How Many Sigma Bonds Are Present In The Following Compound
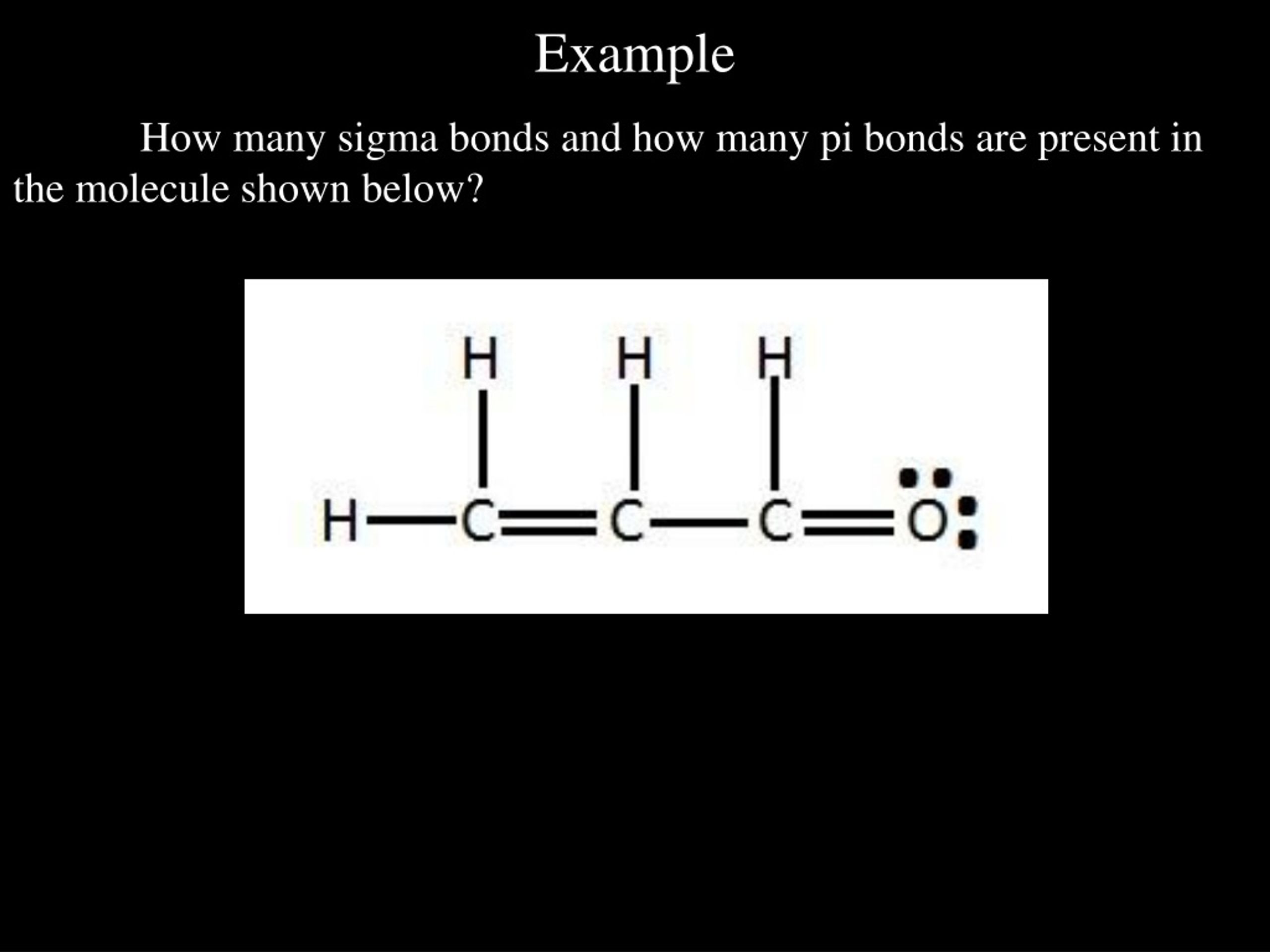
Alright, gather 'round, you magnificent nerds and curious cats! Today, we're diving headfirst into the wonderfully weird world of molecular structures, and I promise, it's going to be more exciting than watching paint dry, but with a lot more electrons. We're talking about sigma bonds, those unsung heroes of chemistry. They’re the glue holding everything together, the steady Eddies of the covalent world. Think of them as the handshake between atoms – firm, reliable, and utterly essential.
