How Many Molecules Are There In 2.30 Grams Of Nh3

Get ready for a mind-blowing adventure into the teeny-tiny world of chemistry! We're about to dive headfirst into a question that might sound a little bit like a riddle, but trust me, it's going to be so much fun. We're going to figure out just how many molecules are packed into a tiny, unassuming pile of ammonia, or as the chemists affectionately call it, NH3. Imagine holding a pinch of salt – now imagine that pinch is actually bursting with more tiny bits than you can even begin to count! That’s the kind of magic we’re exploring today.
So, you've got this 2.30 grams of NH3. Sounds like a pretty small amount, right? Maybe something you'd barely notice on a kitchen scale. But hold onto your hats, because what’s hidden inside that little pile is utterly, fantastically enormous. We're talking about a number so big, it makes the number of stars in the entire universe look like a shy little speck in comparison. Yes, you read that right! Get ready to have your jaw drop.
First things first, let's chat about what a molecule actually is. Think of it like a LEGO brick, but on a scale so small, you'd need the most powerful microscope in the galaxy to even catch a glimpse. A molecule is the smallest unit of a substance that still has the properties of that substance. For ammonia (NH3), it's made up of one nitrogen atom and three hydrogen atoms all holding hands in a special, cozy arrangement. These little LEGOs are what make up our 2.30 grams of NH3.
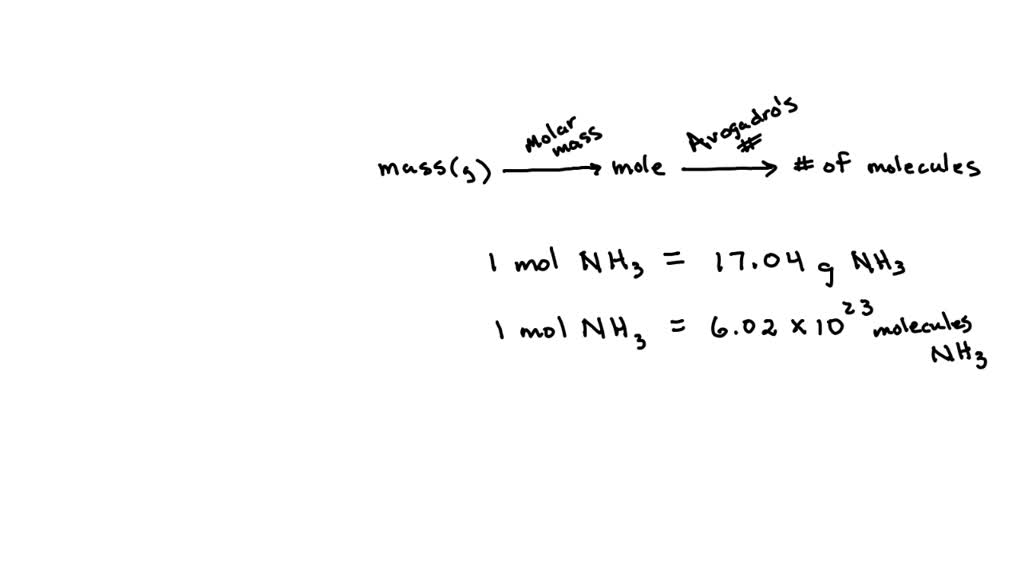
Now, how do we go from grams (which we can see and feel) to molecules (which are practically invisible)? This is where a super-duper important tool in chemistry comes into play: the mole. Don't let the name fool you; it's not about those creepy, wriggly things that live in gardens! In chemistry, a mole is a unit of counting. It's like saying "a dozen" when you mean 12 eggs. A mole just means a huge number of things.
And what's that huge number? It's called Avogadro's Number, and it's approximately 6.022 x 1023. Say that number out loud! Go on, give it a try! It's 602,200,000,000,000,000,000,000. It’s a 6 followed by 23 zeros! If you had a mole of tiny little pebbles, you could build a mountain range taller than Mount Everest, and then some. It’s an almost unfathomably large quantity, and it’s the secret handshake between the world of grams and the world of molecules.
So, a mole of anything is exactly 6.022 x 1023 of that thing. Whether it’s a mole of electrons, a mole of donuts (oh, wouldn't that be amazing!), or a mole of ammonia molecules, it's always that same mind-boggling number of individual items. This is the key to unlocking our mystery. We need to figure out how many moles of NH3 we have in our 2.30 grams.
To do that, we need to know the "weight" of one mole of NH3. This is called the molar mass. Think of it as the specific weight of a baker's dozen of those LEGO molecules. We find this by adding up the atomic weights of all the atoms in the molecule. For NH3, it's one nitrogen atom (which weighs about 14.01 grams per mole) plus three hydrogen atoms (each weighing about 1.01 grams per mole). So, 14.01 + (3 x 1.01) = 17.04 grams per mole. That means 17.04 grams of NH3 is equal to one mole.
