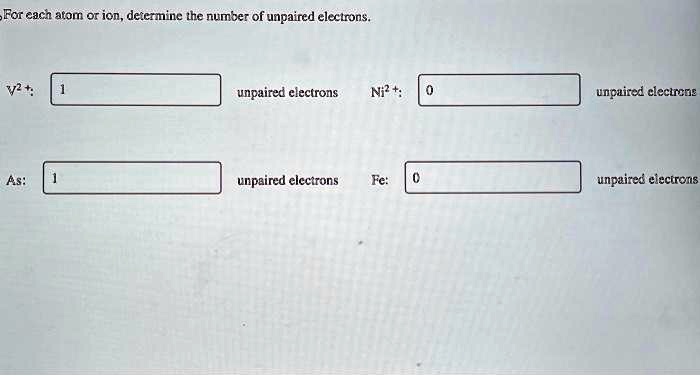
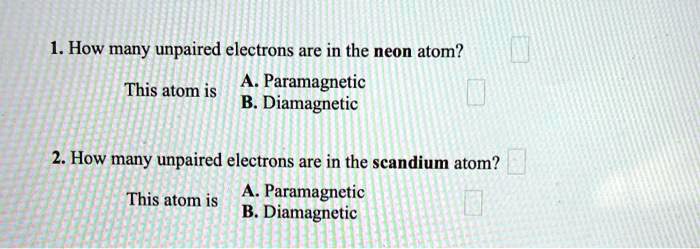
How Many Unpaired Electrons Are In The Neon Atom

Ever stared at one of those fancy neon signs, the kind that practically glows with personality, and wondered what’s really going on inside that tube? It’s not just magic, folks. It’s all about the tiny, invisible world of atoms, and today we’re going to peek into the life of a neon atom, specifically its quest for… well, for not being alone. Think of it like this: if atoms had social lives, neon would be the person who’s always got their friend group perfectly sorted, never needing to awkwardly ask someone to join a game of charades.
So, the burning question: how many unpaired electrons are in a neon atom? Let’s break it down, but keep it light, like a fluffy cloud of helium, not a heavy lead weight. We’re not talking rocket science here, just good old atomic gossip.
First off, what even is an electron? Imagine them as super, super tiny, hyperactive little kids. They’re always zipping around the nucleus of an atom, which is like the atom’s cool parent, minding its own business. These electrons love to pair up. They’re like those married couples who finish each other’s sentences, or maybe those best friends who always show up in matching outfits. It’s all about harmony and stability. When electrons are paired up, they’re happy. They’re settled. They’ve found their atom-mate.
Now, neon. Neon is a bit of a celebrity in the atomic world. It’s part of a special club called the noble gases. You know how some people are just naturally popular and don’t have to try? That’s neon. It’s got this inherent chill vibe. It’s the life of the party without even trying to be. It’s the person who can walk into any room and instantly feel at home, never needing to awkwardly hover by the snack table.
The reason neon is so cool and collected, so unbothered, is because of its electrons. Atoms, you see, have these different "shells" or "energy levels" where electrons hang out. Think of them like floors in a building. The first floor is the closest to the nucleus (the atom’s core), and it can only hold a couple of electrons. The next floor up can hold more, and so on. It’s like a tiny, atomic apartment building, and everyone wants a perfectly filled apartment.
For neon, its electron setup is just chef’s kiss. It’s got its first floor nicely filled with two electrons. Then, it’s got its second floor, which is the outermost one, and it’s jam-packed with eight electrons. Eight! That’s the magic number for this outer shell. It’s like having the perfect number of guests for a board game – everyone has a spot, no one’s left standing awkwardly. This full outer shell is what makes neon a noble gas. It’s achieved atomic nirvana. It’s like reaching the top of the mountain after a long hike, finally able to just sit back and enjoy the view without any more climbing.
So, if all those electrons in the outer shell are paired up, and it’s a full house, what does that mean for unpaired electrons? An unpaired electron is like that one friend who’s always single at a wedding. They’re not sad, necessarily, but they’re also not part of a matching pair. They’re looking for a partner, ready to mingle, perhaps to form a new bond. They have a little bit of restless energy, a desire to connect.
In the case of neon, because its outer shell is perfectly filled with eight electrons, and these electrons like to pair up in twos, all eight of those electrons have found a buddy. They’re all comfortably nestled in their little electron couples. There are no lonely electrons hanging around, no odd numbers looking for a dance partner. It’s like a ballroom where every single person is already paired up. No awkward introductions needed, no one left on the sidelines.

