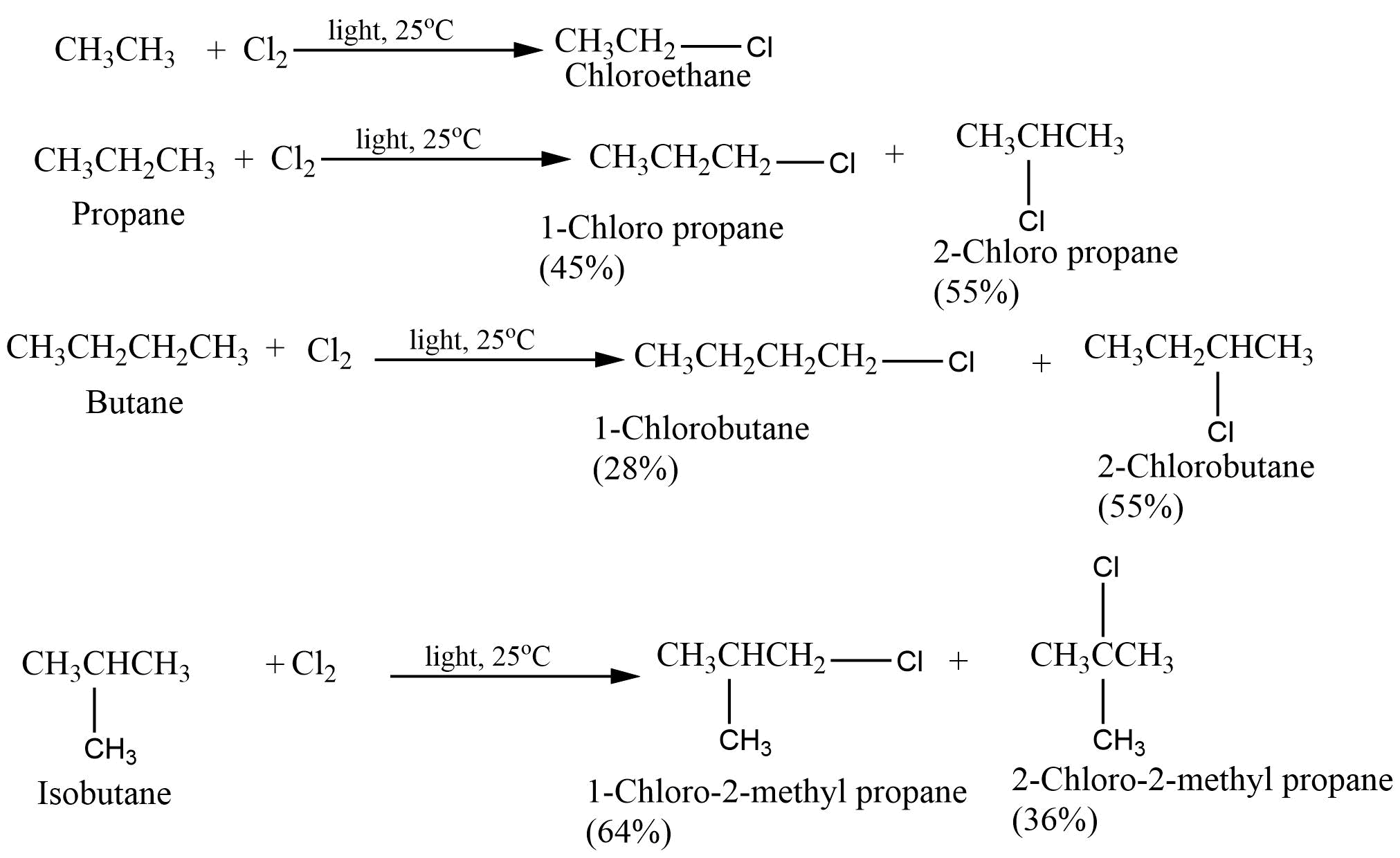
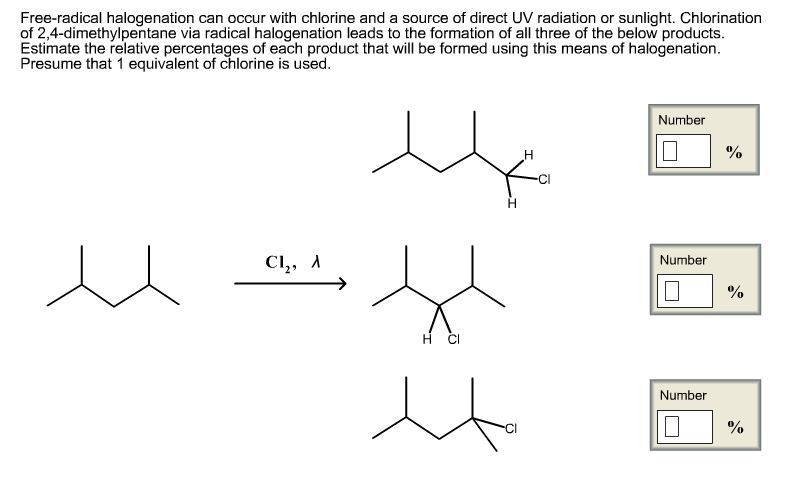
How Many Halogenation Products Can Ch4 Have With Cl2

