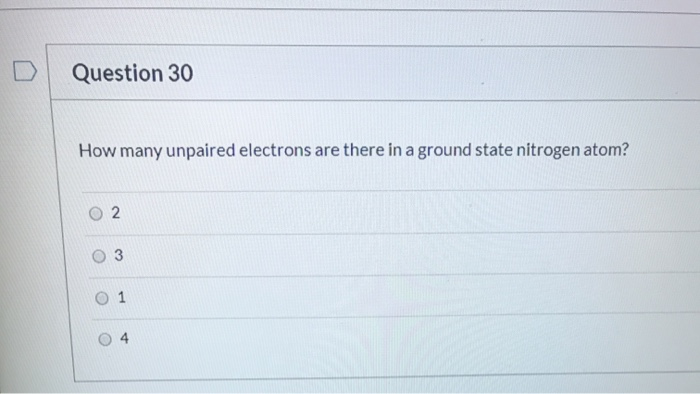
How Many Unpaired Electrons Are In A Nitrogen Atom
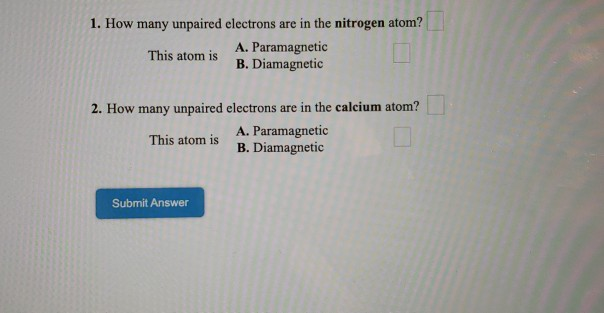
Okay, let's talk about something that might sound a bit… well, nerdy. But stick with me! We're diving into the teeny-tiny world of atoms. Specifically, we're going to play a little guessing game. The question on everyone's mind, or at least on mine during a quiet Tuesday afternoon, is: How many unpaired electrons does a Nitrogen atom have hiding in its shell?
Now, I know what you're thinking. "Unpaired electrons? Is this a dating app for atoms?" Kind of! But way less awkward. Imagine electrons as little rebels. They like to hang out in pairs, like best buddies. But sometimes, they end up going solo. And those solo acts? That's what we call unpaired electrons. And our focus today is the mighty Nitrogen.
Let's get a little visual. Think of an atom like a tiny apartment building. The nucleus is the landlord, always chilling in the middle. Then you have different floors, or "shells," where the electrons live. These shells have different numbers of rooms, called "orbitals." And in each orbital, only two electrons can cram in, no more. It's like a tiny hotel with strict occupancy rules.
So, where does Nitrogen fit into all this? Nitrogen is element number seven on the periodic table. That means it has seven protons in its nucleus, and usually, seven electrons buzzing around. That's our magic number: seven!
Now, these seven electrons aren't just scattered randomly. They fill up those orbital "rooms" in a specific order. It's a bit like the early bird gets the worm, or in this case, the inner shells get filled first. The first shell, the one closest to the landlord, is small. It only has one orbital, a cozy little studio apartment. This orbital can hold a maximum of two electrons. So, the first two electrons of our Nitrogen atom are going to team up and pair up in this first shell. They're like the perfect couple, happy as can be.
That leaves us with five more electrons to find a home for. These five will move to the next shell, the second shell. This shell is a bit bigger. It has more "rooms." It has one "s" orbital (another studio apartment) and three "p" orbitals (like little one-bedroom flats). So, in total, the second shell has four orbitals.
Here's where it gets interesting, and where our unpaired electrons might start to emerge. The "s" orbital in the second shell will get filled first. Two more electrons will pair up here. So now we have our first two electrons happily paired in the first shell, and another two electrons happily paired in the "s" orbital of the second shell. That's four electrons accounted for, all in pairs.