Therefore, the answer to our burning question, drumroll please… a neon atom has zero unpaired electrons. Absolutely, positively, none. Nada. Zilch. It’s like asking how many single socks you have when all your socks are perfectly paired up in the drawer. You’d just shrug and say, "Nope, all good here!"
This lack of unpaired electrons is the secret sauce behind why neon is so… well, so neon. These noble gases are notoriously unreactive. They’re the chemists’ equivalent of saying, "I'm good, thanks." They don't really want to bond with other atoms. Why would they? They’re already perfectly content with their electron situation. It’s like a person who’s just bought their dream house, furnished it perfectly, and now they’re just chilling, not looking to move or share their space. They’ve reached peak contentment.
Think about it this way: when atoms are looking to bond, it’s often because they need to gain, lose, or share electrons to achieve that stable, full outer shell. It’s like a group of people trying to complete a puzzle – they need all the pieces to make the picture whole. But neon? Neon’s puzzle is already complete. It’s got all its pieces, perfectly placed. It’s not looking to trade pieces, borrow pieces, or give away pieces. It’s just… complete.
This is why neon gas is used in those iconic signs. When you zap neon gas with electricity, it gets excited. The electrons get all jazzed up, jump to higher energy levels, and then, when they calm back down, they release that energy as light. And because neon atoms are so stable and don’t want to react with anything else in the tube, you get that pure, vibrant neon glow. It’s the atom equivalent of a perfectly choreographed dance – every move is precise, and there are no fumbles.
Contrast this with an atom that does have unpaired electrons. Imagine oxygen. Oxygen has two unpaired electrons. This makes oxygen a bit more of a social butterfly, more inclined to mingle and form bonds. It’s the reason why we can breathe oxygen – it readily bonds with other atoms, including those in our bodies, to form essential molecules. Oxygen is like that enthusiastic friend who’s always up for an adventure, ready to jump into anything.
Or consider hydrogen, with its single, lonely electron. Hydrogen is like the ultimate bachelor, always looking to pair up. It’s the foundation of so many compounds, like water (H₂O), where two hydrogens are happy to share their lone electron with oxygen. They’re the ultimate team players, always willing to contribute to something bigger.

But neon? Nah. Neon is the ultimate introvert who’s also somehow the most popular person at the party. It’s the person who shows up, looks amazing, and then just stands in the corner, perfectly content, and everyone else is just drawn to its aura of self-sufficiency. It doesn’t need to do anything to be admired; its very existence is enough.
So, the next time you see a vibrant neon sign, remember the quiet, self-contained atom within. It’s not performing some complex chemical ballet; it’s just being its noble self, with its perfectly paired-up electrons, a testament to atomic contentment. It's a reminder that sometimes, being perfectly complete is the coolest thing you can be. It’s the atomic equivalent of wearing a perfectly tailored suit – effortlessly stylish and utterly unconcerned with anyone else’s opinion.
And that, my friends, is the simple, elegant truth about neon. Zero unpaired electrons. It’s the atomic version of a mic drop. It’s the ultimate "I've arrived" statement, written in the language of electron shells. So go forth, and appreciate the stability, the coolness, the nobility of neon. It’s a gas, sure, but it’s one that’s truly got it all figured out. It’s the calm in the chemical storm, the steady eddy in the flow of atomic reactions. And it’s all thanks to those perfectly happy, paired-up electrons. No awkward singles’ mixers in the neon atom, thank you very much. Just pure, unadulterated, atomic bliss. It’s the atomic equivalent of a perfectly brewed cup of tea – warm, comforting, and completely self-sufficient.
