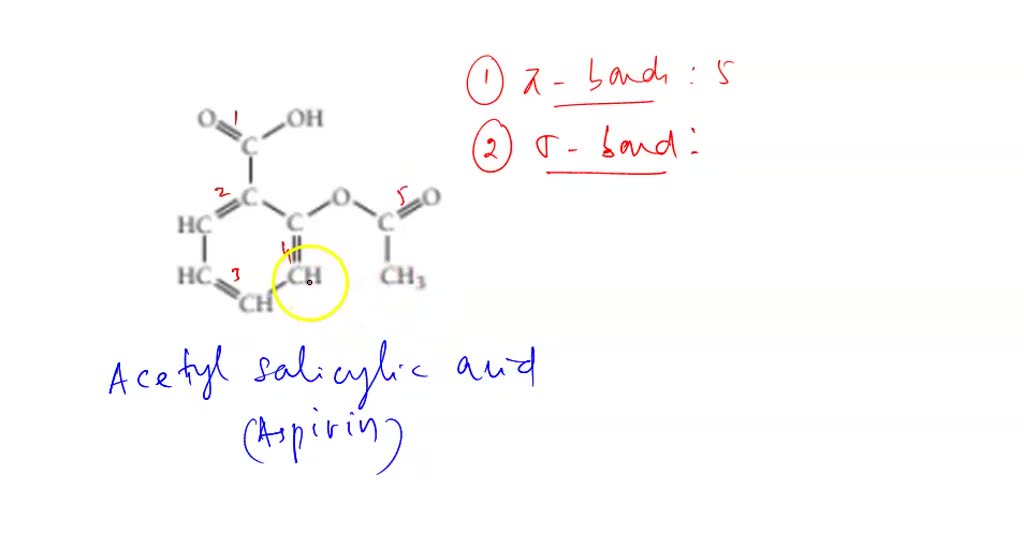
How Many Sigma Bonds Are Present In Acetylsalicylic Acid
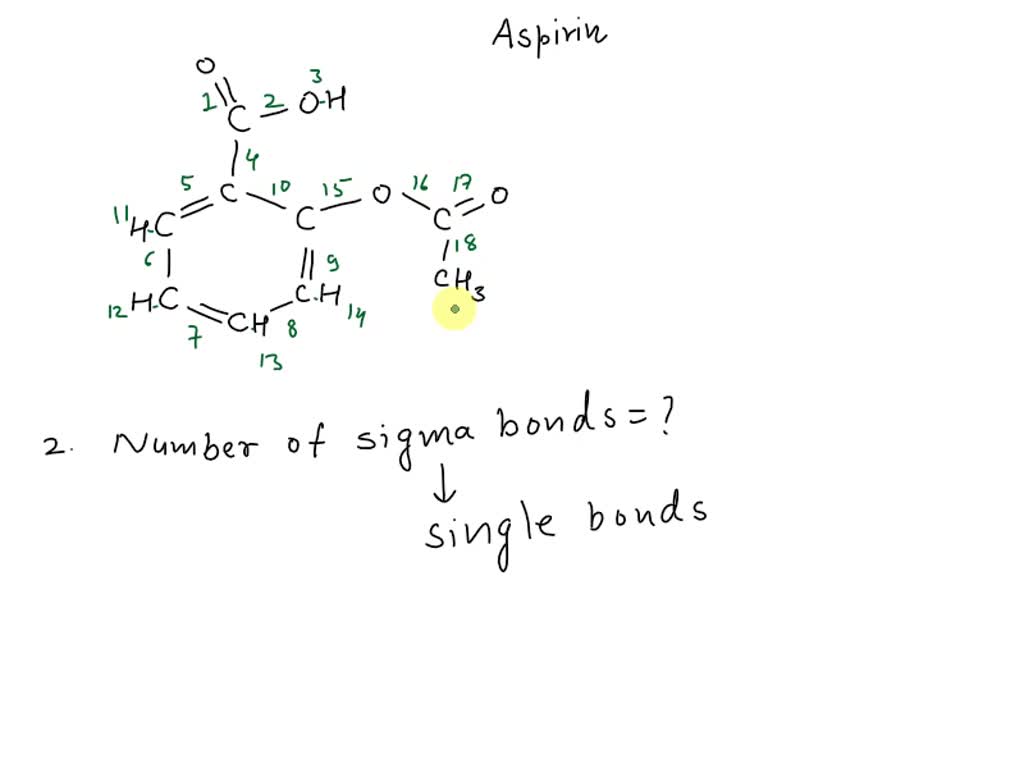
Hey there, fellow molecule marvels! Today, we're diving headfirst into the wonderfully wacky world of chemistry, specifically focusing on a little something called acetylsalicylic acid. Now, you might know it by its more common moniker: aspirin! Yep, that trusty little pill that’s probably hiding in your medicine cabinet right now. Pretty cool, right? We’re going to figure out just how many sigma bonds are lurking within this everyday superstar. Don't worry, we'll keep it super chill, like a lazy Sunday afternoon. No need for lab coats or super-serious expressions here!
