How Many Moles Of Bromine Gas Are In 37.7 Grams

Okay, so picture this: I'm in my kitchen, trying to make this fancy new recipe for, like, lavender-infused sea salt caramel. Sounds gourmet, right? Anyway, the recipe calls for a pinch of something. A pinch! And I'm staring at this tiny little jar of this super-concentrated, almost iridescent purple powder. It smells… well, it smells like a chemistry lab after a particularly enthusiastic experiment. And my first thought is, "How much is a pinch, really?" Is it five crystals? Ten? A hundred? My brain just… short-circuits. Precision is not my strong suit when it comes to kitchen alchemy. So, I end up just eyeballing it, hoping for the best. Thankfully, it was fine. But it got me thinking. Sometimes, in science, we deal with things in quantities that are just as mind-bogglingly small, or in amounts that seem ridiculously large, and we need a way to talk about them that makes sense. It's not just about "a pinch" or "a smidge"; it's about knowing the exact amount. And that, my friends, is where our trusty concept of the
You know, sometimes I feel like science tries to trick us with its big words. Like, "mole." My first thought? Fuzzy little garden critters. But no, in chemistry, a mole is something totally different. It's basically a unit of measurement, like a dozen eggs or a ream of paper. Except, instead of eggs or paper, it’s for… well, atoms and molecules. And the number is, let's just say, a tad bigger than a dozen.
So, if you've ever found yourself wondering how many of those teeny-tiny building blocks of matter are hanging out in a certain amount of stuff, you're in the right place. Today, we're going to dive into the fascinating world of bromine gas and figure out, with a little bit of calculation and a whole lot of curiosity, just how many moles are lurking in a specific amount of it. Don't worry, no actual lab coats or bubbling beakers required for this little adventure. We're keeping it strictly mental, and hopefully, a little bit fun.
The Tiny Giants We Can't See
Let’s be honest, the world of atoms and molecules is a bit like a secret society. They’re everywhere, they’re doing all the important stuff, but you can’t actually see them. Not with your regular eyes, anyway. You need some seriously fancy equipment for that. And that’s where the mole becomes our superhero. It’s the way scientists, and now us, can count these invisible participants in everyday reactions and substances.
Think about it. If you had a bag of marbles, you could count them easily. One, two, three… you get the idea. But if you had a bag of atoms? Forget about it. Even a tiny speck of dust contains billions upon billions of atoms. Trying to count them individually would be… well, a lifetime (or several) well spent and still incomplete. So, we needed a shortcut. A clever way to group them into manageable numbers.
And that, my curious companions, is Avogadro's number. It’s a ridiculously large number: 6.022 x 1023. So, if you were to write it out, it would be a 6 followed by 23 zeros. Twenty-three zeros! Imagine that. That’s how many particles (atoms, molecules, ions, whatever you’re counting) are in one mole. It’s like a cosmic odometer for the microscopic world. Pretty wild, right?
So, when we talk about moles, we’re really talking about this specific, humongous number of particles. It’s a chemical convention, a way to bridge the gap between the macroscopic world we can measure (like grams) and the microscopic world of atoms and molecules. It’s the chemist’s dozen, but on an epic, universe-sized scale.
Bromine: The Colorful, Slightly Feisty Element
Now, onto our star of the show (or at least, the gaseous star): bromine. Bromine is a chemical element, with the symbol Br. It’s a halogen, which means it likes to hang out with other elements in Group 17 of the periodic table. Think of it as part of a slightly dramatic family – fluorine, chlorine, iodine, and astatine are its siblings. They're all quite reactive, and bromine is no exception.
At room temperature, bromine is a bit of a showstopper. It's the only non-metal element that’s a liquid under these conditions. And it's not just any liquid; it's a dense, reddish-brown, volatile liquid. Volatile, in chemistry terms, means it evaporates easily, producing a reddish-brown vapor. So, if you ever see that color and think "huh, that's unusual," it might just be bromine doing its thing. It's also known for its pungent, irritating odor. So, definitely not something you want to be taking deep inhales of without proper safety precautions! It's a bit like that friend who has a really strong personality – you notice them, and they can be a little overwhelming.

