How Many Peptide Bonds Are Present In Pentapeptide
So, I was at this super chill coffee shop the other day, you know, the kind with mismatched furniture and baristas who actually smile? I overheard a conversation between two students, hunched over laptops, looking utterly perplexed. One of them, let's call her Maya, sighed dramatically and said, "I swear, this biochemistry is trying to break me. It's all these fancy words… like 'peptide bonds' and 'pentapeptides.' What even is a pentapeptide? And how many of these 'peptide bond' things are lurking in it?" Her friend, bless his soul, just shrugged, looking equally lost. I chuckled to myself, grabbed my latte, and thought, "Ah, the joys of learning biochemistry!" It’s like trying to decipher a secret code, isn't it? But don't worry, Maya and her friend, and anyone else who's ever felt that little pang of confusion, I'm here to break it down for you. No advanced degrees required, just a curious mind and maybe a snack.
You see, the world of proteins and peptides can seem a bit intimidating at first. They’re these fundamental building blocks of life, and understanding how they're put together is like learning the alphabet before you can read a novel. And at the heart of this building process are… you guessed it, peptide bonds! They’re the tiny, yet mighty, connectors that hold these chains of amino acids together. Think of amino acids as individual LEGO bricks, and peptide bonds are the little bumps and holes that snap them into place, forming a longer, more complex structure. Pretty cool, right?
Now, Maya specifically mentioned "pentapeptide." That word itself gives us a HUGE clue, doesn't it? Like a detective spotting a fingerprint at a crime scene. The prefix "penta-" is a pretty universal signal for the number five. We see it everywhere – think of a pentagon (a shape with five sides), or a pentameter (a line of verse with five metrical feet). So, a pentapeptide, by its very name, tells us it's a peptide made of five amino acids. Simple as that!
But the real question, the one that had Maya and her friend scratching their heads, was about the peptide bonds. How many of these crucial connections are actually in a pentapeptide? This is where we need to put on our slightly more analytical hats, but don't worry, we're not going to need calculators, just a bit of logical thinking. It’s like counting the number of links in a chain you're making. If you have five beads (amino acids) and you want to connect them all in a line, how many connections do you need?
The LEGO Analogy Strikes Again!
Let’s go back to our trusty LEGO analogy. Imagine you have five individual LEGO bricks. You want to connect them end-to-end to make a single, longer brick. How many times do you have to snap them together?
