How Many Primary Alcohols Are Possible For C4h10o
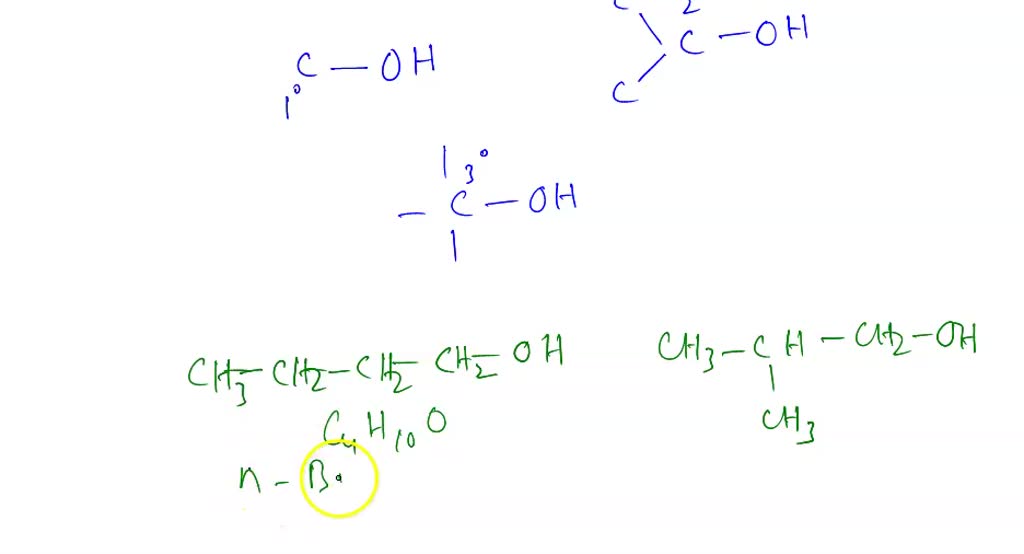
Ever looked at a simple molecular formula like C4H10O and wondered how many different ways you can arrange those atoms to make something real? It’s a bit like having a box of LEGO bricks and trying to build as many unique structures as possible. This isn't just a fun brain teaser for chemists; understanding these different arrangements, called isomers, is super important in fields from medicine to materials science. Knowing how many possibilities exist helps scientists discover new compounds, predict their properties, and even design entirely new molecules for specific jobs. It’s a fundamental way we explore the vast chemical universe!
Today, we're going to dive into a specific corner of this molecular playground: the world of primary alcohols with the formula C4H10O. Why primary alcohols? Because they're a great starting point for understanding isomerism. A primary alcohol is a type of organic molecule where a hydroxyl group (that's the -OH part) is attached to a carbon atom that is itself bonded to only one other carbon atom. Think of it as the hydroxyl group hanging off the end of a carbon chain. It’s a relatively simple structure, but even at this level, things get interesting!
Unlocking the Possibilities: C4H10O and Primary Alcohols
So, let's get down to business. We have our building blocks: 4 carbon atoms (C), 10 hydrogen atoms (H), and 1 oxygen atom (O). Our mission is to assemble these into primary alcohols. This means the -OH group must be on a carbon that's connected to just one other carbon. This is a key rule we need to follow.
First, let's consider the basic carbon skeleton. With 4 carbon atoms, the most straightforward way to arrange them is in a straight line. This gives us a chain of four carbons:
C - C - C - C
Now, where can we attach our -OH group to make it a primary alcohol? Remember, it has to be on a carbon bonded to only one other carbon. In our straight chain, the carbons at the very ends fit this description perfectly. They are each connected to just one other carbon atom.
So, we can attach the -OH group to the first carbon:
OH - C - C - C - C
This molecule is called butan-1-ol. Notice the "-1-" in the name? That tells us the hydroxyl group is on the first carbon of the four-carbon chain. This is our first primary alcohol!
What about the other end of the chain? If we attach the -OH group to the last carbon (which is the fourth carbon in our numbering), it's structurally identical to attaching it to the first carbon. It's still a primary alcohol, just flipped around. We still call it butan-1-ol.

So, with a straight four-carbon chain, we’ve found one unique primary alcohol.
Branching Out: The Other Carbon Skeleton
But wait, are there other ways to arrange 4 carbon atoms? Absolutely! We don't have to make a straight line. We can introduce branching. The next simplest arrangement for 4 carbons is a three-carbon chain with one carbon attached as a branch to the middle carbon. This looks like:
C | C - C - C
This carbon skeleton is called isobutane. Now, let’s try to attach our -OH group to make a primary alcohol. We need to find a carbon atom that is bonded to only one other carbon atom.

Let's examine the carbons in our branched skeleton:
- The two carbons at the ends of the three-carbon chain (the "arms" of the Y shape) are each bonded to only one other carbon atom. These are prime candidates for our primary alcohol!
- The central carbon atom is bonded to three other carbon atoms. Attaching an -OH here would make it a tertiary alcohol, not a primary one, so we can't do that.
So, we can attach the -OH group to either of the "arm" carbons. Since these two carbons are identical due to the symmetry of the molecule, attaching the -OH to either one will result in the same molecule. Let's attach it to one of them:
CH2OH | CH3 - CH - CH3
This molecule is called 2-methylpropan-1-ol. The "2-methyl" tells us there's a methyl (CH3) group on the second carbon, and the "-1-ol" tells us the hydroxyl group is on the first carbon of the main three-carbon chain. This is our second unique primary alcohol!
The Verdict
We've explored the two possible ways to arrange the 4 carbon atoms (a straight chain and a branched chain) and considered where we can attach the -OH group to satisfy the primary alcohol condition. In both cases, we found distinct and valid primary alcohols.
Therefore, for the molecular formula C4H10O, there are exactly two possible primary alcohols. These are:
- Butan-1-ol (from the straight chain of carbons)
- 2-Methylpropan-1-ol (from the branched chain of carbons)
Isn't it amazing how just changing the arrangement of the same atoms can lead to different compounds with potentially different properties? This is the beauty and power of isomerism, and primary alcohols with C4H10O give us a clear and simple illustration of this fascinating concept!
