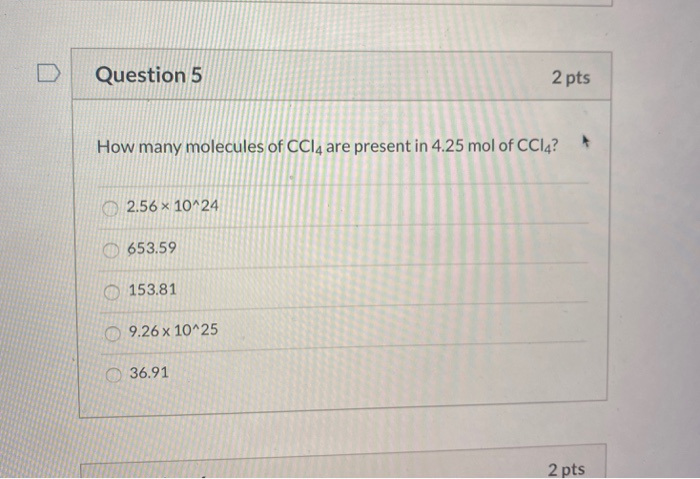
How Many Molecules Are Present In 4.25 Mol Of Ccl4
Hey there, fellow curious minds! Ever found yourself staring at something, perhaps a recipe, a science experiment, or even just a really cool gadget, and wondering, "How does this really work?" That same spark of curiosity often leads us down fascinating paths, and today, we're diving headfirst into the wonderfully microscopic world of chemistry. It might sound intimidating, but understanding a bit about the building blocks of everything around us can be surprisingly satisfying and even quite useful.
So, why do we bother with things like moles and molecules? Well, it’s all about understanding the quantities of substances. Think of a mole as a super-convenient chemist's dozen. Instead of a dozen (12) eggs, a mole is a ridiculously large number, approximately 6.022 x 1023. This number, known as Avogadro's number, helps us count and measure tiny particles like atoms and molecules in a manageable way. This is absolutely crucial for everything from baking the perfect cake (precise ingredient ratios matter!) to developing life-saving medicines, and even in manufacturing the plastics that shape our modern world.
Let’s take a common example: carbon tetrachloride, or CCl4. While less common in everyday household products today due to environmental concerns, it’s historically been used as a solvent and in refrigerants. Understanding how many CCl4 molecules are in, say, 4.25 moles of it, is a fundamental step for chemists working with this compound. It tells them the scale of their reactions and the potential impact of the substance. Imagine a chef needing to know precisely how many grains of salt are in a cup – it’s that kind of precision, just on a much, much smaller scale!
Now, how do we actually figure out how many molecules are in 4.25 moles of CCl4? It’s a straightforward calculation that unlocks the secrets of the microscopic. We use that handy Avogadro's number we talked about. The formula is simple: Number of Molecules = Moles x Avogadro's Number. So, for 4.25 moles of CCl4, we multiply 4.25 by 6.022 x 1023. Get your calculators ready! This gives us a staggering number: 25.5935 x 1023 molecules, or more conventionally written, 2.559 x 1024 molecules of CCl4. That's a lot of tiny particles!
To enjoy this whole "molecule counting" adventure more effectively, embrace the curiosity! Don't be afraid of the numbers. Think of it like solving a puzzle. Start with smaller, more familiar compounds. Many online resources and even basic chemistry textbooks offer practice problems. The more you practice, the more intuitive it becomes. And remember, understanding these fundamental concepts is like learning the alphabet of the universe – once you’ve got it, a whole new world of understanding opens up!