When we're talking about bromine gas, we're usually referring to diatomic bromine, which is written as Br2. This means that in its gaseous state, bromine exists as molecules, with two bromine atoms bonded together. It's like the bromine atoms decided to hold hands and go on an adventure together in the gas phase. This diatomic nature is super important when we do our calculations, so keep that in the back of your mind. It’s the difference between counting individual atoms and counting pairs of atoms, which changes our overall numbers.
Bromine has a variety of uses, from flame retardants to water purification, and even in some pharmaceuticals. It’s a bit of a workhorse element, despite its slightly intimidating nature. And understanding how much of it we have in terms of moles helps us understand its potential reactions and applications. It’s all about quantifying the invisible!
The Bridge Between Mass and Moles: Molar Mass
So, we have grams (which we can weigh on a scale, like my sugar for cookies) and we have moles (which are our giant counting units for atoms and molecules). How do we get from one to the other? This is where the concept of molar mass comes to the rescue. It’s the bridge, the magical translation tool.
The molar mass of a substance is essentially the mass of one mole of that substance, expressed in grams per mole (g/mol). It’s like saying, "If I have a dozen eggs, they weigh approximately X grams." For elements, the molar mass is numerically equal to their atomic mass found on the periodic table. But remember, we're dealing with bromine gas, which is Br2. So, we need to consider the mass of two bromine atoms.
Let’s do a quick check on the periodic table. The atomic mass of bromine (Br) is approximately 79.904 grams per mole. Since bromine gas is Br2, one mole of bromine gas contains two moles of bromine atoms. Therefore, the molar mass of bromine gas (Br2) is:
Molar Mass of Br2 = 2 * (Atomic Mass of Br)
Molar Mass of Br2 = 2 * (79.904 g/mol)
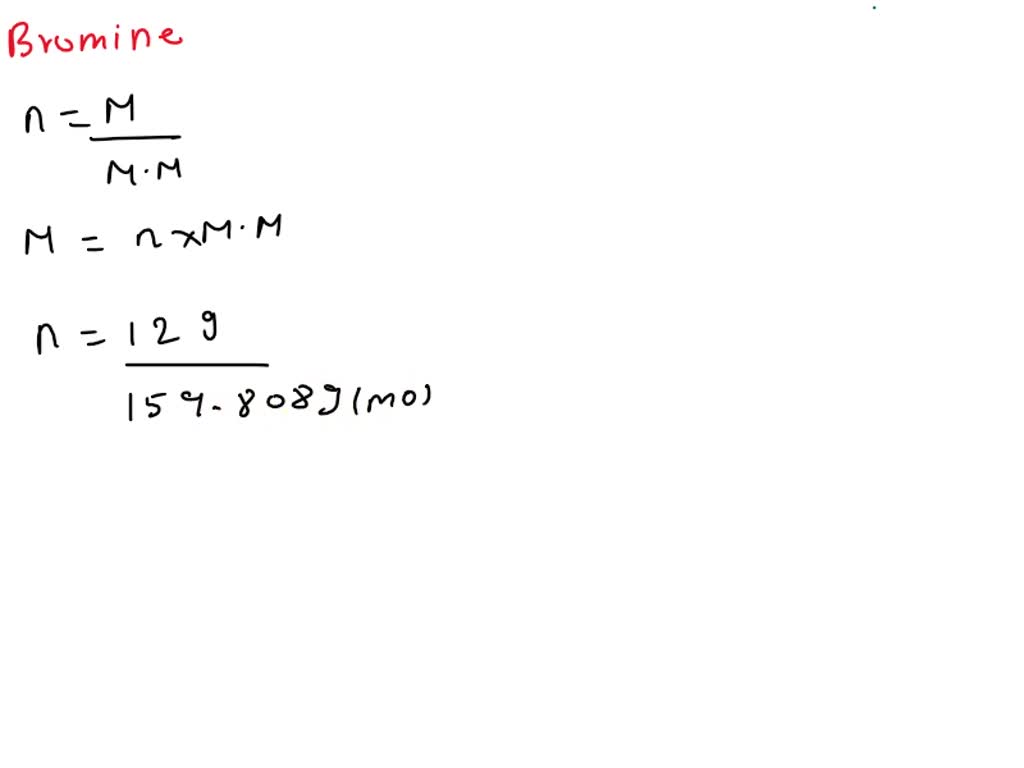
Molar Mass of Br2 = 159.808 g/mol
So, for every one mole of bromine gas molecules, you’ve got about 159.808 grams of it. This number is our key to unlocking the mystery of how many moles are in our given 37.7 grams.