We still have three electrons left! These three are going to take up residence in the three "p" orbitals of the second shell. Now, here's the quirky rule of "p" orbitals, and this is where the fun really begins. Electrons, when they enter a set of "p" orbitals, are a little bit like people entering a room with multiple identical chairs. They tend to spread out. They're a bit shy at first, or maybe just want their own personal space.
So, each of these three remaining electrons will occupy one of the three "p" orbitals. They're not going to double up and pair up immediately. They're going to take their own seats. Think of it as them saying, "Nah, I'm good over here."
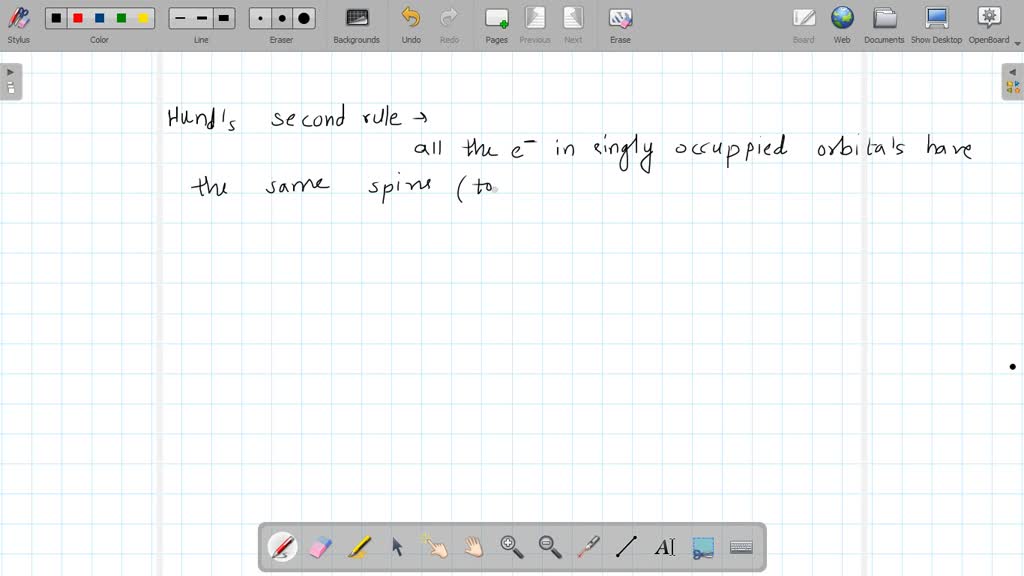
And that, my friends, is the moment of truth! We have three electrons that have each found their own "p" orbital. They are not sharing. They are not paired up. They are, you guessed it, unpaired.
So, the answer to our burning question is: a Nitrogen atom has three unpaired electrons.
It’s not a super common thing to ponder, I'll grant you that. You're probably not going to be quizzed on this at your next barbecue. But isn't it kind of neat? This simple atom, so fundamental to, well, everything around us (including you and me!), has this little inner dance of paired and unpaired electrons. It’s like a tiny, atomic secret.

Maybe it's an unpopular opinion, but I think it’s way more interesting than celebrity gossip. While everyone else is talking about who’s dating whom, we can be quietly smug, knowing the electron situation in a Nitrogen atom. We're basically science detectives, unraveling the mysteries of the universe, one electron at a time.
And the beauty of it is, these unpaired electrons are actually super important. They're like the social butterflies of the atomic world. They make Nitrogen very reactive. They're eager to find a partner, to form bonds, to make connections. This is why Nitrogen is so essential in forming all sorts of molecules, from the DNA in your cells to the proteins that build you up. It's all thanks to those three little rebels, dancing solo in their orbitals.
So, next time you breathe in, or eat something, or just generally exist, give a little nod to Nitrogen. And remember its little secret: three unpaired electrons, ready to mingle and make the world go 'round. It’s a small detail, but sometimes, it’s the small details that are the most fascinating, don't you think? I certainly do. It’s the little things, after all!
