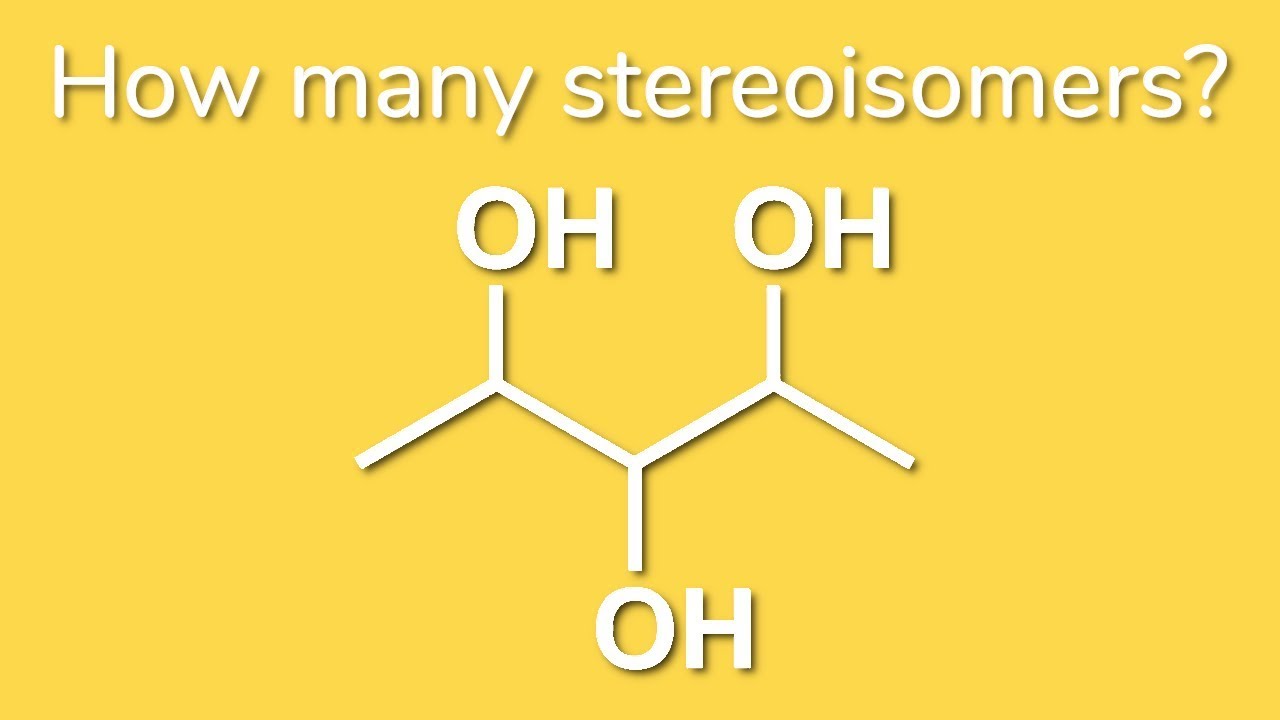
How Many Stereoisomers Are Possible For The Following Molecule
Hey there, science curious folks! Ever look at a molecule and wonder, "Is that it?" Like, could it be arranged a little differently and still be the same chemical building block, but with, you know, a totally different vibe?
Well, today we're diving into the fascinating world of stereoisomers. Don't let the fancy name scare you! Think of it like this: imagine you have a Lego creation. You can take the same bricks and put them together in slightly different ways, right? The bricks are the same, but the final structure can look and behave differently. That's kind of what stereoisomers are all about in the molecular realm.
So, we've got this particular molecule we're gonna dissect today. It's not like, a super complicated behemoth, but it has enough going on to be a really neat example of how these different spatial arrangements can pop up. And the question is, how many different versions of this molecule can we actually make, just by tweaking how its atoms are arranged in 3D space? It's like a molecular puzzle!
Unpacking the Mystery: What Makes a Stereoisomer?
Okay, so what exactly makes two molecules stereoisomers? The key thing is that they have the exact same chemical formula – meaning they have the same number of each type of atom. They're also connected in the same order. The difference, and this is the juicy part, lies in the three-dimensional arrangement of their atoms. It’s all about their spatial orientation.