This is the beauty of chemistry, right? We take these abstract concepts and give them concrete values that we can work with. It’s like having a secret code to understand the universe at its most fundamental level. And the molar mass is a crucial part of that code.
Cracking the Code: Calculating Moles of Bromine Gas
Alright, time for the main event! We have 37.7 grams of bromine gas, and we know that 159.808 grams of bromine gas equals 1 mole of bromine gas. We want to find out how many moles are in our 37.7 grams. This is a classic conversion problem, and it’s surprisingly straightforward once you have the right tools.
The formula we use to convert mass to moles is:
Number of Moles = Mass (in grams) / Molar Mass (in grams/mole)
Let’s plug in our numbers:
Number of Moles = 37.7 g / 159.808 g/mol
Now, for the moment of truth. Let’s grab our trusty calculator (or do some fancy mental math if you're feeling brave!).
37.7 divided by 159.808 is approximately 0.23589…
So, in 37.7 grams of bromine gas, there are approximately 0.236 moles of bromine gas (rounding to three significant figures, which is usually a good practice in science unless otherwise specified).
Isn't that neat? We went from a weight measurement, something tangible, to a count of invisible particles. It’s like looking at a pile of Lego bricks and figuring out exactly how many complete sets you have based on their total weight. A little bit of math, and suddenly, the microscopic world becomes a little more… countable.
It's important to note that this calculation is an approximation because the molar mass we used is based on the average atomic mass of bromine. But for most practical purposes, this is perfectly accurate. The beauty of science is that even with approximations, we can still get incredibly precise answers about the world around us.

Why Does This Even Matter?
You might be thinking, "Okay, that's cool, but why do I need to know how many moles of bromine gas are in 37.7 grams?" And that’s a fair question! It’s not like you’re going to be measuring out bromine gas for your morning coffee. (Please, for the love of all that is good, do not do that.)
But this skill, this ability to convert between mass and moles, is fundamental to so many areas of chemistry. It's the backbone of understanding chemical reactions. When chemists design experiments, they need to know precisely how much of each reactant to use to get the desired product. They don't just guess. They calculate in moles.
Imagine you're baking that lavender sea salt caramel again, but this time, you need a specific amount of vanilla extract. If the recipe said, "Add 0.05 moles of vanilla extract," you’d need to know how to convert that into a measurable amount, like milliliters or grams. The mole concept is the universal language of chemistry, allowing scientists to communicate and replicate experiments accurately, no matter what language they speak.
It also helps us understand the scale of things. 0.236 moles of bromine gas might not sound like much, but remember what a mole is. That's 0.236 times 6.022 x 1023 molecules of Br2. That's a lot of tiny, gas-filled packages! It gives us a real appreciation for just how much matter is packed into seemingly small quantities.
So, while you might not be weighing out bromine gas in your daily life, the principles you've just explored are the same ones that drive scientific discovery, industrial processes, and even the development of new medicines. It’s all about understanding the building blocks and how they interact, and the mole is our essential tool for doing just that.
And hey, next time you hear about a chemical reaction or a scientific measurement, you’ll have a little bit of insight into the invisible world that makes it all happen. You’ll know that behind those numbers, there’s a whole universe of atoms and molecules, meticulously counted and understood, thanks to our little friend, the mole.
So, to recap: we’ve gone from a relatable kitchen mishap about imprecise measurements to the mind-boggling scale of atoms, learned about the feisty element bromine, understood the crucial role of molar mass, and finally, calculated the number of moles in a given mass. It’s a journey, right? And it all started with a simple question about quantity. Pretty cool stuff, if you ask me. Keep asking those "how many" questions, because sometimes, they lead to the most interesting discoveries!