You pick up the first brick. Then you take the second brick and snap it onto the first. That's one connection, right?
Now you have a chain of two bricks. You take the third brick and snap it onto the end of the second. That’s your second connection.
You continue this process. The fourth brick snaps onto the third (third connection). And finally, the fifth brick snaps onto the fourth (fourth connection).
See? You have five bricks, and you made four connections to link them all together. It's not five connections, it's one less than the number of individual units.
Peptides: The Backbone of Proteins
This is precisely how peptide bonds work in biochemistry. When amino acids join together to form a peptide or a protein, they link up sequentially. Each link is a peptide bond. So, if you have a chain of 'n' amino acids, you will have 'n-1' peptide bonds holding them together.
This is a fundamental principle in understanding the structure of peptides and proteins. Proteins are just very, very long chains of amino acids, and the number of peptide bonds in a large protein can be in the hundreds or even thousands! Mind-boggling, isn't it?
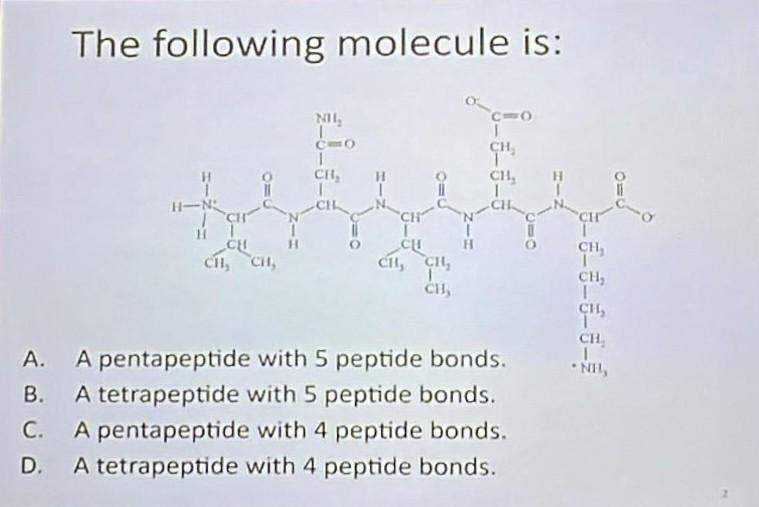
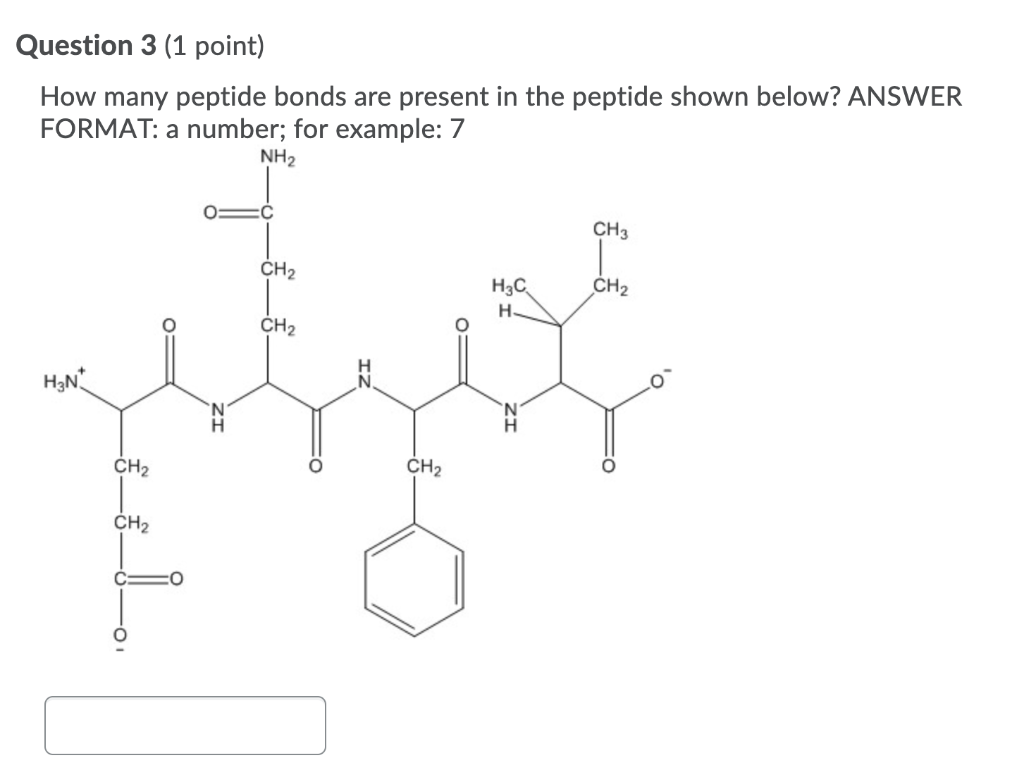
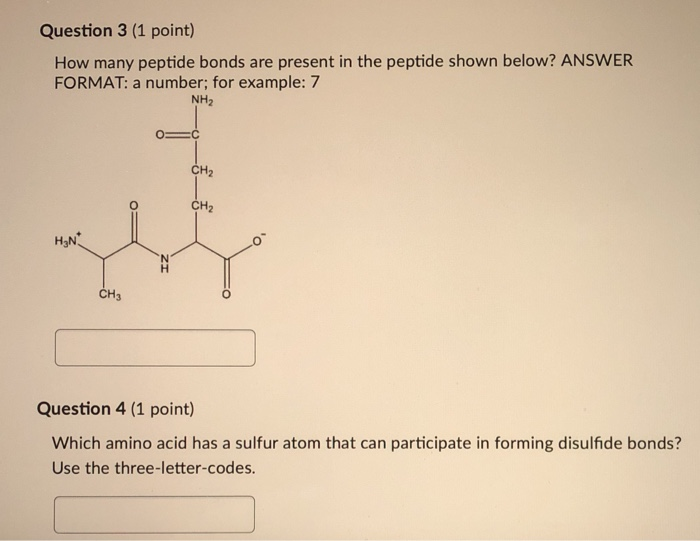
For our specific case, the pentapeptide, we have five amino acids. Applying our little formula (n-1), we get 5 - 1 = 4. Therefore, a pentapeptide contains four peptide bonds.
It's like when you're baking a loaf of bread. If you want to make a loaf with five slices, you need to make four cuts in the dough before baking. The cuts represent the peptide bonds, and the slices represent the amino acids. Each cut creates two pieces from one, just as each peptide bond links two amino acids together.
How Does a Peptide Bond Actually Form? (The Sciencey Bit, but not too sciencey)
Alright, let's peek under the hood just a tiny bit, shall we? It's not as scary as it sounds, I promise! Peptide bonds are formed through a chemical reaction called a dehydration reaction (also known as a condensation reaction). This is where a molecule of water (H₂O) is removed when the bond is formed. Hence, "dehydration."
Each amino acid has a central carbon atom, an amino group (-NH₂), a carboxyl group (-COOH), a hydrogen atom, and a side chain (R-group) that makes each amino acid unique. When two amino acids come together, the carboxyl group of one amino acid reacts with the amino group of the other amino acid.

Specifically, a hydroxyl group (-OH) from the carboxyl group and a hydrogen atom (-H) from the amino group are removed, forming a molecule of water. The remaining carbon atom from the carboxyl group and the nitrogen atom from the amino group then form a covalent bond – the glorious peptide bond (-CO-NH-).