Now, someone, somewhere, probably while wrestling a particularly stubborn test tube, asked a question that sounded innocent enough: "How many sigma bonds are present in the following compound?" A seemingly simple query, right? Like asking how many sprinkles are on a donut. But in chemistry, even the simplest questions can lead you down a rabbit hole lined with atoms, electrons, and maybe even a stray beaker or two. And today, we're going to tackle that very question, but we're going to do it with pizzazz! Because, let's face it, chemistry can be a bit like dating – it's way more fun when there's a good story involved.
The Case of the Mystery Molecule
So, the question hangs in the air, a tantalizing puzzle. We need to identify the compound first, of course! Imagine it’s a molecule that looks a bit like a tiny, elaborate sculpture. Maybe it's something you'd find in a fancy science museum exhibit, or perhaps something a mad scientist conjured up in their underground lab. For our purposes, let’s conjure up a magnificent, fictional compound. Let’s call it… "Acetone-Hexatriene-Dimethanol Super-Sparkle". Yes, I know, it sounds like something you’d buy at a magical potion shop, but it gives us something to sink our teeth into!
Now, before you panic and start Googling "how to escape chemistry class," relax! We're not going to draw out this monstrosity. We just need to understand its basic building blocks. Think of it like deciphering a recipe. You don’t need to be a Michelin-star chef to count the number of eggs, right?
What Exactly Is a Sigma Bond?
Let’s get down to brass tacks. What makes a sigma bond so special? Imagine two atoms, feeling a bit lonely, and deciding to share some of their electron buddies. When they do this, their electron clouds overlap. A sigma bond, or σ bond, is formed when this overlap happens directly along the imaginary line connecting the two atomic nuclei. It's like they’re holding hands, perfectly aligned. It’s the strongest, most basic type of covalent bond.
Think of it this way: you have a bunch of atoms, and they’re all trying to get cozy. A sigma bond is the first hug they give each other. It’s the foundation, the bedrock, the very first step in forming a stable molecule. Every single bond between two atoms, whether it's a single, double, or triple bond, will have at least one sigma bond. It’s like the original gang, the OGs of molecular bonding.
