How Many Structural Isomers Are Possible For C4h10

Hey there, fellow curious minds! Ever wondered why the world around us isn't just one giant, uniform blob? Think about it – we've got fluffy clouds, sturdy mountains, tiny little seeds that grow into towering trees, and us, a whole spectrum of amazing humans. It's all thanks to the incredible way things can be put together, and that's exactly what we're diving into today. We're going to talk about something super cool from the world of chemistry, but don't worry, we'll keep it as breezy as a summer picnic.
We're going to explore a question that might sound a little bit like a riddle: How many structural isomers are possible for C4H10? Now, before you picture a bunch of intimidating chemical formulas and equations, let's break it down. Think of it like LEGOs. You have a certain number of bricks, right? If you have, say, 4 red bricks and 10 blue bricks, you could arrange them in all sorts of ways to build different things. You could make a little car, a tall tower, or maybe even a wobbly robot. Each of those is a different structure, even though you're using the exact same set of bricks.
In chemistry, atoms are our "bricks." C4H10 is like our specific set of bricks: 4 carbon atoms (the "C"s) and 10 hydrogen atoms (the "H"s). The amazing thing is, these atoms can link up in different ways, creating different molecules. These different arrangements are called structural isomers. They have the same ingredients, but they're assembled differently, leading to slightly (or sometimes, very) different properties. It’s like having a recipe for cookies that calls for flour, sugar, and eggs. You can make chocolate chip cookies, oatmeal raisin cookies, or even peanut butter cookies – all using the same basic ingredients, but the structure of how you mix and bake them changes the final delicious outcome.
The Humble Building Blocks: Carbon and Hydrogen
Our main characters in the C4H10 story are carbon and hydrogen. Carbon is like the ultimate connector. It loves to bond with other atoms, especially itself. Think of carbon atoms as little social butterflies, always ready to link arms with their friends. Hydrogen, on the other hand, is a bit more of a one-on-one kind of atom. It usually just needs one partner to be happy.
So, when we have 4 carbons and 10 hydrogens, the first and most straightforward way they can arrange themselves is in a nice, neat, straight line. Imagine lining up four friends holding hands, and then a bunch of other friends (the hydrogens) come and hold the free hands of the first four. This is our first possible structure for C4H10.
We call this one butane. It's a simple, unbranched chain. Picture a perfectly straight noodle. That's kind of what this molecule looks like when you draw it out.
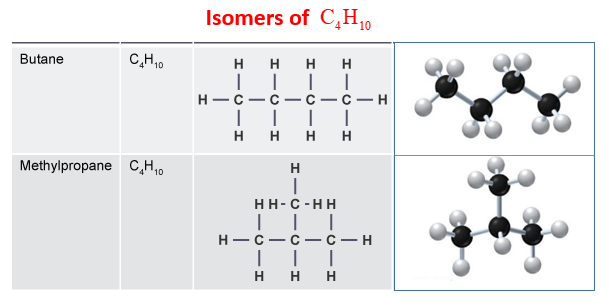
This straight chain arrangement is pretty stable and common. It’s like the default setting for these atoms. But here’s where the magic of isomerism kicks in!
Branching Out: The First Twist
Now, what if our carbon atoms decide not to form a perfect straight line? What if one of them decides to go off on its own little side adventure? Imagine our four friends holding hands in a line, but then one of them decides to hold the hand of a friend who is also holding the hand of someone else. This creates a branch!
So, instead of a 4-carbon straight chain, we can have a 3-carbon chain with one carbon atom attached to the middle carbon of that chain. Think of it like a T-junction on a road. You have the main road (the 3-carbon chain), and then a smaller road branching off from the middle.
This is our second structural isomer for C4H10. We call this one isobutane. It's got the same 4 carbons and 10 hydrogens, but the way they're connected is different. It's like having the same ingredients for a cake, but instead of a round cake, you make a mini-muffin shape. The ingredients are the same, but the form is distinct.

Why should you care about this little difference? Well, even though they have the same atoms, butane and isobutane behave a little differently. For instance, they have slightly different boiling points, meaning they turn into gas at slightly different temperatures. This might not seem like a big deal, but in the world of chemistry and industry, these small differences can be super important!
Are There Any More Twists?
So, we've got our straight chain (butane) and our one branch (isobutane). This is where the question gets really interesting. Can we rearrange these 4 carbon and 10 hydrogen atoms in any other way? We've tried a straight line and a single branch. What about more complex branching?
Let's try to imagine another arrangement. We have 4 carbons. If we try to put two branches off the same carbon, we run into a problem. Carbon can only make a certain number of connections (usually four). If we have a central carbon with two branches, and those branches are also carbons, we quickly run out of room and the hydrogen count gets all messed up. It’s like trying to fit too many people onto a small seesaw – it just doesn't work!
In fact, no matter how creatively you try to rearrange those 4 carbon atoms and 10 hydrogen atoms, you'll find that you can only come up with these two distinct patterns of connection. You can't create a third unique structure using only these building blocks in different connection orders.
So, the answer to our question, How many structural isomers are possible for C4H10? is two.
Why Should We Even Bother Knowing This?
You might be thinking, "Okay, so there are two ways to connect these atoms. So what?" This is a totally fair question! It's like asking why it matters if your favorite shirt has a different stitch pattern or if your sandwich has the lettuce on top or on the bottom. It seems minor, right?
But this concept of structural isomers is everywhere. It's fundamental to understanding the world of chemistry, and chemistry is fundamental to pretty much everything!

Think about medicines. The difference between a helpful drug and something that doesn't work (or worse, is harmful) can be as subtle as a slight change in how atoms are arranged. It's like having a key that fits perfectly into a lock versus a key that looks almost the same but has a tiny difference – the wrong key just won't open the door, no matter how hard you try.
It also affects the materials we use every day. The plastics in your toys, the fuel in your car, the fibers in your clothes – all these are made of molecules, and the way those molecules are structured dictates their properties. Imagine trying to build a strong bridge out of marshmallows versus steel. The difference in structural arrangement leads to vastly different strengths and uses.
Even in nature, this is a huge deal. The difference between the sugar that gives you energy and a toxic substance might be a matter of a few atoms being hooked up in a slightly different order. It's the difference between a helpful friend and someone you’d rather avoid, all based on subtle structural cues.
So, while C4H10 might seem like a simple case with just two isomers, it’s a fantastic gateway to understanding a much bigger and more fascinating concept. It’s a reminder that even with the same basic ingredients, the way things are put together can create an incredible diversity of forms and functions. It’s the art of molecular architecture, and it shapes the world we live in, one atom at a time!
