How Many Protons Neutrons And Electrons Does Iridium Have
Imagine you’re at a fancy jewelry store, admiring the dazzling display of necklaces and rings. You might be drawn to the brilliant shine of gold or the deep, captivating hue of sapphires. But have you ever spotted a piece with a more subdued, yet undeniably elegant, silvery-white gleam? That, my friends, could very well be Iridium!
Now, Iridium isn't as famous as gold or silver. It’s a bit of a shy rockstar, more often found in the background, doing important work. But like any good rockstar, it has its own unique personality, and a big part of that personality is locked away inside its tiny, microscopic heart – its atoms.
Think of an atom like a miniature solar system. In the very center, we have the nucleus, which is like the sun. And orbiting around it, like planets, are the electrons. But the nucleus isn’t just empty space; it’s packed with two types of tiny particles: protons and neutrons.
So, how many of these little guys are hanging out inside an Iridium atom? Let’s dive in!
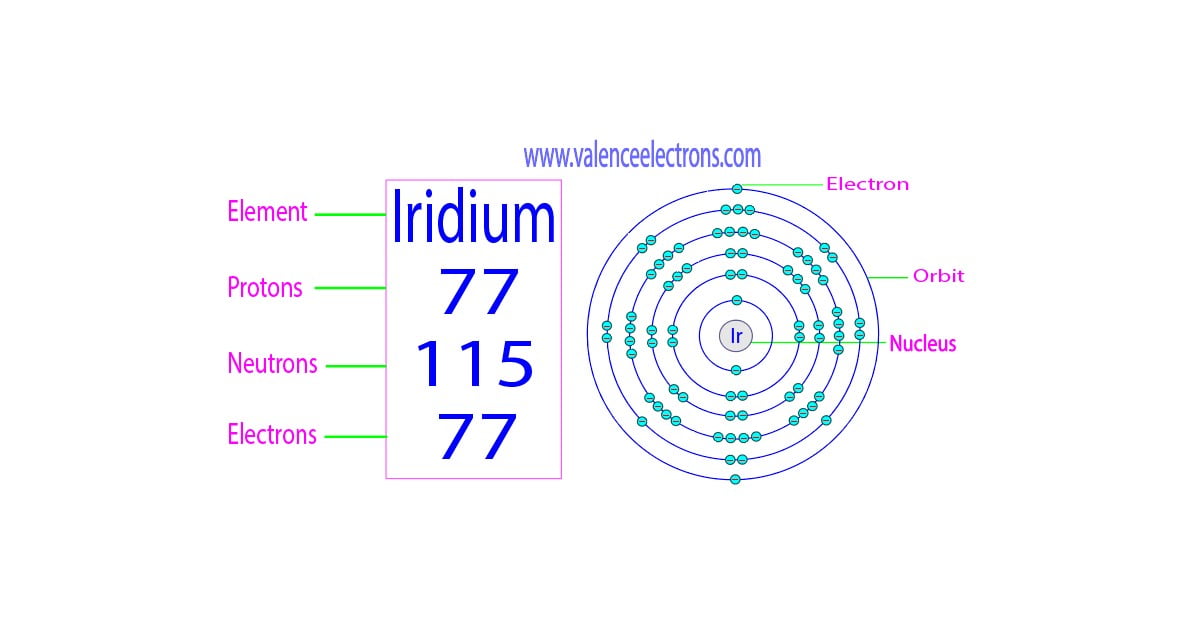
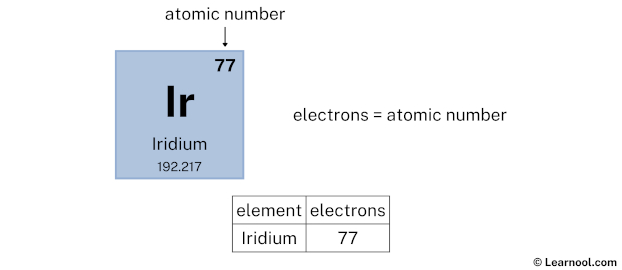
First up, let’s talk about the protons. These are the really important guys when it comes to identifying an element. It’s their number that tells us, “Yep, this is definitely Iridium and not, say, a sneaky piece of platinum trying to impersonate it!” For Iridium, the magic number of protons is 77. Yep, seventy-seven tiny, positively charged particles all huddled together in the nucleus. That’s a pretty specific number, isn't it? It's like having 77 friends all agreeing to wear the exact same uniform – it makes them instantly recognizable!
Now, these protons don't usually hang out alone. They like to bring along some buddies, and those buddies are the neutrons. Neutrons are like the quiet, steady friends in the group. They don't have any electrical charge, which is a bit of a superpower because it means they can help hold the nucleus together without causing a fuss. The most common type of Iridium atom has 114 neutrons. So, add those to our 77 protons, and you've got a nucleus with a whopping 191 particles packed inside! Imagine trying to fit 191 bouncy balls into a tiny little box – that’s a lot of jostling and bumping going on in there!
It's kind of like a secret handshake. Those 77 protons are the key that unlocks the identity of Iridium. Without them, it just wouldn't be itself.
How Many Protons, Neutrons and Electrons Does Iridium Have?
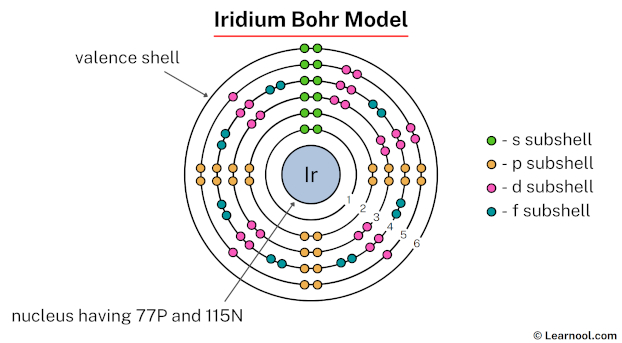
But the story doesn't end with the nucleus. We have to talk about the orbiting electrons. These are the super speedy, negatively charged particles that zip around the nucleus. In a happy, balanced atom, the number of electrons is always the same as the number of protons. Why? Because the positive charge of the protons and the negative charge of the electrons cancel each other out, making the atom electrically neutral. So, just like our proton count, Iridium has 77 electrons! That’s 77 tiny dancers twirling around the nucleus in a very specific pattern. They’re not just randomly flitting about; they have their own orbits, like different highways in the atomic city.
So, to recap our amazing Iridium atom: we have 77 protons, 114 neutrons (in the most common version, because sometimes atoms like to be a little quirky and have a different number of neutrons!), and 77 electrons. That’s a total of 191 tiny particles making up the core, with another 77 whizzing around on the outside!
It’s pretty mind-boggling when you think about it. This incredibly hard, dense metal, used in things like spark plugs, fountain pen tips, and even for the tiny parts in your smartphone, is made up of these incredibly small, energetic pieces. It's like a mighty oak tree being built from millions of tiny, invisible LEGO bricks!
And here’s a fun little tidbit: Iridium is incredibly rare. It’s much rarer than gold! This is partly because most of the Iridium on Earth came from outer space, carried here by meteorites. So, every time you see something made of Iridium, you’re looking at a little piece of history, a visitor from the cosmos that decided to stick around and be part of our world.
Next time you’re admiring something shiny and silvery, take a moment to appreciate the incredible cosmic dance happening within its atoms. Those 77 protons, 114 neutrons, and 77 electrons are all working together, perfectly balanced, to create the unique properties of Iridium. It’s a little reminder that even the most humble-seeming materials have a universe of complexity and wonder hidden within them, just waiting to be discovered.