Think about your left hand and your right hand. They’re made of the same “parts” (bones, muscles, skin), but you can’t perfectly superimpose one on the other. They are mirror images, but they’re not identical. This is a classic analogy for a specific type of stereoisomer called an enantiomer. If a molecule has this kind of "handedness," it means it has enantiomers.
But it’s not just about mirror images! Sometimes, molecules can be arranged in different ways that aren't mirror images. These are called diastereomers. Imagine a car. It has wheels, an engine, doors – all the same parts as another car of the same model. But maybe one car has the steering wheel on the left, and the other has it on the right. They're still cars, but their "arrangement" of controls is different, and they aren't mirror images of each other. See? It’s all about the spatial game!
The Molecule in Question: Let's Get Specific!
Alright, let's peek at our star molecule for today. (Don't worry, we won't get bogged down in jargon). We're looking at a molecule that has a few key features that allow for these stereoisomeric variations. It has a central atom, a chiral center, and then a bunch of different things attached to it.
What’s a chiral center, you ask? Think of it as a junction point. If an atom is connected to four different groups, it's often a chiral center. This is a huge clue that handedness might be involved! It’s like a crossroads where the path taken can lead to a different destination, even if you started from the same spot.
Our molecule has a central carbon atom. And this carbon atom is attached to four distinct things. This is the magic ingredient for creating stereoisomers. If that carbon was attached to, say, two identical groups, then no matter how you spun it around, it would look the same. But with four different groups? That's where the fun begins!

Counting the Possibilities: The Math Behind the Magic
So, how do we figure out the maximum number of stereoisomers possible? For molecules with a single chiral center, it's pretty straightforward. If you have one chiral center, you can generally have up to two stereoisomers. These will be a pair of enantiomers – our left and right-hand versions.
Imagine our chiral carbon is like a spinner on a board game. You have four different colors on the spinner, and they can point in different directions. But for this specific type of stereoisomerism (enantiomers), it’s like the spinner can only be in two distinct "modes" that are mirror images of each other. You can't get more than that from a single chiral center.
Now, what if our molecule had more than one chiral center? This is where things get really interesting and the number of possibilities can explode! For every additional chiral center, the number of potential stereoisomers can double. It's like adding more spinners to the board – the number of combinations goes up significantly!
Let's say our molecule had two chiral centers. Theoretically, you could have up to 22 = 4 stereoisomers. And if it had three chiral centers? That’s 23 = 8 possible stereoisomers! It's like a branching decision tree, where each chiral center is a new fork in the road.

The Nitty-Gritty for Our Molecule
For the specific molecule we're looking at, it has one chiral center. This means that, in principle, we can have up to two stereoisomers. These two stereoisomers will be enantiomers – they will be non-superimposable mirror images of each other.
It's like having a key. You can have the standard key (let's call it R), or you can have its mirror image (let's call it S). Both are keys, they're connected to the same lock, but you can't just swap them out and expect them to work in exactly the same way. In biological systems, this difference can be HUGE. Think about how your body recognizes specific molecules. One enantiomer might fit perfectly into a receptor like a glove, while its mirror image might be completely useless, or even harmful!
So, for our molecule, the answer is two. It's a simple number, but it represents a fundamental concept in chemistry that explains a lot about how the world works, from the taste of our food to how medicines function.
Why Is This Even Cool?
You might be thinking, "Okay, two isomers. So what?" Well, this is where it gets really fascinating! As I mentioned, biological systems are incredibly sensitive to molecular shape. Enzymes, which are biological catalysts, are like highly specific puzzle pieces. They can only interact with molecules that have the exact right shape.
Imagine trying to unlock a door with the wrong key. It just won't turn. Similarly, if a drug molecule has the wrong stereochemistry, it might not bind to its target in the body, rendering it ineffective. In some cases, the "wrong" isomer can even have completely different, and sometimes dangerous, effects. This is why understanding stereoisomers is so crucial in drug development and understanding biological processes.
It's like the difference between a left-handed baseball glove and a right-handed one. They're both gloves, they're both for baseball, but you can't really use one if you're designed for the other! The difference in spatial arrangement matters profoundly.
So, the next time you hear about a molecule, remember that it’s not just a collection of atoms. It’s a 3D object, and its spatial arrangement can make all the difference in the world. And for our humble molecule, there are exactly two distinct spatial arrangements, each with its own potential role to play!