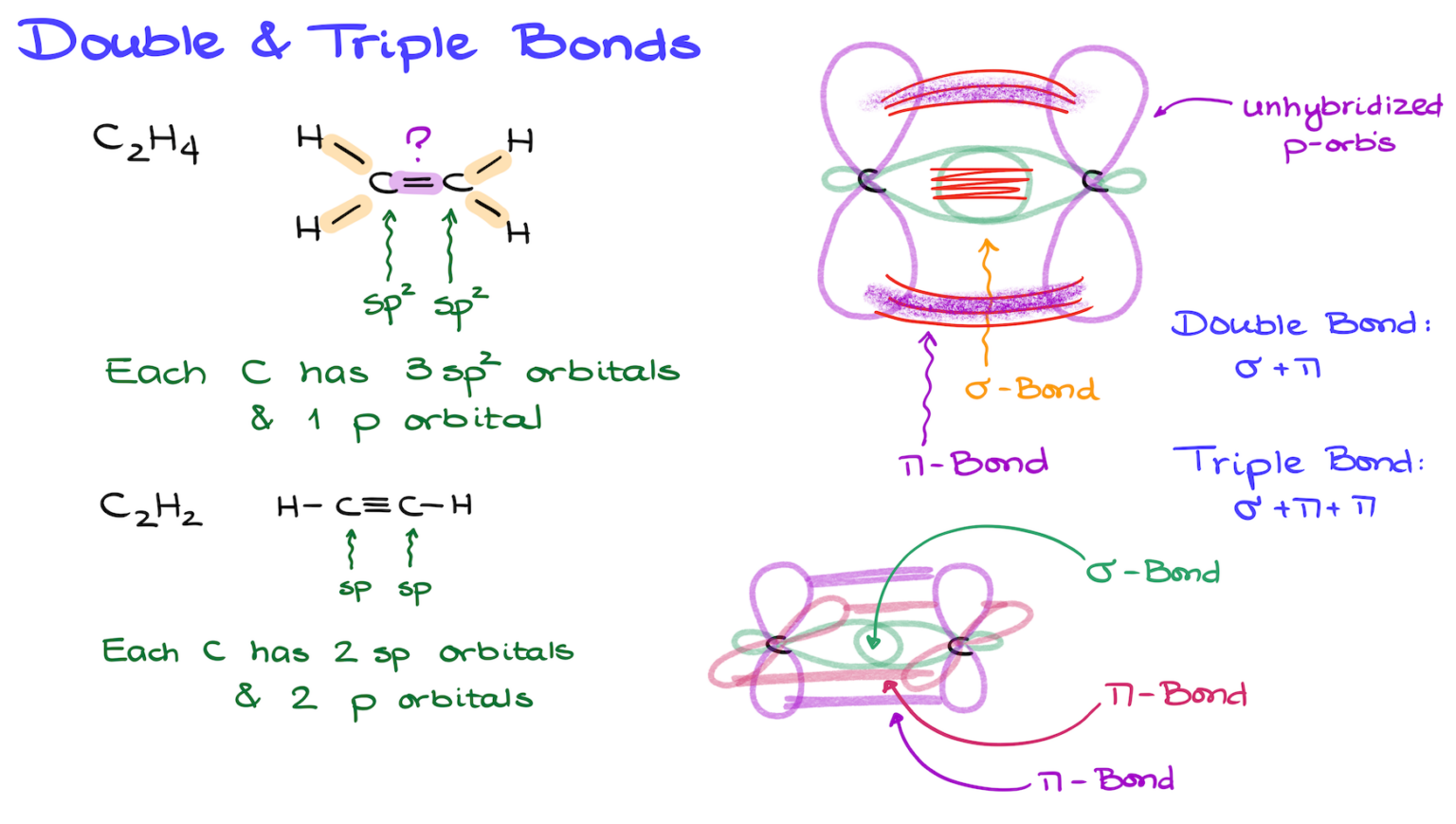
Fun fact for you: the word "sigma" comes from the Greek letter, which looks a bit like the number 6. And in many molecular structures, these bonds can have a radial symmetry, sort of like… well, a six-pointed star. Coincidence? Probably. But it’s a cool coincidence, right?
The Sneaky Secrets of Multiple Bonds
Now, here’s where things get a little spicy. Sometimes, atoms don't just want a simple handshake. They want a full-on embrace! This is where double bonds and triple bonds come in. A double bond is like a sigma bond plus another bond called a pi bond (π bond). A triple bond is a sigma bond plus two pi bonds. It’s like the atoms are really, really committed to sharing their electrons!
These pi bonds are formed by the sideways overlap of atomic orbitals, kind of like two shy people bumping shoulders from a distance rather than a direct head-on collision. They’re important, sure, but they’re not as strong or as foundational as the trusty sigma bond. So, when we’re counting sigma bonds, we’re looking for that primary, direct overlap. The pi bonds are extra credit, the sprinkles on top of the sigma bond ice cream cone.
Let's Count Some Bonds! (The Fun Part!)
Okay, back to our imaginary friend, "Acetone-Hexatriene-Dimethanol Super-Sparkle." Since we don't have a real picture, let's break down its components and imagine a simplified structure. We need to count the atoms and how they're connected. For this exercise, we'll focus on carbon (C), oxygen (O), and hydrogen (H) atoms, as they're the usual suspects in organic chemistry.
A key rule to remember is that each single bond between two atoms is one sigma bond. When you have a double bond, there's one sigma bond and one pi bond. For a triple bond, there's one sigma bond and two pi bonds. See? The sigma bond is always there, the generous giver!
Let's pretend our compound has a basic structure like this (and by "pretend," I mean I'm making it up for the sake of our delightful narrative):
- A central carbon backbone.
- Some hydrogens happily attached to carbons.
- An oxygen or two doing their oxygen thing.
- Maybe a double bond somewhere, just to keep things interesting!
Imagine a simple molecule like methane (CH4). It’s just one carbon atom with four hydrogen atoms attached. Each C-H bond is a single bond, so that's four sigma bonds. Easy peasy, lemon squeezy!
