How Many S Electrons Does P Atomic Number 15 Possess
Alright, settle in folks, grab your lattes, and let's dive into something that might sound as exciting as watching paint dry, but I promise, it's got more sparkle than a disco ball in a diamond mine. We're talking about atoms. Yeah, I know, groan all you want, but these little guys are the building blocks of everything. And today, we're going to chat about a particular atom that goes by the rather snazzy, if slightly uninspired, name of Phosphorus. Specifically, we're going to get our hands dirty – figuratively, of course, we're in a café – and figure out how many s electrons this chap, whose atomic number is a cool 15, has got in his arsenal.
Now, before your eyes glaze over like a donut in a sugar storm, let's break this down. Atomic number 15. What does that even mean? It's like the atom's ID number, the universal postcode. It tells us it's got 15 protons buzzing around in its nucleus like a toddler who’s just discovered a jumbo pack of fizzy sweets. And, in a happy, neutral atom (which is what we're usually dealing with in these general chats, unless we're talking about atoms that have lost their chill and become ions), the number of electrons is also a whopping 15. These are the little fellas that zip around the nucleus, playing a cosmic game of hide-and-seek in specific orbital paths. Think of them as tiny, energetic satellites.
The Shell Game: Atoms Edition
So, we've got 15 electrons to account for. Where do they all go? This is where things get a bit like a cosmic game of Tetris, or perhaps more accurately, like trying to fit all your holiday souvenirs into a single suitcase. Atoms don't just shove all their electrons into one big pile. Nope. They're organized. They live in "shells," which are basically energy levels. The closer you are to the nucleus, the lower the energy. Think of it as prime real estate – the closer to the sun (the nucleus), the more sought-after the spot.
These shells are numbered: 1, 2, 3, and so on. Shell number 1 is the innermost, the most exclusive penthouse suite. Shell number 2 is the slightly larger, still quite nice, apartment. And as you go further out, the shells get bigger and can hold more electrons. It’s like a cosmic onion, with layers of electron goodness.
Introducing the Sub-Atomic Stars: s, p, d, and f
But wait, there's more! Within these shells, things get even more interesting. Each shell is divided into different types of "sub-shells" or orbitals. These are the specific shapes and energy states where electrons can hang out. For our purposes today, the two most important players in this orbital drama are the s orbitals and the p orbitals. Imagine them as different types of rooms in your apartment complex: some are cozy studios (s orbitals), and some are more spacious multi-bedroom suites (p orbitals).
Here's the crucial bit: each s orbital is like a tiny, perfectly spherical VIP lounge. It's simple, elegant, and can only hold a maximum of two electrons. No more, no less. It's a strictly-two-per-s-lounge policy. These s orbitals are found in every shell. Shell 1 has one s orbital, Shell 2 has one s orbital, Shell 3 has one s orbital – you get the picture.
