How Many Stereoisomers Are There For The Compound Shown
Imagine you’re at a party, and you meet someone who looks exactly like your best friend, but they have a slightly different accent and a penchant for pineapple on pizza. They’re clearly not your friend, but the resemblance is uncanny. In the quirky world of chemistry, we have a similar phenomenon, but instead of people, it's molecules! And when it comes to a particular molecule, things get a bit like a family reunion where everyone looks similar but has their own unique personality.
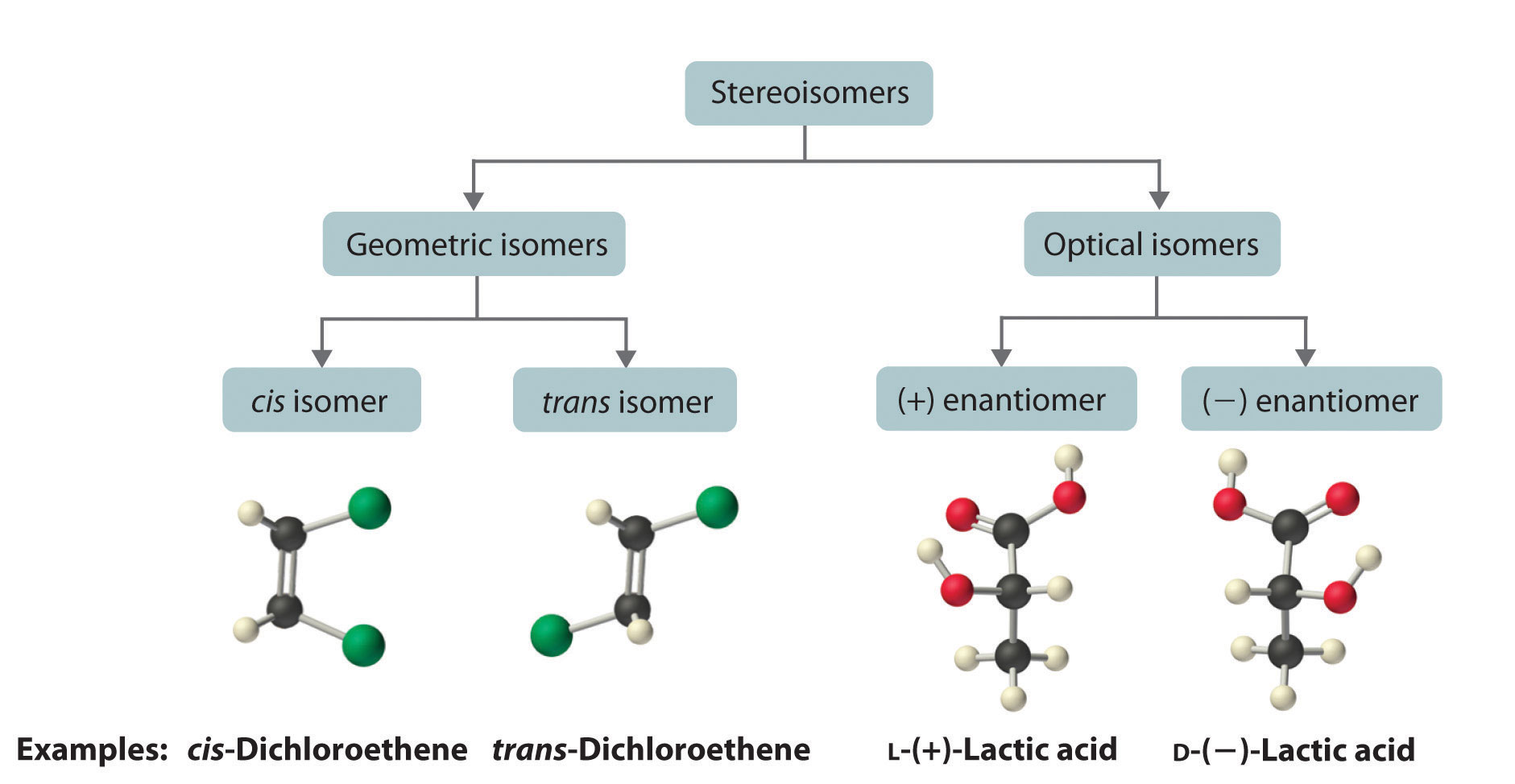
Let’s talk about our star of the show, a rather unassuming chemical compound. Think of it like a tiny LEGO creation, but with four different "studs" (atoms or groups of atoms) attached to a central brick. Now, the fun part is how these studs can be arranged. They can point up, down, left, or right, and depending on how you twist and turn our little LEGO creation in three-dimensional space, you can end up with different arrangements. These different arrangements are what we call stereoisomers. They’re like molecular twins, but not identical. They have the same "building blocks" but are put together in a slightly different way, like mirror images that can’t quite be perfectly superimposed, much like your left and right hands.
