How Many Orbitals Are There In The 4p Subshell
Ever found yourself staring up at the night sky, wondering about the hidden structures that make up everything around us? It's a pretty grand thought, right? Well, the universe has its own intricate organizational system, and understanding it, even in the smallest ways, can be incredibly fascinating. Today, we're diving into a tiny but mighty part of that system: the 4p subshell. It might sound super technical, but stick with me, because learning about it is like unlocking a secret code to how atoms behave, and honestly, it's a pretty fun piece of trivia for any curious mind.
So, what's the big deal with a "subshell"? Think of an atom like a miniature solar system. The electrons, instead of orbiting the nucleus in neat, predictable paths, are found in different "energy levels" or "shells." Within each shell, there are even more specific regions where electrons are likely to be found, and these are called orbitals. The 4p subshell is a particular arrangement of these orbitals within the fourth energy level. Its purpose is to house a specific number of electrons in a particular shape and orientation, influencing how atoms interact and bond with each other.
Understanding the number of orbitals in a subshell, like the 4p, helps scientists predict and explain all sorts of chemical phenomena. For instance, it's crucial for understanding chemical bonding – how atoms join forces to create molecules like water or the complex structures in our own bodies. In education, this knowledge forms the bedrock of chemistry and physics curricula, helping students grasp concepts from basic atomic structure to advanced quantum mechanics. You might not see a "4p subshell" on your grocery list, but its principles are at play in the materials that make up your phone, the medicines that keep you healthy, and even the way plants perform photosynthesis. It's the invisible architecture of the tangible world!
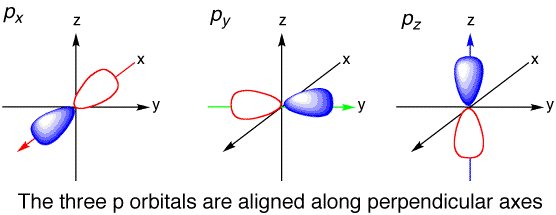
The beauty of this topic is that you don't need a fancy lab to appreciate it. When you're learning about atomic structure in school, you'll encounter diagrams showing these different shells and subshells. Pay close attention to how the 'p' subshell is typically represented. You'll notice it usually consists of three distinct, dumbbell-shaped orbitals. That's right, the 4p subshell, just like any other 'p' subshell (2p, 3p, etc.), contains three orbitals. Each of these orbitals can hold a maximum of two electrons, meaning the 4p subshell can accommodate a total of six electrons. It's a simple, elegant structure that has profound implications.
Want to explore this further without getting bogged down in complex math? Look up simple animations or diagrams of atomic orbitals online. You'll see these 'p' orbitals depicted as three lobes oriented along the x, y, and z axes. Imagine them as three balloons tied together at the center, pointing in three different directions. It's a wonderfully visual way to grasp a fundamental concept in atomic physics. So, the next time you encounter the term "4p subshell," you'll know it's home to a well-defined set of three orbitals, each ready to play its part in the grand molecular dance of the universe.