Then you have the p orbitals. These are a bit more flamboyant, existing in sets of three, oriented along different axes (imagine them like three arrows pointing in x, y, and z directions). Each of these p orbitals can also hold two electrons, meaning the entire p sub-shell can hold a grand total of six electrons. But, and this is a big but, p orbitals don't show up until you get to Shell number 2. Shell 1 is a bit of a minimalist, only having its s orbital. It’s like the trendy bachelor pad of the atomic world.
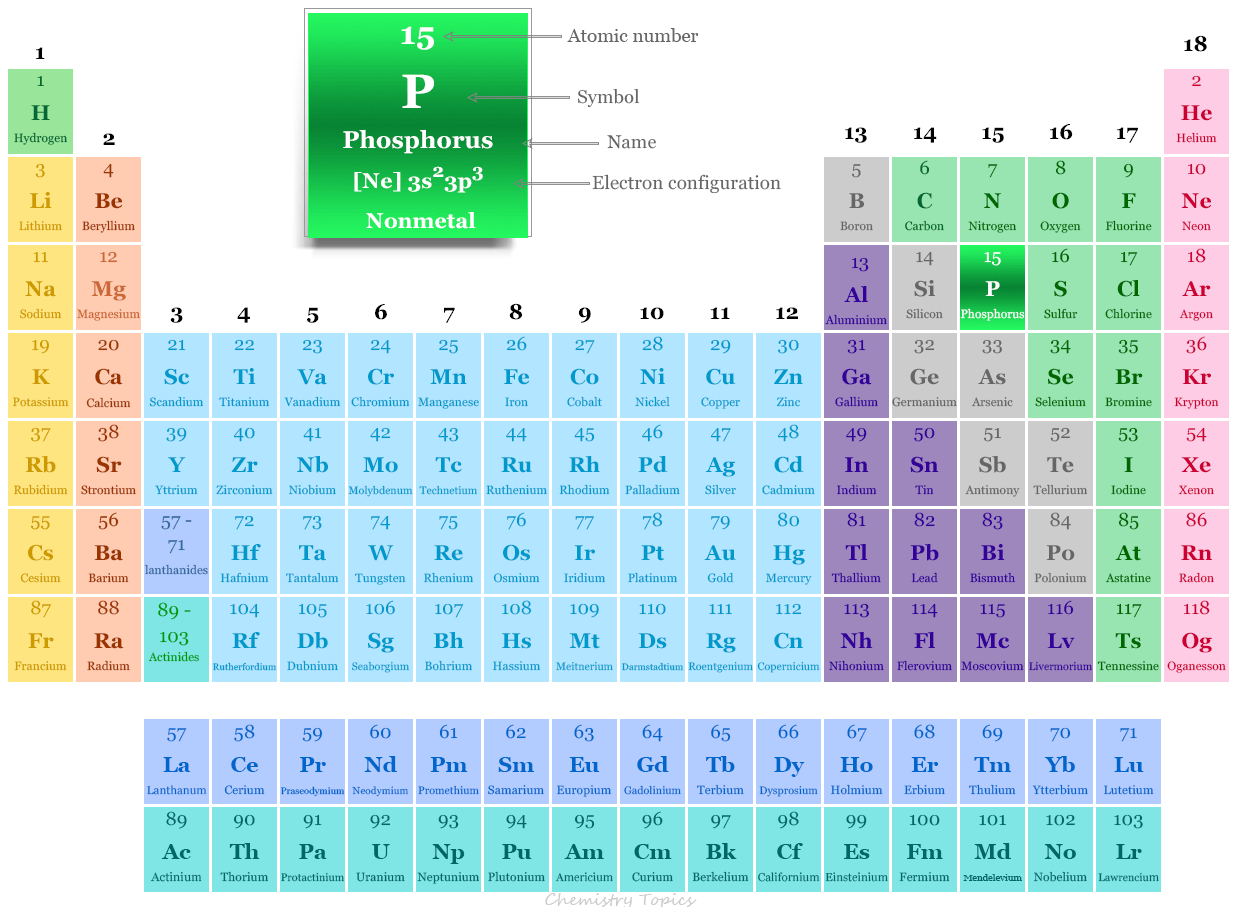
Let’s Count Some Electrons for Phosphorus (Atomic Number 15)!
Now, let's get down to business and populate our Phosphorus atom with its 15 electrons. We'll do this shell by shell, like a patient librarian shelving books.
Shell 1: This is the first stop. It's got one s orbital, and remember, that s orbital is a stickler for having exactly two electrons. So, 2 electrons go into the 1s orbital. We've got 13 electrons left to place. Easy peasy!
Shell 2: Next up is Shell 2. This shell is a bit more generous. It has its own s orbital, and that s orbital can hold 2 electrons. So, we put 2 electrons in the 2s orbital. Now we've used up 2 + 2 = 4 electrons. We still have 11 more to go! Also in Shell 2, we get our first taste of the p orbitals. Remember, a p sub-shell has three p orbitals, and each can hold 2 electrons, for a total of 6 electrons. So, the 2p sub-shell gets completely filled with its maximum of 6 electrons. We've now accounted for 2 (from 1s) + 2 (from 2s) + 6 (from 2p) = 10 electrons. We're getting closer! We have 5 electrons remaining.
Shell 3: This is where the remaining electrons will reside. Shell 3 starts with its own s orbital, which, as we know, can hold a maximum of 2 electrons. So, we pop 2 electrons into the 3s orbital. That brings our total accounted for to 10 + 2 = 12 electrons. We have 3 electrons left!
The Remaining Electrons: We've got 3 electrons left, and the next available sub-shell in Shell 3 are the p orbitals. Remember, p orbitals can hold up to 6 electrons. Since we only have 3 electrons left, they'll happily settle into the 3p sub-shell. They won't fill it completely, but they'll find their designated spots within the 3p orbitals. So, 3 electrons go into the 3p sub-shell.
The Grand Total of s Electrons!
So, let's tally up our s electrons for Phosphorus (atomic number 15)!
From Shell 1, we had the 1s orbital with 2 electrons.
From Shell 2, we had the 2s orbital with 2 electrons.
From Shell 3, we had the 3s orbital with 2 electrons.
Add them all up: 2 + 2 + 2 = 6 s electrons!
There you have it! Phosphorus, the atom with 15 electrons, has a grand total of 6 s electrons. They're diligently doing their job in their spherical little homes within the first three shells. So next time you’re munching on a healthy snack that contains phosphorus (like nuts or dairy – yum!), you can impress your friends by knowing that the atom responsible for all that goodness has 6 s electrons. Pretty neat, huh? Now, who wants another coffee? This atomic gossip is making me thirsty!