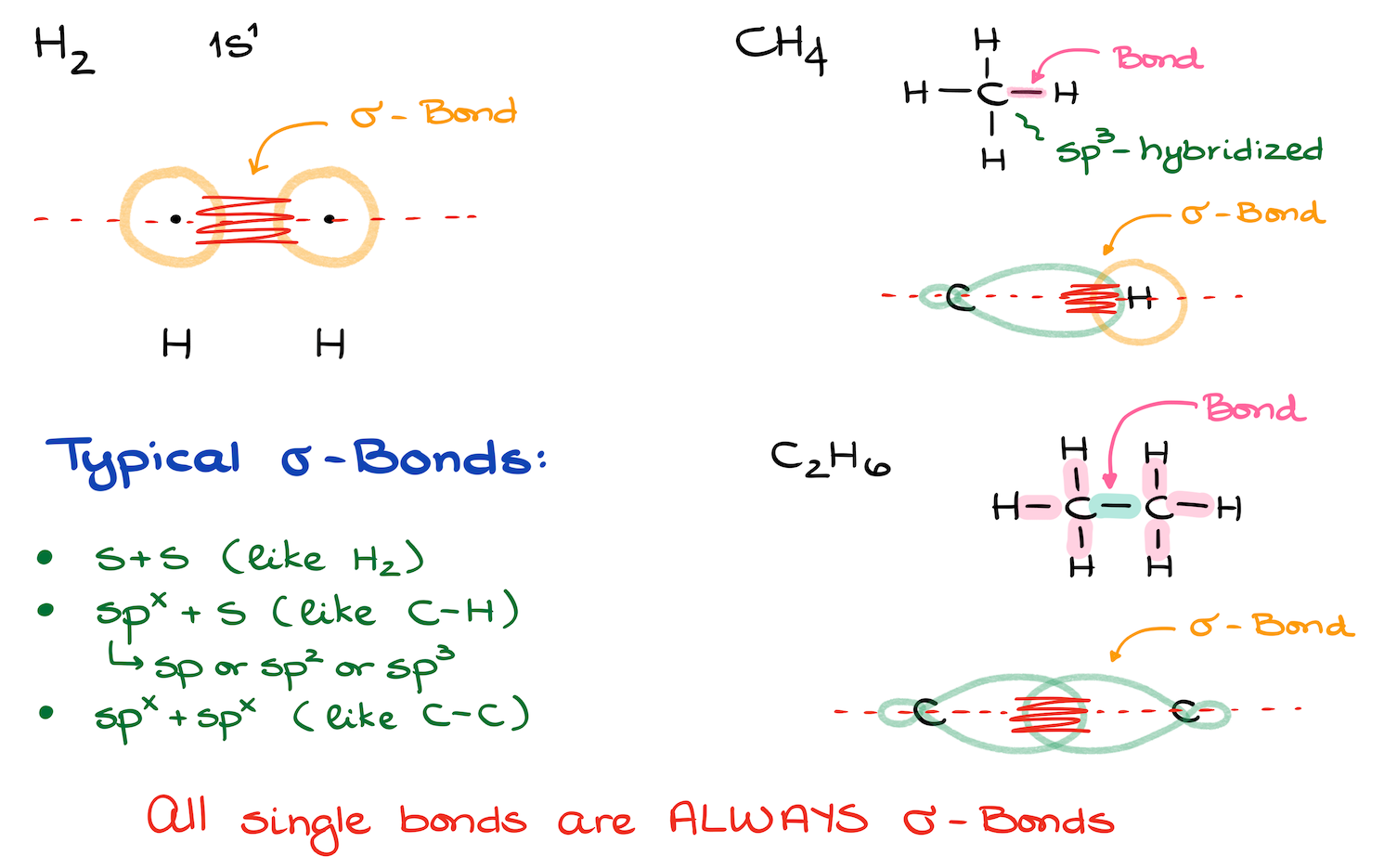
Now, let’s think about ethane (C2H6). Two carbons, and each has three hydrogens. So, we have C-C single bond, plus six C-H single bonds. That’s 1 + 6 = seven sigma bonds. See, the sigma bond count grows as the molecule gets bigger!
What about something with a double bond, like ethene (C2H4)? It has a C=C double bond and four C-H single bonds. The C=C double bond has one sigma bond and one pi bond. Then we have the four C-H single bonds, each being a sigma bond. So, 1 (from C=C) + 4 (from C-H) = five sigma bonds. The double bond didn't double the sigma count, just added one to it!
The Grand Finale: Counting Sigmas in Our Super-Sparkle!
So, for our made-up "Acetone-Hexatriene-Dimethanol Super-Sparkle," we’d approach it systematically. We’d break down the molecule into its constituent parts and count every single bond. Every line drawn between atoms represents a bond. We'd go through each atom, tracing the connections.