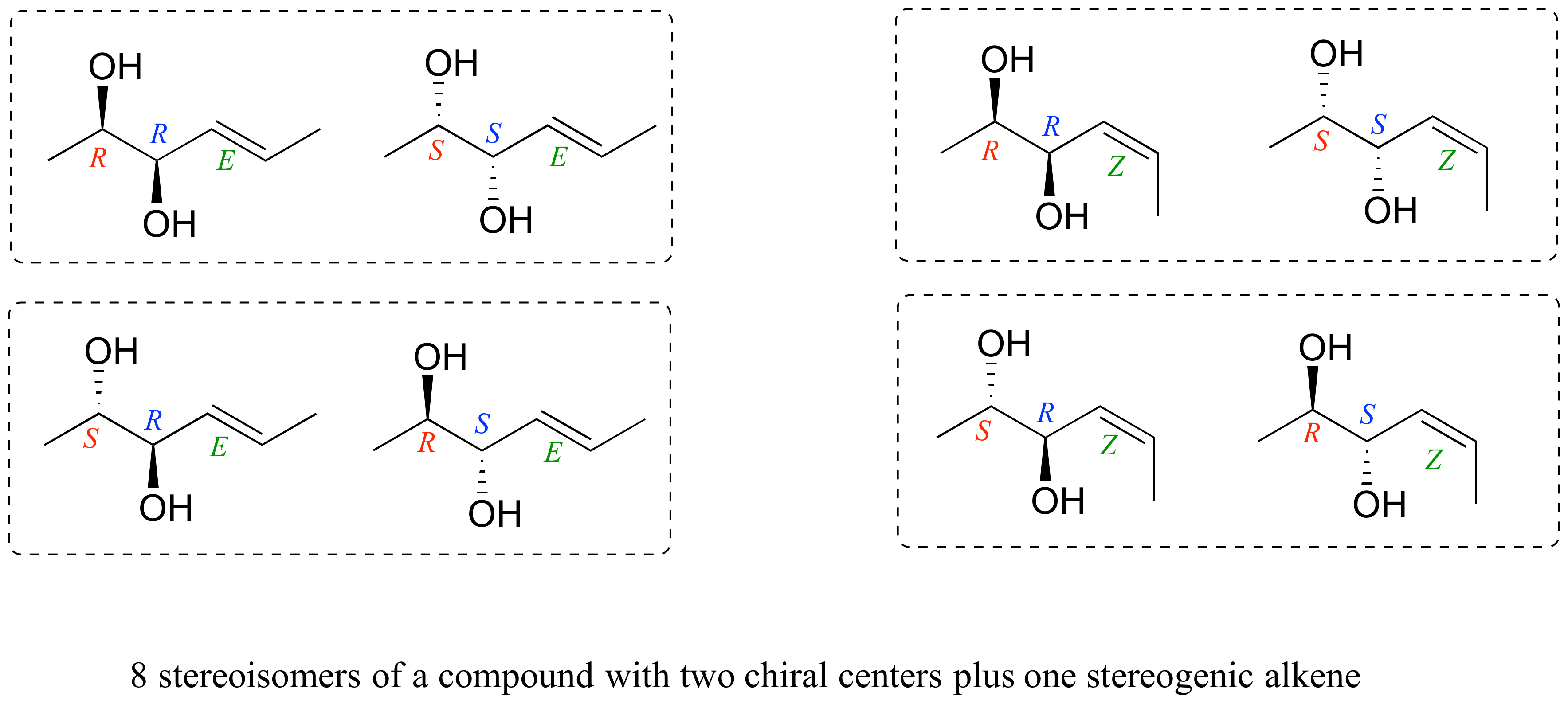
So, for our humble molecule with its four distinct studs, how many of these "molecular twins" or "cousins" can we create? The answer, surprisingly, is quite a few! It’s like having a box of the same LEGO set, but instead of just one way to build the spaceship, you discover you can build several different, yet structurally similar, versions. For our specific compound, the magic number is 16. Yes, sixteen different ways to arrange those four studs! That’s a whole party of molecules, all looking vaguely familiar but each with their own subtle (or sometimes not-so-subtle) differences.
Think about it this way: each of those four studs has two possible positions (think of them as "up" or "down" relative to a certain plane). For the first stud, you have 2 choices. For the second, another 2. The third, a third 2. And the fourth, a fourth 2. It’s a multiplicative effect! So, 2 x 2 x 2 x 2 = 16. It’s like a combinatorial explosion of molecular possibilities! This might sound a bit daunting, but it’s also incredibly fascinating. It means that even with a relatively simple building block, nature (or a chemist in a lab!) can create a surprising amount of variety.
Now, you might be wondering, why does this even matter? Well, these tiny differences in arrangement can have a HUGE impact. Imagine a key fitting into a lock. If the key is slightly bent or has a different shape, it might not open the door. In our bodies, molecules often work like keys fitting into very specific "locks" on our cells. A molecule that’s a perfect fit might be a life-saving drug, while its slightly different stereoisomer could be inactive, or even worse, have harmful side effects. It’s like the difference between a perfect handshake and an awkward elbow bump – both involve hands, but the outcome is entirely different!

This is why understanding stereoisomers is so crucial in fields like medicine and pharmaceuticals. Scientists have to be incredibly precise. They’re not just building molecules; they’re building specific versions of molecules, the ones that will do the job they’re intended for. It's like being a master chef, knowing that using the wrong spice, even if it's a related one, can completely change the flavor of your dish.
For our particular compound, with its 16 potential stereoisomers, it’s like having a whole buffet of molecular options. Some might be sweet, some might be savory, and some might be… well, let’s just say they don’t taste very good! The beauty of it all is that nature, through evolution, has figured out how to use these different molecular shapes to its advantage. Think about the incredible diversity of life around us, from the scent of a rose to the complex signaling within our brains. So much of that relies on the precise three-dimensional arrangements of molecules.
So, the next time you hear about a chemical compound, remember that it's not just a collection of atoms. It’s a miniature sculpture, a complex arrangement in space. And for our featured compound, it’s a whole party of 16! It’s a testament to the incredible intricacy and elegance of the molecular world, a world where even the slightest twist can lead to a whole new personality. It’s a reminder that in chemistry, as in life, sometimes the smallest differences can make the biggest impact. It’s a bit like a family gathering where you have the sensible cousin, the adventurous one, the quiet observer, and the life of the party – all made of the same "stuff," but each uniquely themselves.
