How Many Moles Are Represented By 11.5 G Of C2h5oh
So, picture this: I'm rummaging through my kitchen cabinets the other day, looking for that one specific spice I know I have. You know the one, the mysterious jar that only seems to appear when you're in the middle of a complex recipe and suddenly realize you're missing a crucial element. I'm talking about that moment of mild panic, right? Anyway, I stumble across a dusty old bottle of ethanol. You know, the stuff you might find in a science lab, or maybe for cleaning certain things. It wasn't a giant industrial-sized container, just a regular, albeit slightly forgotten, bottle. My first thought was, "Huh, wonder how much of this is actually in here?" And then, my brain, which apparently has a persistent chemistry background hum, immediately jumped to: "Okay, but in moles?"
It’s a weird thought, I grant you. Most people would probably just think, "It's a bottle of liquid, there's some in there." But that's where the fun begins, isn't it? That little voice in the back of your head, the one that’s always asking "but why?" or "what does that mean?" That’s the voice that turns a simple observation into a mini-science lesson. And today, my friends, we're going on a little journey with that voice. We're going to figure out how many of those tiny, invisible, yet incredibly important, chemical building blocks – moles – are represented by a seemingly ordinary amount of a common substance: 11.5 grams of ethanol, or C2H5OH.
Now, before you start picturing little Lego bricks of ethanol floating around, let's get one thing straight. A mole isn't a physical thing you can hold in your hand. It's a unit of measurement. Think of it like a baker's dozen. A dozen eggs is 12 eggs, right? A baker's dozen is 13. A mole is just a really, really, ridiculously big number. It's Avogadro's number, to be precise: approximately 6.022 x 1023. Yeah, that's a six with 23 zeros after it. It's the number of "things" – atoms, molecules, ions, whatever – in one mole of a substance. So, when we talk about moles, we're talking about an immense quantity of incredibly tiny particles.
Why do chemists even bother with such a colossal number? Well, because atoms and molecules are so minuscule. Imagine trying to count individual grains of sand on a beach. It's impossible! Chemists need a way to group these tiny particles into manageable quantities that we can actually weigh and work with. The mole provides that bridge between the microscopic world of atoms and molecules and the macroscopic world of grams and liters that we experience.
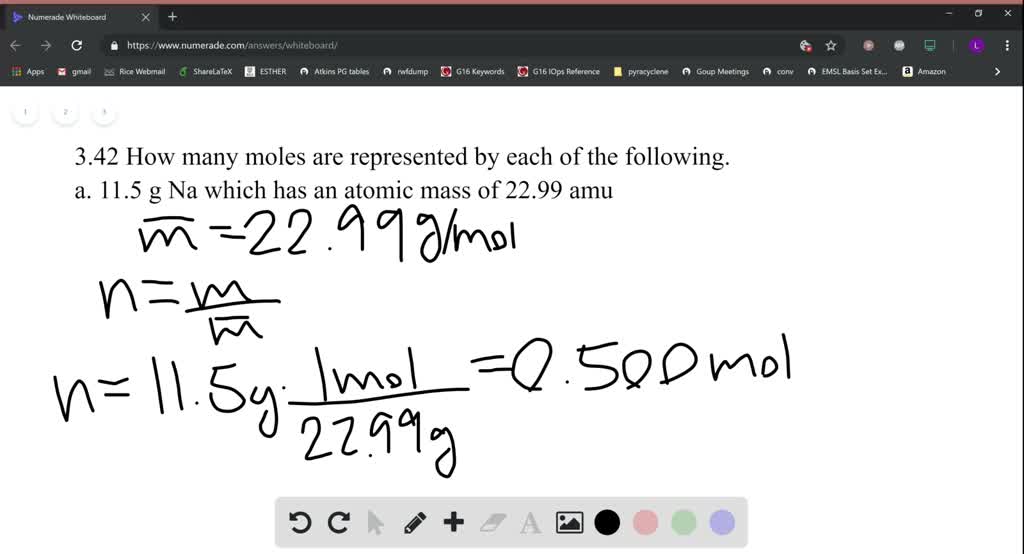
So, back to our dusty bottle. Let's say I actually scooped out exactly 11.5 grams of that ethanol. How do we translate that weight into a number of moles? This is where the concept of the molar mass comes into play. Every substance has a unique molar mass, which is essentially the weight of one mole of that substance. It's expressed in grams per mole (g/mol).
To find the molar mass of ethanol (C2H5OH), we need to look at the atomic masses of each element present in its formula. We’ll need our trusty periodic table for this, the chemist's best friend (alongside a good calculator, of course!).
Ethanol has the chemical formula C2H5OH. Let's break that down:
- There are two carbon (C) atoms.