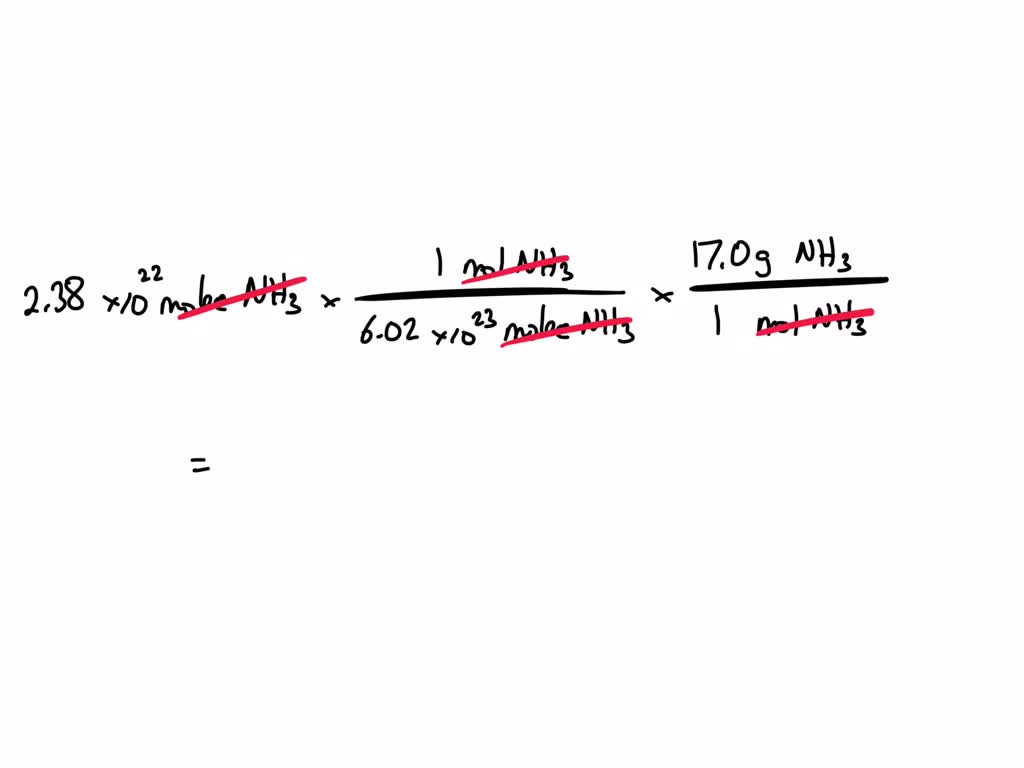
Now, we have 2.30 grams of NH3. We want to know how many moles that is. We can simply divide our sample's weight by the molar mass: 2.30 grams / 17.04 grams/mole. This calculation will tell us how many "dozens" of molecules we have. Drumroll please… it comes out to be approximately 0.135 moles of NH3.
We're getting so close! We know we have about 0.135 moles of ammonia. And we know that one mole is equal to 6.022 x 1023 molecules. So, to find the total number of molecules, we just multiply the number of moles we have by Avogadro's Number! It’s like saying, if you have 0.135 "dozens," and each dozen has 12 items, how many items do you have in total? 0.135 x 12. But here, instead of 12, we have that humongous number: 6.022 x 1023.
So, let's do the final calculation: 0.135 moles x 6.022 x 1023 molecules/mole. This is where the real magic happens! Get ready for a number that will make your head spin in the best possible way.
And the answer is… approximately 8.13 x 1022 molecules of NH3! Yes, you heard that right! In your 2.30 grams of ammonia, there are roughly 81,300,000,000,000,000,000,000 molecules. That's an 8 followed by 22 zeros!
![[ANSWERED] There are molecules in 30.0 grams of NH3. Select the correct](https://media.kunduz.com/media/sug-question/raw/56261918-1659287570.989619.jpeg?h=512)
Just to put that into perspective, imagine trying to count them all. If you could count one molecule every second, without ever stopping, not even for a nap or a snack, it would take you… wait for it… billions of years. Seriously! It would take longer than the age of the universe to count all those tiny little NH3 LEGO bricks!
Think about it: that little sprinkle of ammonia is a bustling metropolis of unimaginable tiny particles, all zipping around and doing their chemical thing. It’s a hidden universe within your grasp. Every time you encounter a substance, whether it's water, sugar, or even the air you breathe, it's teeming with these incredible numbers of molecules. It’s a constant reminder of the astonishing complexity and sheer abundance of matter all around us.
Isn't that just the coolest thing ever? Chemistry, at its heart, is about understanding these fundamental building blocks and how they interact. And sometimes, the most ordinary-looking things contain the most extraordinary amounts of… well, everything! So, the next time you hear about grams or moles, remember the mind-boggling universe that's packed inside.
We've gone from a tiny 2.30-gram sample to a number so vast it’s almost comical. We've learned about molecules, the building blocks of everything. We've met the mighty mole and its superhero, Avogadro's Number. And we've calculated a number so enormous it deserves its own universe.
So, there you have it! The answer to our fun chemical puzzle. You've successfully navigated the world of atomic weights, molar masses, and the incredible scale of the molecular realm. Feel that sense of wonder? That's the feeling of understanding something truly amazing. Go forth and share your newfound appreciation for the countless molecules that make up our world!
The sheer number of molecules in even a tiny amount of substance is a testament to the incredible scale of the microscopic world. It's like discovering a secret, bustling city hidden within a single grain of sand!
So, to recap: 2.30 grams of NH3 isn't just a small amount of stuff. It's a vibrant, bustling community of approximately 8.13 x 1022 individual ammonia molecules. Each one doing its own little dance, contributing to the whole. It’s a beautiful, chaotic, and utterly astonishing reality, all thanks to the magic of chemistry and a little bit of math!