So, what exactly is a sigma bond? Think of it as the original, bedrock bond between two atoms. It's the strong, stable connection that forms when two atomic orbitals overlap directly, face-to-face, along the imaginary line connecting the two nuclei. Imagine two magnets snapping together – that’s kind of the vibe. It’s the foundation upon which all other, fancier bonds are built. We’re talking about the bread and butter of chemical connections, the reliable workhorses of the molecular world. Without sigma bonds, molecules would just fall apart like a poorly constructed Lego castle. And who wants that?
Now, acetylsalicylic acid, or aspirin, is a bit of a fancy molecule. It's got a few different parts, like a miniature organic city. To count those sigma bonds, we need to get a little closer and inspect its structure. Don't worry, it's not as intimidating as it sounds. We'll break it down, piece by piece, like unwrapping a delicious (but molecular!) present.
Let's start with the core structure. Aspirin is essentially made up of a benzene ring, a carboxylic acid group, and an ester group. Sounds like a mouthful, right? But trust me, it's less scary than trying to assemble IKEA furniture without the instructions. The benzene ring, in particular, is a super common and important part of many organic molecules. It's a six-membered ring of carbon atoms, and it’s remarkably stable.
In a benzene ring, each carbon atom is bonded to two other carbon atoms and one hydrogen atom. Now, here's a little chemistry tidbit for you: carbon loves to make four bonds. It’s like the social butterfly of the periodic table, always looking to connect. In the benzene ring, each carbon atom forms single bonds with its neighboring carbons. These single bonds are our trusty sigma bonds. So, we’ve got six carbons in the ring, and each one is connected to two other carbons via a sigma bond. That’s 6 carbons x 2 sigma bonds/carbon = 12 bonds. But wait, we've counted each bond twice (once for each carbon involved). So, we divide by two, giving us 6 sigma bonds just within the carbon-carbon framework of the ring itself. Easy peasy, right? Almost too easy, I'm starting to get suspicious!
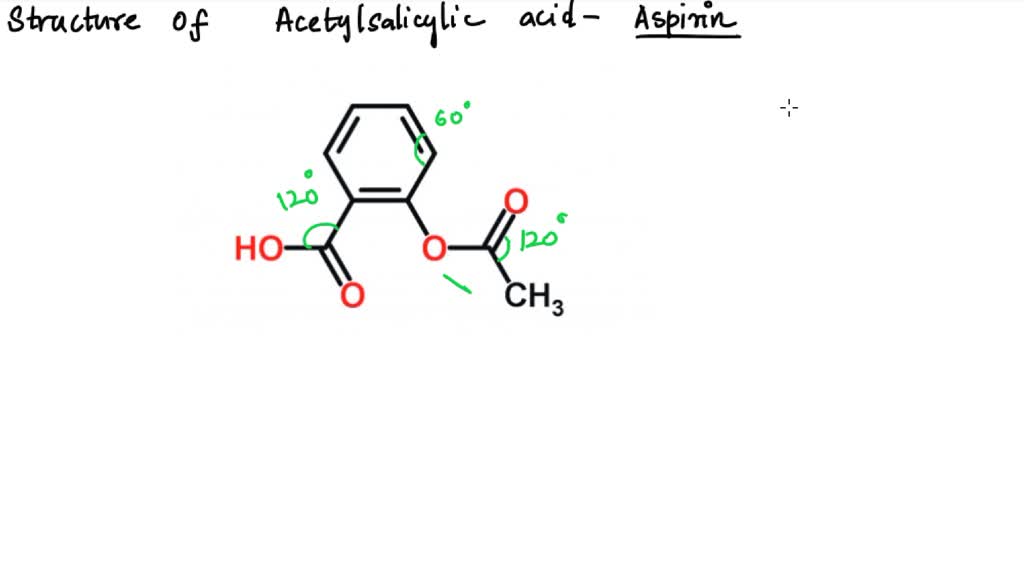
But that's not all for our benzene buddy! Remember, each carbon in the benzene ring also needs to be bonded to something else to complete its quad. In the case of benzene, it's usually a hydrogen atom. So, we have six carbon atoms, and each is bonded to one hydrogen atom. These are also sigma bonds! So, that's another 6 sigma bonds (6 C-H bonds). Add those to our C-C sigma bonds, and we're already at 12 sigma bonds just from the benzene ring and its attached hydrogens. We're on a roll, people! Who knew counting could be this exciting?
Okay, so the benzene ring is looking pretty well-connected with its 12 sigma bonds. But acetylsalicylic acid is a bit more complex than just a bare benzene ring. We have those extra bits attached, remember? Let’s tackle the carboxylic acid group. This group looks like this: -COOH. It's got a carbon atom, two oxygen atoms, and a hydrogen atom.
