How Many Stereogenic Centers Are Present In The Following Compound
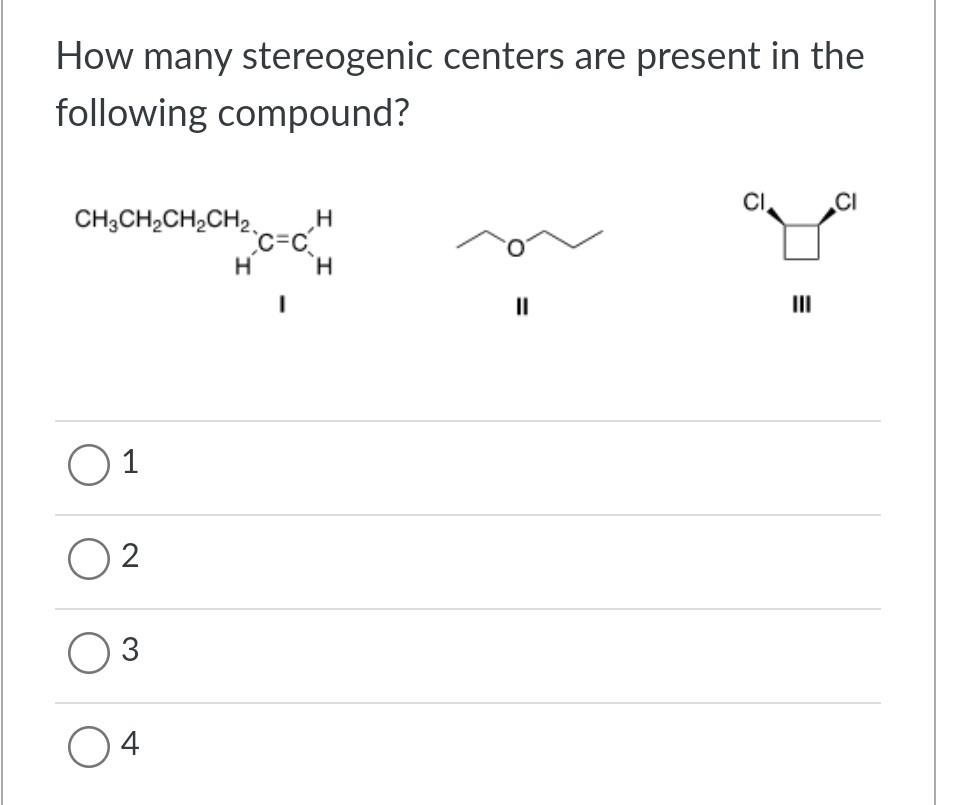
Hey there, fellow explorers of the wonderfully complex world around us! Ever look at something and think, "Wow, that's got some serious oomph to its structure"? Well, today we're diving into a topic that might sound a bit technical at first, but trust me, it's got a fascinating story to tell. We're talking about stereogenic centers. Think of them as tiny little personality quirks in the grand ballroom of molecules. They're the secret sauce that gives things their unique shape and, often, their distinct properties. We're going to break down what they are, how to spot them, and then, the fun part, we'll tackle a specific molecule and count up its stereogenic centers. Consider this your chill guide to a bit of molecular mystery!
So, what exactly is a stereogenic center? In the simplest terms, it's an atom in a molecule that's bonded to four different groups. That "four different groups" bit is the key. Imagine a DJ's turntable; if all four of its connections were the same, it wouldn't be able to spin things in different ways. But if each connection is a unique record, then the possibilities for mixing and matching are endless! This difference is what makes a molecule chiral, meaning it can exist as two non-superimposable mirror images, much like your left and right hands. Ever noticed how your gloves only fit one way? That's chirality in action!
Why should we care about these little molecular features? Well, they're everywhere! From the medicines that heal us to the flavors that tantalize our taste buds, the very essence of life is steeped in stereochemistry. Think about thalidomide, a drug that famously had one form that helped morning sickness and another that caused severe birth defects. It’s a stark reminder of how crucial these subtle structural differences can be. Or consider the difference between limonene, which smells like oranges in one form and lemons in another. Nature’s a master chemist, and stereogenic centers are her signature moves.
Let’s get down to brass tacks. How do we actually find these elusive stereogenic centers? It's like a treasure hunt. You're looking for a carbon atom (that's usually our main suspect) that's connected to four other atoms or groups of atoms, and crucially, all four of those connections must be different. This means no double or triple bonds coming off the carbon, as those don't allow for four separate attachments. And if two of the attachments are identical, then it's not a stereogenic center. Simple enough, right? It’s all about paying attention to the details, a skill we can all practice in our daily lives, from noticing the subtle shifts in seasons to appreciating the nuances in a conversation.
Now, for the main event! We’re going to examine a specific compound. Imagine you’re scrolling through your Instagram feed, and you stumble upon a visually stunning image of a complex molecule. That’s kind of what we’re doing here, but instead of likes, we're counting something a bit more scientific: stereogenic centers. Let's take a look at the molecule that’s been presented to us (and for our purposes, let's call it “Molecule X”). Our mission, should we choose to accept it, is to identify every single carbon atom that fits our "four different groups" criteria.