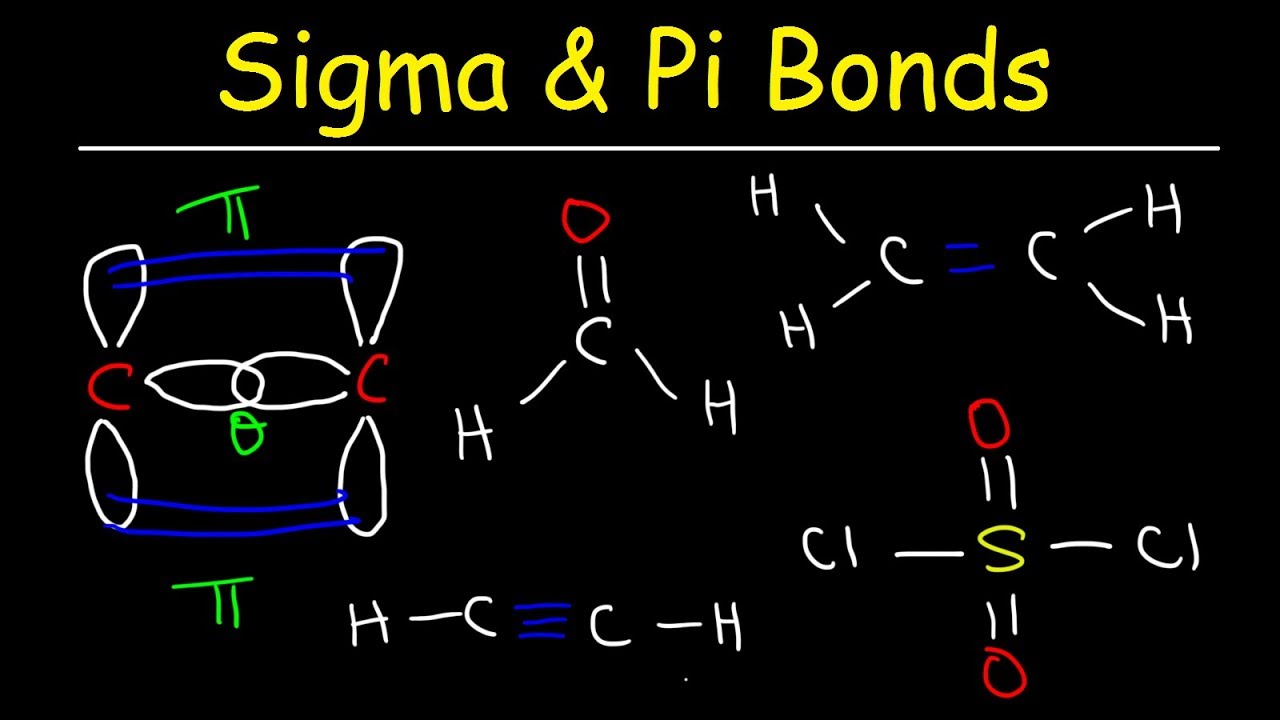
If we had a simple chain of carbons like in hexatriene (six carbons), and assume it’s all single bonds for a moment: C-C-C-C-C-C. That’s five C-C bonds. If each carbon is also attached to hydrogens to satisfy its valency (usually four bonds for carbon), we’d count those too. Every single line you see in a Lewis structure, unless it's part of a double or triple bond, is a sigma bond. If it is part of a double or triple bond, count the first bond as a sigma bond. The subsequent bonds are pi bonds, which we ignore for this specific count.
Let's imagine our Super-Sparkle has a structure that’s roughly like this: a few carbons in a ring, some hooked up in a chain, maybe an alcohol group (-OH), and a double bond thrown in for good measure. For every C-C, C-H, C-O, O-H single bond, that’s one sigma bond. For every C=C or C=O double bond, that’s one sigma bond. For any hypothetical C≡C or C≡N triple bond, that’s also one sigma bond. We just don't count the pi bonds.
So, the process is: 1. Identify all the atoms in the molecule. 2. Determine how they are bonded to each other (single, double, or triple bonds). 3. Count every single bond as one sigma bond. 4. For double bonds, count one sigma bond. 5. For triple bonds, count one sigma bond. 6. Sum them all up!
And there you have it! The grand total of sigma bonds for our hypothetical "Acetone-Hexatriene-Dimethanol Super-Sparkle" would be the sum of all these individual sigma bonds. It might be a big number, but it’s a number built on the solid foundation of those amazing, reliable sigma bonds. So next time you look at a molecule, remember: it’s all held together by these fantastic, fundamental connections. They might not be the flashiest, but they are, without a doubt, the backbone of the molecular universe. Cheers to sigma bonds!
