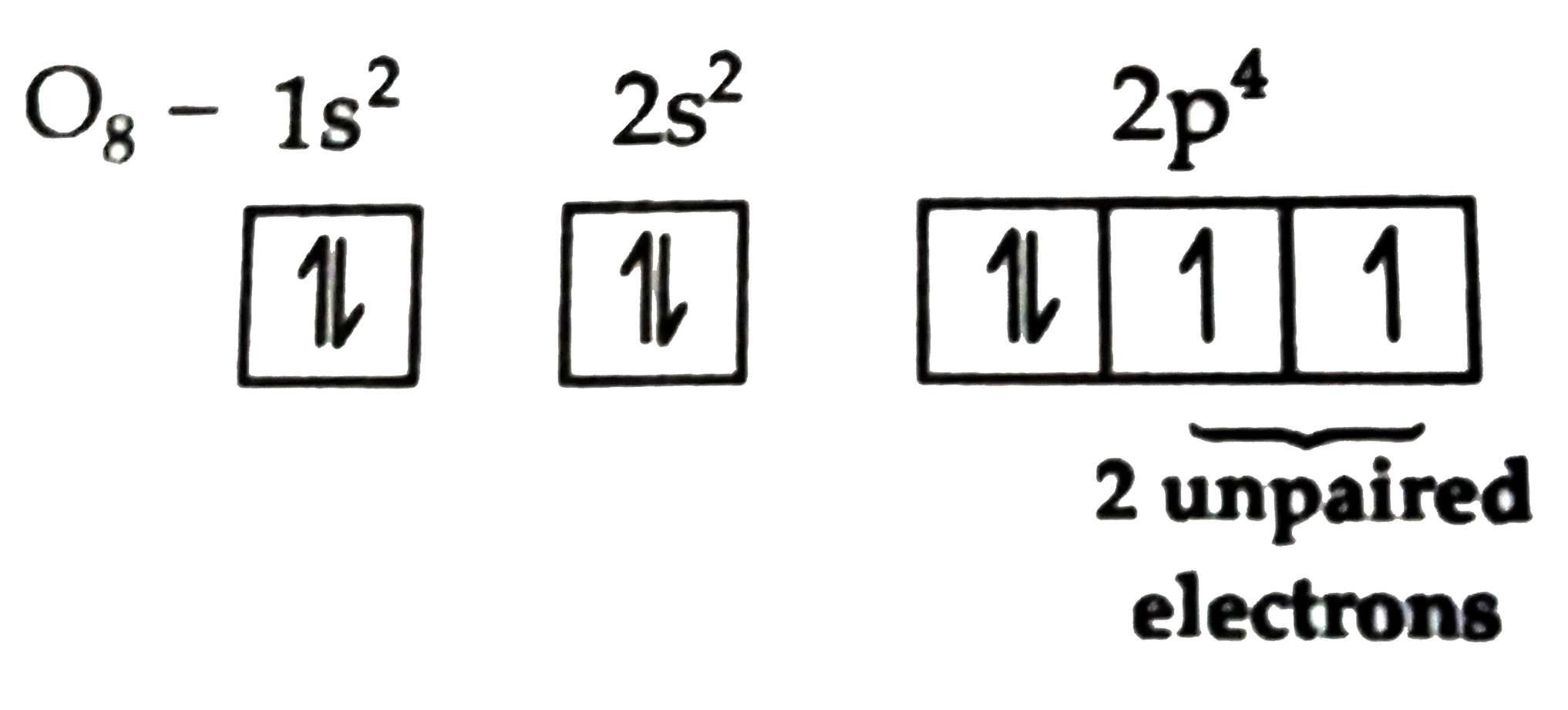
Hey there, fellow wanderers of the modern world! Ever found yourself staring at a perfectly brewed cup of coffee, or perhaps taking a deep breath of that crisp morning air, and wondered about the tiny, invisible forces at play? We’re talking about the unsung heroes of our everyday existence, the building blocks that make everything, well, everything. Today, we’re diving into the surprisingly chill world of atoms, specifically, our good ol’ friend, oxygen.
Now, before you start picturing complex chemistry textbooks and stuffy lecture halls, let's put on our comfy slippers. We're not here to cram for an exam. We're here to explore, to discover, and to find the fun in the fundamental. Think of it like a backstage pass to the universe. And our backstage pass today? It’s all about a little something called unpaired electrons in an oxygen atom.
You might be thinking, “Electrons? Unpaired? Is this going to get complicated?” Absolutely not! We're keeping it breezy, like a Sunday afternoon picnic. So grab your favorite beverage, lean back, and let’s unravel this cosmic little mystery together.
Oxygen: The Breath of Life, But What Makes It Tick?
We all know oxygen is pretty crucial, right? Without it, our planet would be a very different, and much quieter, place. It’s the lifeblood of our bodies, the fuel for our fires, and the silent partner in so many of the chemical reactions that keep our world spinning. But have you ever stopped to consider why oxygen is so darn good at doing all these things? It all boils down to its atomic personality, and a big part of that personality is tied to its electrons.