The carbon in the carboxylic acid group is bonded to the benzene ring. This is a sigma bond. Then, this carbon is also bonded to two oxygen atoms. One of these oxygen atoms is double-bonded to the carbon, and the other is single-bonded. Now, hold on a sec! A double bond is actually made up of one sigma bond and one pi bond. So, the carbon-oxygen double bond contributes one sigma bond. The other carbon-oxygen bond is a single bond, which is a sigma bond as well. Phew, got that? One sigma, one pi, and then another sigma.
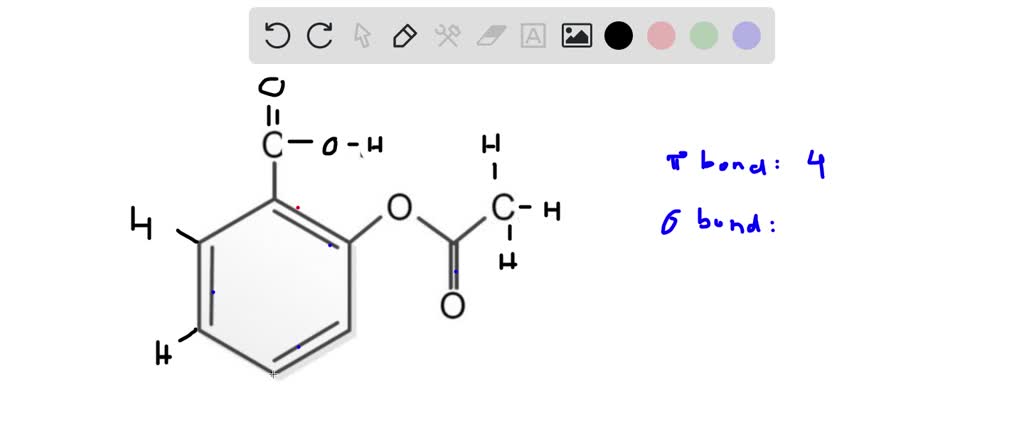
And what about that hydrogen atom attached to one of the oxygens in the carboxylic acid group? Yep, you guessed it – that's another sigma bond (an O-H bond). So, for the carboxylic acid group, we've got: one C-C sigma bond connecting it to the benzene ring, one C-O sigma bond, one C=O sigma bond (remember, only one of the double bond is sigma), and one O-H sigma bond. That's a total of 4 sigma bonds in the carboxylic acid part alone. Not too shabby!
Now, let's move on to the ester group. In acetylsalicylic acid, this group is attached to the benzene ring as well. It looks like this: -OCOCH3. This is where things get a tiny bit more involved, but we’re pros now, so we can handle it. The ester group is connected to the benzene ring via an oxygen atom. So, that's our first sigma bond: a C-O sigma bond linking the ester group to the benzene ring.
This oxygen atom is also bonded to a carbon atom. This is another sigma bond (O-C sigma bond). This carbon atom is part of a methyl group, which is -CH3. So, this carbon is bonded to the oxygen and to three hydrogen atoms. Each of these C-H bonds is a sigma bond. That's 3 C-H sigma bonds. And, of course, the carbon itself is bonded to the oxygen, forming that O-C sigma bond we just mentioned.
Let's recap the ester group, shall we? We have: 1. A C-O sigma bond connecting the ester group's oxygen to the benzene ring. 2. An O-C sigma bond connecting the ester’s oxygen to its carbon. 3. Three C-H sigma bonds within the methyl group (-CH3). So, in the ester group, we have a grand total of 1 + 1 + 3 = 5 sigma bonds. See? You're practically a chemistry whiz already!
Alright, time for the grand total! We've meticulously counted the sigma bonds in each part of our aspirin molecule. Let's add them all up:
- Benzene ring (C-C and C-H bonds): 12 sigma bonds
- Carboxylic acid group (-COOH): 4 sigma bonds
- Ester group (-OCOCH3): 5 sigma bonds
Total sigma bonds = 12 + 4 + 5 = 21 sigma bonds. Ta-da! We've officially discovered that there are 21 sigma bonds in a molecule of acetylsalicylic acid. Isn't that just amazing? We took this complex-looking molecule and broke it down into its fundamental connections. It's like uncovering the secret blueprint of a tiny, pill-shaped superhero!
So, the next time you reach for an aspirin, you can remember the intricate network of 21 sigma bonds working diligently to bring you relief. Each of those strong, stable connections is crucial for holding the molecule together, allowing it to do its job. It’s a beautiful reminder of the power and elegance of chemistry, even in the simplest of things.
And hey, if you found this little journey into sigma bonds fun and not too brain-bending, that’s fantastic! It just goes to show that even the seemingly complicated corners of science can be explored with a bit of curiosity and a playful spirit. So keep exploring, keep asking questions, and never underestimate the awesome power of understanding the tiny building blocks that make up our world. You’ve got this!