Ever looked at a molecule and thought, "Wow, that's a lot of potential for change!"? Today, we're going to chat about something that might sound a bit science-y at first glance – the humble methane molecule, affectionately known as CH4, and its dance with chlorine gas, Cl2. Now, don't let the "chemistry" label scare you. Think of it like this: we're going to explore how many different "makeovers" our little methane can get when a chlorine buddy comes along.
Imagine methane as a little kid, let's call him "Mikey." Mikey is super simple. He's got one carbon atom at his core, like a tiny, round head, and four hydrogen atoms, like four little arms reaching out. He's pretty stable, like a perfectly balanced game of Jenga – all the pieces are in just the right place.
Now, imagine chlorine gas, Cl2, as a pair of energetic twins, "Clara and Carl." They're a bit mischievous and love to swap places. When Mikey and Clara and Carl meet, things can get interesting. They don't just go for a polite handshake; they might decide to replace one of Mikey's arms with one of theirs.
So, the first thing that can happen is that one of Mikey's hydrogen arms gets swapped out for a chlorine arm. We're left with a molecule that's a bit like Mikey, but now he's got a chlorine accessory. This is called monochloromethane. Think of it like Mikey getting a cool new blue glove instead of one of his red ones. It's still Mikey, just with a little twist.
But Clara and Carl are twins for a reason – they're eager! If the conditions are right, they might come back and decide to swap out another one of Mikey's remaining hydrogen arms for a chlorine arm. Now Mikey has two chlorine accessories. This is where things start to get a little more complex. We're not just talking about one glove anymore; maybe Mikey now has a blue glove and a green glove. This molecule is called dichloromethane.
Why should you care about this? Well, these molecules, even though they sound like they belong in a lab, are actually pretty common in our world. They're used in all sorts of things, from cleaning solvents (think of that strong smell when you're using certain degreasers) to even being ingredients in some pharmaceuticals. So, understanding how they're made is like understanding how your favorite everyday items come to be.
Let's keep going with our makeover analogy. Mikey can't hold onto all those hydrogen arms forever when Clara and Carl are around. If the chlorine twins keep at it, they can replace a third hydrogen arm. Now Mikey is sporting three chlorine accessories! This molecule is called trichloromethane. Imagine Mikey now has a blue glove, a green glove, and a yellow glove. He's looking quite flamboyant!
And then, the grand finale! If Clara and Carl are really determined, they can replace the very last hydrogen arm on Mikey. Now, Mikey is entirely surrounded by chlorine! This final molecule is called tetrachloromethane (or carbon tetrachloride, if you prefer its older name). It's like Mikey has completely ditched his red gloves and is now rocking a full chlorine suit. He's a whole new person, or rather, a whole new molecule!
So, to recap, our simple Mikey (CH4) can go through a series of transformations with Clara and Carl (Cl2). He can end up with:
1. Monochloromethane:
One chlorine arm. Like Mikey swapping a red glove for a blue one.
2. Dichloromethane:
Two chlorine arms. Mikey now has a blue and a green glove.
3. Trichloromethane:
Three chlorine arms. Mikey is rocking a blue, green, and yellow glove.
4. Tetrachloromethane:
Four chlorine arms. Mikey is in a full chlorine suit!

Each of these is a distinct product, a unique molecule with its own properties and uses. It’s like having four different outfits for Mikey, all created by the same pair of energetic twins.
Now, you might be wondering, "Is this a random process, or is there some control?" In the real world of chemistry, we can often influence how many swaps happen. It's like telling Clara and Carl, "Okay, just one swap today, please!" or "Let's go for the full chlorine makeover!" We can control the conditions, like the temperature and the amount of light, to encourage one product over another.
This is a bit like baking. You can follow a recipe to make a simple cookie (monochloromethane), or you can add more ingredients and bake it differently to get a more complex pastry (say, dichloromethane or beyond). The basic ingredients are there, but the process dictates the final outcome.

The reason this matters to us, even if we're not chemists in a lab coat, is because these halogenated methane compounds have had and continue to have a significant impact on our lives. For instance, some of these compounds, especially tetrachloromethane, were once widely used as cleaning agents and refrigerants. However, we later discovered that they could be quite harmful to our health and the environment. This is why understanding these reactions is crucial for developing safer alternatives and for managing the chemicals we use responsibly.
Think about it like understanding how a car engine works. You don't need to be a mechanic to drive, but knowing the basics helps you appreciate its complexity and understand when something might be going wrong. Similarly, knowing that methane can be converted into these various chlorinated forms helps us understand the chemical landscape around us and the implications of chemical processes.
It’s a fascinating glimpse into the world of molecular transformations, showing how a simple starting point can lead to a variety of different destinations. And all it takes is a little bit of energy and a willing dance partner, like our friendly chlorine gas!
So, the next time you hear about methane or chlorine in the news, or perhaps see a product that uses a chlorinated compound, you'll have a little secret knowledge: you know that simple CH4 molecule can get quite dressed up, with up to four different chlorine outfits!