At its core, every atom is like a miniature solar system, with a nucleus at the center and electrons zipping around in orbits, or shells. These electrons are like tiny, energetic particles that dictate how an atom interacts with others. They’re the social butterflies, the matchmakers, the ones who decide whether an atom is going to be shy and keep to itself, or bold and jump into a reaction.
Now, electrons don't just hang out randomly. They like to pair up, like best buds chilling on the couch. In fact, most electrons prefer to be in pairs within their orbital shells. This pairing makes them feel stable and content. Think of it like a perfectly matched pair of socks – they’re happier together, fulfilling their sock destiny.
The Curious Case of the Unpaired Electron
But here’s where things get interesting. Not all electrons manage to find a dance partner. Some are loners, flying solo. These are our unpaired electrons. And in the case of an oxygen atom, this little quirk is a huge deal.
Let’s do a quick peek at oxygen’s electron situation. An oxygen atom, in its most common form, has 8 electrons. These electrons are arranged in different energy levels or shells. The outermost shell, which is the one that really matters when it comes to bonding and reactions, is called the valence shell. Oxygen’s valence shell has 6 electrons.
Now, these valence shells have specific capacities for electrons. The outermost shell of oxygen can hold up to 8 electrons to achieve a state of maximum stability, often called a "full octet." Imagine a really popular party; everyone wants to be in the room where all the cool kids are, which is the full octet. Oxygen, with its 6 valence electrons, is like a party host who’s still looking for two more guests to make the party perfect.
So, these 6 electrons in the outer shell arrange themselves. They try to pair up as much as possible. But here's the kicker: the way these electrons are arranged in their orbitals means that even when the shell isn't full, there’s a tendency for them to occupy specific sub-orbitals. And when oxygen tries to accommodate its 6 valence electrons into these sub-orbitals, it ends up with a certain configuration.
Ready for the big reveal? A standard oxygen atom has two unpaired electrons. Yep, two little electrons rocking the solo life. Think of them as the intrepid explorers of the atomic world, always ready for an adventure. This unpaired status is what gives oxygen its super reactive personality. It’s like having two free agents always looking for a team to join.
Why Should We Care About These Lone Wolves?
Okay, so oxygen has two unpaired electrons. Big deal, right? Well, yes, it’s a very big deal. These unpaired electrons are the reason oxygen is so keen on forming bonds. They’re constantly looking for another electron (or two!) to complete their pairs and achieve that coveted stable state.

This desire to pair up is what makes oxygen a powerful oxidizing agent. It readily "steals" electrons from other atoms or molecules. This process, called oxidation, is fundamental to so many things we experience daily. Think about:
- Rusting: That classic orange-brown film on your old bicycle? That’s iron reacting with oxygen. The oxygen, with its eager unpaired electrons, pulls electrons from the iron, causing it to break down. It’s a slow dance of electron exchange.
- Burning: Fire is essentially a rapid oxidation process. When you burn wood or gas, oxygen atoms are enthusiastically taking electrons, releasing energy in the form of heat and light. It’s the ultimate team-up for an energetic show!
- Our Metabolism: Inside your body, a complex dance of oxidation is happening constantly. Your cells use oxygen to break down food molecules, releasing the energy you need to think, move, and even just exist. It's a highly controlled, life-sustaining chemical reaction, all thanks to those eager electrons.
It’s kind of like how in a group project, the person who finishes their part early is always looking for something else to contribute to, or someone to collaborate with. Those unpaired electrons are oxygen’s way of saying, "Hey, I’ve got some energy to share, and I’m ready to team up!"
The Science Behind the Spark: A Little Deeper Dive (Optional!)
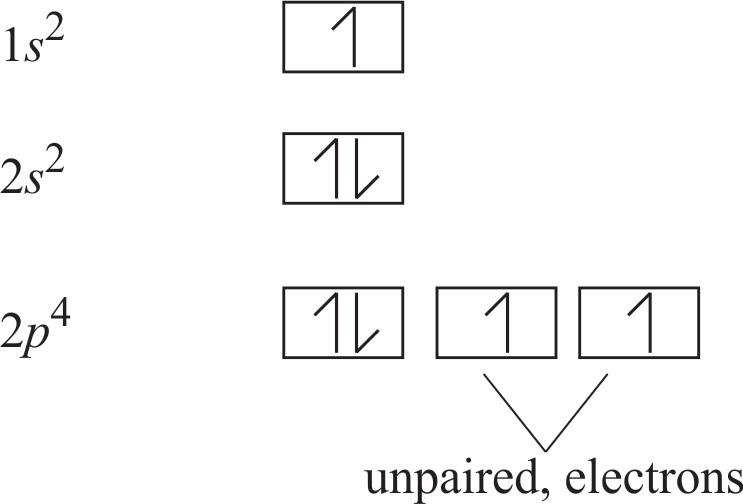
For those who love a sprinkle more detail, let’s touch on the concept of orbitals. Electrons don't orbit like planets; they exist in regions of probability called orbitals. Oxygen has s orbitals and p orbitals. The outermost shell has one 2s orbital and three 2p orbitals. Each orbital can hold a maximum of two electrons.