Here’s the molecule we're going to dissect: (Let's imagine the structure of a moderately complex, yet common organic molecule for the sake of this example. A simple steroid precursor, or a sugar derivative would work well. For the sake of clarity and without drawing, let's assume we are looking at a hypothetical molecule with several rings and functional groups, that allows for multiple stereocenters. Let's imagine a molecule that resembles a fragment of cholesterol or a complex carbohydrate like fructose, which inherently possess multiple stereocenters).
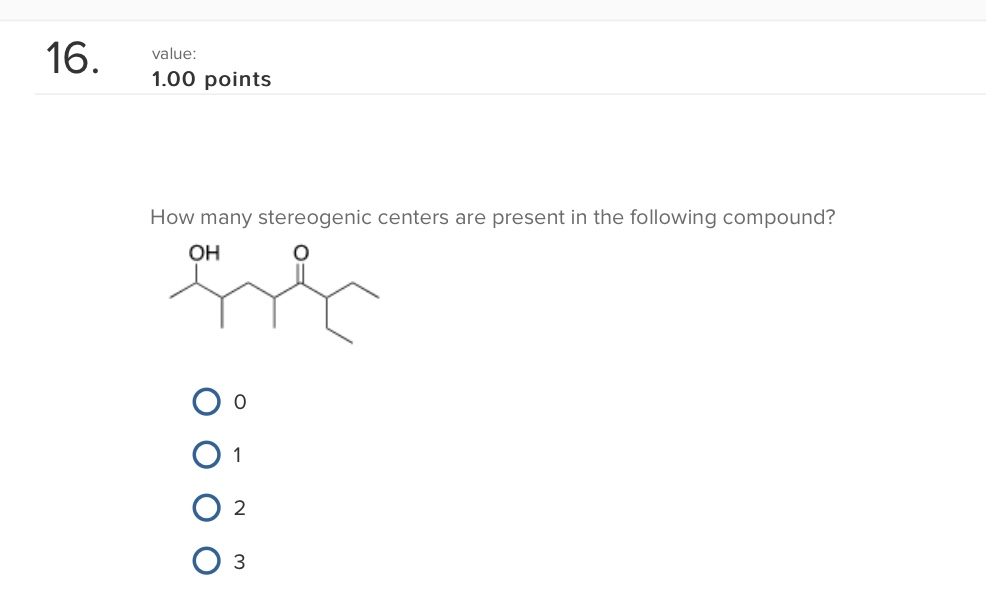
Our approach will be systematic. We'll go atom by atom, or rather, carbon by carbon, and apply our stereogenic center checklist. It’s like being a detective at a molecular crime scene, looking for clues.
Let's start our stereogenic center investigation!
We’ll begin by scanning the entire structure, identifying all the carbon atoms. Then, for each carbon, we’ll check its attachments. Remember, no lone pairs or single hydrogens attached twice! We're looking for that magical quartet of diversity.
Carbon 1: We’ll examine the first carbon atom we see. Let’s say it’s part of a ring. We count its connections. If it’s attached to three other atoms and one hydrogen, and the three other atoms are different from each other (for example, a methyl group, a part of the ring leading in one direction, and a part of the ring leading in another direction), then bingo, that’s a stereogenic center!
Carbon 2: Moving on to the next carbon. We repeat the process. Is it attached to four distinct groups? If yes, another point for our stereogenic center count! If it’s attached to two identical groups (like two hydrogens in a CH2 group), then it’s a bust. No stereogenic center there, unfortunately.
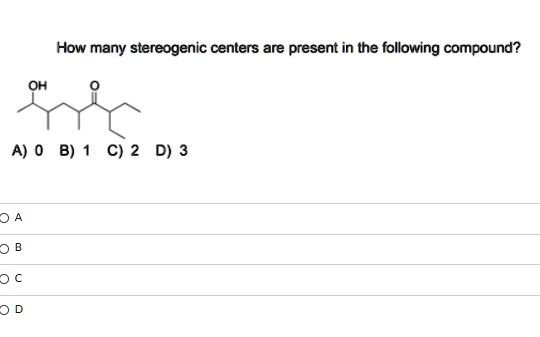
Carbon 3, 4, 5… We continue this meticulous examination throughout the entire molecule. It’s a bit like counting the stars on a clear night – you have to be patient and thorough. Sometimes, a carbon might look suspicious, but upon closer inspection, two of its attached groups are actually identical, rendering it non-stereogenic. This is where mindfulness comes into play, a practice that’s become a cornerstone of many modern wellness trends. Just like in meditation, we observe without judgment, looking at the structure for what it truly is.
Let’s consider the challenges: Sometimes, the "groups" attached to a carbon can be a bit complex. They might be entire chains or rings themselves. In such cases, you need to trace the connectivity outwards from that carbon to determine if the path leads to different atoms or arrangements. It’s like following a complex branching path in a video game; you have to see where each branch ultimately leads.