It's a pretty neat trick of chemistry, allowing these building blocks to link up and create the vast diversity of molecules that make up living organisms. Think about it: the same basic reaction forms the peptides in your muscles, the enzymes that digest your food, and the signaling molecules that communicate messages in your body.
The Directionality of Peptides
Another cool thing about peptides and proteins is their directionality. They have a beginning and an end. One end has a free amino group, and the other end has a free carboxyl group. The end with the free amino group is called the N-terminus (N for Nitrogen), and the end with the free carboxyl group is called the C-terminus (C for Carbon).
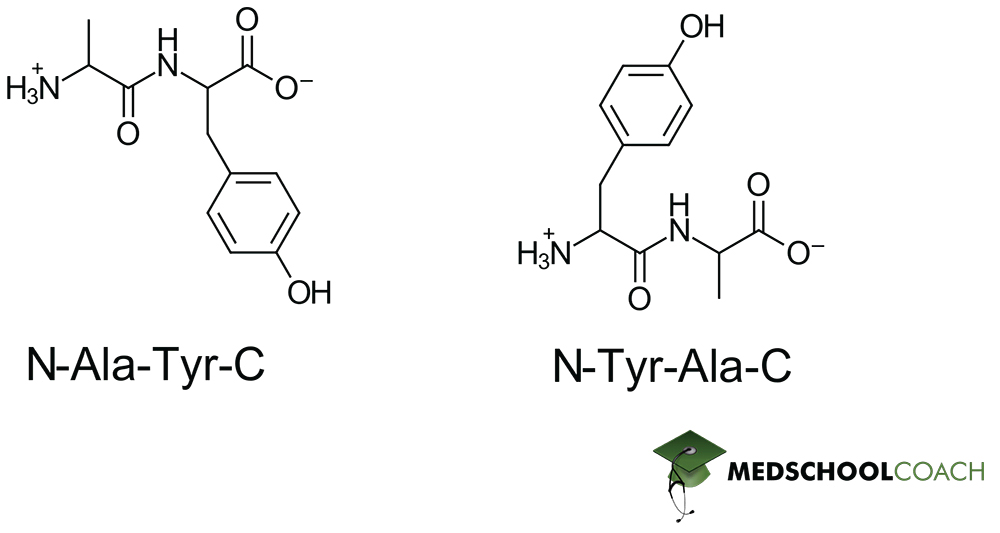
When we talk about a pentapeptide, we're talking about a chain of five amino acids linked in a specific sequence. For example, if we have amino acids A, B, C, D, and E, the pentapeptide might be written as A-B-C-D-E. The peptide bonds would be between A and B, B and C, C and D, and D and E. That's our four peptide bonds!
The order matters! A-B-C-D-E is a different molecule with different properties than, say, E-D-C-B-A. It's like arranging letters to form words. "STOP" is very different from "POTS," even though they use the same letters. This sequence specificity is what gives proteins their unique functions.
Why Does This Even Matter? (Beyond the Coffee Shop Convo)
So, why are we nerding out about pentapeptides and peptide bonds? Well, understanding these fundamental structures is crucial for so many things in biology and medicine. For instance:
- Drug Development: Many drugs are peptides or are designed to interact with peptides. Knowing how they are constructed helps researchers create more effective treatments. Think of insulin, a peptide hormone that is vital for managing diabetes.
- Understanding Diseases: Certain diseases are linked to the misfolding or malfunction of proteins, which are made of many amino acids linked by peptide bonds. Studying these bonds helps us understand the root causes of these conditions.
- Nutrition: Proteins are essential nutrients, and they are broken down into amino acids in our digestive system. Understanding peptide bonds helps us grasp how our bodies process and utilize protein.
- Biotechnology: In the lab, scientists synthesize peptides for various research purposes, from creating antibodies to developing new materials.
It’s like knowing how to build with LEGOs. Once you understand the basic brick and how it connects, you can build anything from a small car to a giant castle. Similarly, understanding peptide bonds allows us to understand the incredible complexity and diversity of life itself.
A Little Irony for Your Day
You know, it’s funny. We’re talking about these incredibly complex molecular structures, the very essence of life, and it all boils down to a simple subtraction problem: n-1. It’s a touch ironic, isn’t it? That something so fundamental and so intricate can be described by such a basic mathematical relationship. It’s a reminder that sometimes, the most profound answers are found in the simplest of observations. Just like Maya and her friend were a bit stuck, but the answer was just a hop, skip, and a minus-one away!
So, the next time you hear the word "pentapeptide," you can confidently say, "Ah, yes, that's five amino acids linked by four peptide bonds!" You've conquered a small piece of the biochemistry puzzle. Give yourself a pat on the back, maybe grab a coffee (or a latte, if you're feeling fancy), and appreciate the elegant simplicity that underlies the complexity of life. And remember, even the most daunting scientific concepts can often be broken down with a bit of curiosity and a good analogy. Now, go forth and impress your friends with your newfound peptide bond knowledge!