Oxygen’s 6 valence electrons fill up these orbitals. The 2s orbital is filled with two paired electrons. Then, the three 2p orbitals get filled. According to Hund's Rule, electrons will individually occupy each orbital within a subshell before doubling up. So, the first three electrons go into separate 2p orbitals, each with one electron. The remaining three electrons then start pairing up. This leaves two of the 2p orbitals with paired electrons, and one 2p orbital with a single, unpaired electron. Wait, I said two unpaired electrons before. Let me re-evaluate.
Ah, a slight correction here! When we talk about the ground state of a neutral oxygen atom (O₂ molecule), it's a bit more complex and fascinating. The molecular orbital theory is needed for the O₂ molecule. In the diatomic oxygen molecule (O₂), which is what we breathe, the electrons are arranged in molecular orbitals. And in this common form of oxygen, there are indeed two unpaired electrons.
This arrangement is a bit unusual in the world of diatomic molecules, where most expect all electrons to be paired. But it's precisely this configuration, with its unpaired electrons, that makes O₂ so reactive and essential for life. It's a testament to how nature often finds elegant solutions in what might seem like an imperfect arrangement.
Think of it like a band. Most bands have all their members playing harmoniously. But an oxygen molecule is like a band with two members who are just a little more spontaneous, a little more willing to improvise, and that's what gives their music (their chemical reactions) its unique energy and power.
Fun Facts and Cultural Connections
Did you know that the word "oxygen" comes from Greek words meaning "acid-former"? This was based on an early, incorrect belief that oxygen was necessary for all acids. Science, like life, is a journey of discovery and sometimes, a few missteps!
In popular culture, oxygen is often portrayed as the pure, life-giving element. Think of those dramatic scenes in movies where a character is gasping for air, or the pristine landscapes depicted as having "pure oxygen." It’s a symbol of vitality and health. Even in wellness culture, the idea of "oxygenating" the body is popular, though it usually refers to improving circulation and respiration rather than literally adding more oxygen molecules.
And for the sci-fi fans out there, the very premise of living on other planets often hinges on the presence or absence of oxygen. Whether it's terraforming Mars or finding alien life, the availability of this simple element is a central plot point. It underscores its fundamental importance to our understanding of life.
Practical Tips for an "Oxygenated" Life
While we can't directly control the unpaired electrons in oxygen atoms, we can certainly appreciate and nurture their effects in our lives. Here are a few easy-going ways to embrace the power of oxygen:
- Get Outdoors: This is the most obvious one, but it bears repeating. Spend time in nature. Go for walks in the park, hikes in the mountains, or simply sit by the sea. Fresh air is packed with oxygen, and your body will thank you. It’s the ultimate free spa treatment.
- Deep Breathing Exercises: When you take a deep, conscious breath, you're optimizing your body's oxygen intake. Try a few minutes of mindful breathing each day. It can reduce stress, improve focus, and even boost your mood. Think of it as a mini-vacation for your lungs.
- Stay Hydrated: Water is essential for transporting oxygen throughout your body. So, keep that water bottle handy and sip throughout the day. Your cells will be happier, and so will you.
- Regular Exercise: Physical activity increases your breathing rate and your body's demand for oxygen. It strengthens your respiratory system, making it more efficient at taking in and utilizing oxygen. Find an activity you enjoy, whether it’s dancing, swimming, or cycling.
- Support Green Spaces: Plants are the Earth's oxygen factories! Support local parks, plant trees, or even start a small herb garden. By nurturing greenery, you’re contributing to a healthier oxygen supply for everyone.
These are not radical life changes; they are small, sustainable habits that can significantly enhance your well-being and your appreciation for the element that sustains us.
A Moment of Reflection
So, the next time you take a breath, perhaps you'll pause for a fleeting moment and consider the incredible atom that is oxygen. Those two tiny, unpaired electrons, dancing on the edge of stability, are constantly at work, enabling rust, fire, and the very life within you. They are a beautiful illustration of how even the smallest, seemingly insignificant details in the universe can have profound and far-reaching consequences.
It’s a humbling thought, isn’t it? That the air we breathe, the energy we feel, and the very existence of so many processes around us are driven by the energetic quests of these solitary electrons. It reminds us that complexity and wonder can be found in the most ordinary of things. Just like the simple act of breathing can be a gateway to understanding the intricate dance of the universe.
Embrace the magic of the everyday. Find joy in the fundamental. And remember, even the most powerful forces often start with a single, unpaired entity looking for its place in the world.