- There are five hydrogen (H) atoms in the ethyl group, plus one more in the hydroxyl group, making a total of six hydrogen atoms.
- There is one oxygen (O) atom.
Now, let's grab those approximate atomic masses from the periodic table:
- Carbon (C): approximately 12.01 g/mol
- Hydrogen (H): approximately 1.01 g/mol
- Oxygen (O): approximately 16.00 g/mol
Ready for some math? Don't panic, it's the good kind of math! We multiply the number of atoms of each element by its atomic mass and then add them all up to get the molar mass of ethanol.
![[ANSWERED] How many moles are there in 5 00 g of ethanol CH3CH OH MM 46](https://media.kunduz.com/media/sug-question-candidate/20220614223439817981-4627221.jpg?h=512)
For carbon: 2 atoms * 12.01 g/mol = 24.02 g/mol
For hydrogen: 6 atoms * 1.01 g/mol = 6.06 g/mol
For oxygen: 1 atom * 16.00 g/mol = 16.00 g/mol
Now, let's sum these up:
Molar mass of C2H5OH = 24.02 g/mol + 6.06 g/mol + 16.00 g/mol = 46.08 g/mol
So, one mole of ethanol weighs approximately 46.08 grams. This is a crucial piece of information, like knowing the weight of a single golf ball if you're trying to figure out how many golf balls are in a bucket. You can't just guess; you need that reference point.

We've got our target weight: 11.5 grams of ethanol. And we know the weight of one mole: 46.08 grams. The question is, how many of these "46.08 gram units" are contained within our 11.5 grams? It's a division problem, plain and simple!
The formula to convert grams to moles is:
Number of moles = Mass (in grams) / Molar mass (in g/mol)
Let's plug in our numbers:
Number of moles = 11.5 g / 46.08 g/mol
And when you crunch those numbers (grab your calculator, no shame in that game!):

Number of moles ≈ 0.249566 moles
So, 11.5 grams of ethanol represents approximately 0.250 moles. See? Not so scary after all!
What does this 0.250 moles mean in terms of actual molecules? Well, remember that Avogadro's number? We just figured out we have about a quarter of a mole. So, the number of ethanol molecules in 11.5 grams would be:
0.249566 moles * 6.022 x 1023 molecules/mol ≈ 1.50 x 1023 molecules of ethanol.
That's 150,000,000,000,000,000,000,000 molecules! Mind-boggling, right? Just a little bit of liquid holding an astronomical number of tiny particles. It’s like finding a tiny pebble on the beach and realizing it’s made up of an almost infinite number of microscopic grains. It really puts things into perspective.
This kind of calculation is fundamental to pretty much everything in chemistry. Whether you're cooking up a new pharmaceutical, designing a new material, or even just trying to understand how a reaction happens in your own body, you’re going to be dealing with moles. It's the currency of the chemical world.
Think about it in a more relatable way. Imagine you're baking cookies, and the recipe calls for, say, 2 cups of flour. Now, you could try to count out individual grains of flour, but that's utterly impractical. Instead, we have units like "cups" or "grams" that represent a manageable quantity of flour. Moles are the chemical equivalent of those units, but instead of counting grains, we're counting molecules.
And that's why understanding molar mass and how to convert between grams and moles is so darn important. It allows us to move from what we can measure (like the weight of a substance) to what's actually happening at the molecular level. It’s the key to unlocking the secrets of chemical reactions and the composition of matter.
So, the next time you see a quantity of a chemical, whether it's in a lab or just a common household item (like my forgotten ethanol bottle), don't just see the grams. Ask yourself, "How many moles is that?" Because within those grams lies a universe of tiny particles, and the mole is your map to navigating it.
It's also worth noting that precision matters in science. Our molar mass calculation used approximate atomic masses. For highly precise work, you'd use more decimal places for the atomic masses. But for most general chemistry calculations, these approximations are perfectly fine and give you a very good understanding of the number of moles involved.
The world of chemistry can seem daunting with all its formulas and numbers, but at its heart, it's about understanding the building blocks of everything around us. And the mole is one of the most powerful tools we have for that understanding. It’s the concept that allows us to make sense of the invisible world and quantify the immeasurable.
So, to recap our little kitchen adventure: 11.5 grams of ethanol is equivalent to about 0.250 moles. That means you have approximately 1.50 x 1023 molecules of ethanol. It's a small number of grams representing a colossal number of molecules. And that, my friends, is the beauty and wonder of the mole concept!
Now, if you'll excuse me, I might actually need to find that spice. This whole detour has made me want to do some actual baking. Maybe with some (measured) ethanol for a special effect. Just kidding… mostly!