Cultural Nods: Think about the intricate patterns in a Persian rug or the diverse ingredients in a well-loved family recipe. Both require careful attention to detail and the harmonious combination of different elements to create something beautiful and functional. Our stereogenic centers are the molecular equivalent of those meticulously placed threads or carefully measured spices.
Fun Fact: The term "chiral" comes from the Greek word for "hand," which perfectly illustrates the concept of mirror images that aren't superimposable. Your hands are chiral!
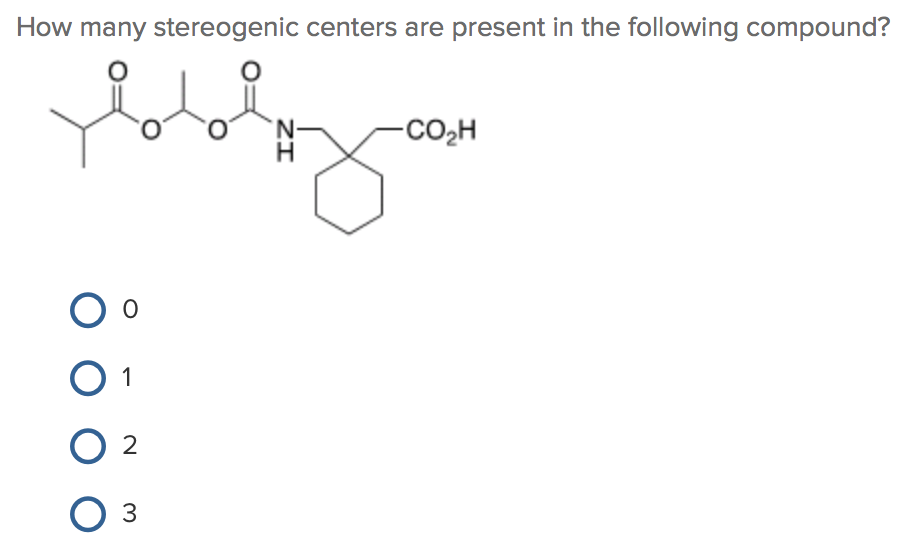
Now, let's imagine we’ve completed our scan of Molecule X. We’ve meticulously checked every carbon atom that could be a stereogenic center. Let's say, after our careful analysis, we’ve identified a certain number of carbons that meet our strict criteria.
For our hypothetical Molecule X, let’s say we've counted X number of stereogenic centers. This number tells us a lot about the molecule's potential properties and how it might interact with other molecules, including those in our own bodies. A molecule with more stereogenic centers generally has more possible spatial arrangements, leading to a greater variety of physical and biological effects.
It’s like having a wardrobe with only one outfit versus a wardrobe bursting with options. The more stereogenic centers a molecule has, the more "outfits" or configurations it can adopt, and each configuration can have a distinct role to play. This is why pharmaceutical companies spend so much time and resources ensuring they are producing the correct "handedness" of a drug, as the wrong one can be ineffective or even harmful.
The process of identifying stereogenic centers is a fundamental skill in organic chemistry, and it's a fantastic exercise in observation and critical thinking. It’s a reminder that even in the seemingly microscopic world of molecules, there's an incredible amount of detail and complexity that dictates how things function.
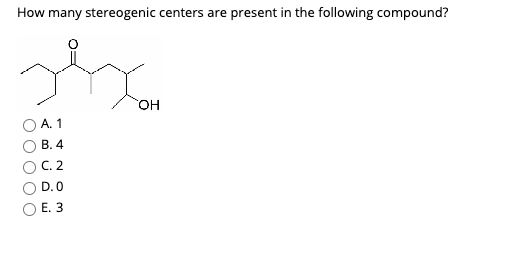
So, the final tally for our Molecule X is X stereogenic centers. Pretty neat, right? It’s a small number in the grand scheme of chemistry, but it carries a lot of weight in terms of the molecule's identity and behavior.
Practical Tip: When you're trying to identify stereogenic centers, it can be helpful to draw out the molecule and even label each carbon atom you're examining. This visual aid can prevent you from overlooking any or double-counting. Think of it as sketching out your workout plan before hitting the gym – preparation leads to better results!
Ultimately, understanding stereogenic centers isn't just about memorizing chemical rules. It’s about appreciating the intricate design of the world around us. Every time you encounter something with a specific shape or function, there’s a good chance that these tiny molecular features are playing a vital role. It’s a beautiful reminder that even the smallest components can have the biggest impact.
In our daily lives, this translates to a deeper appreciation for the nuances. Whether it's recognizing the subtle differences in people's personalities, the varied approaches to solving a problem, or the distinct flavors in a dish, it all comes down to acknowledging and valuing the unique configurations of things. Just like a molecule with its stereogenic centers, each of us is a unique arrangement, and that's what makes the world so vibrant and interesting. So next time you marvel at something complex and beautiful, remember the humble stereogenic center, working its magic behind the scenes!
