How Many Half Filled Orbitals Are In A Bromine Atom
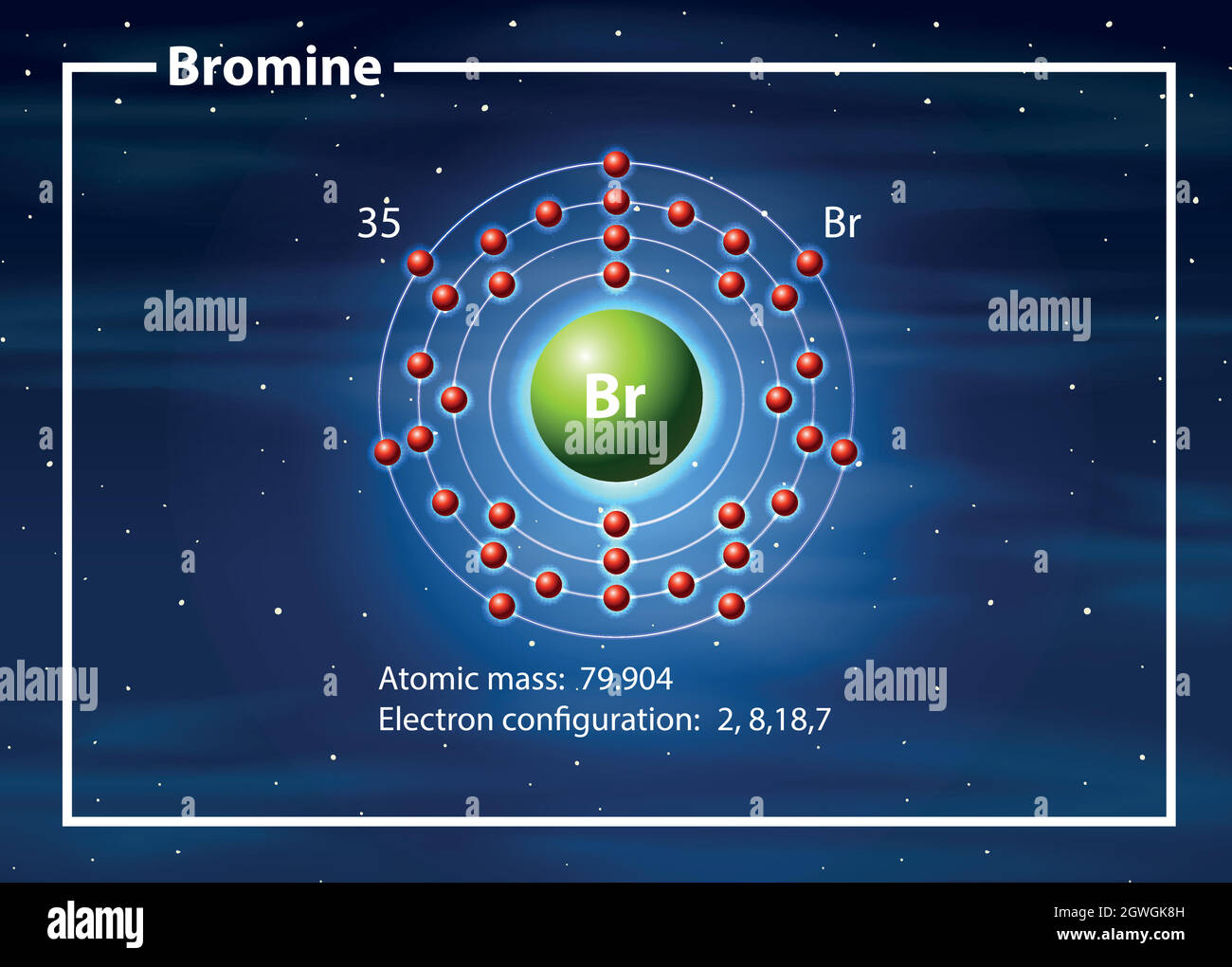
Alright, science fans and occasional science-curious folks! Let's dive into the wonderfully weird world of atoms. We're going to talk about a particularly chatty element today: Bromine.
Now, I know what you're thinking. "Orbitals? Half-filled? This is starting to sound like a college textbook I definitely didn't skim." But stick with me, because we're not here for a lecture. We're here for a little bit of fun.
Think of atoms like tiny, busy little apartments. Inside these apartments are the electrons, always zipping around. And these electrons have their own special rooms, which we call orbitals.
Some of these rooms are perfectly tidy, holding their full capacity of two electrons. Others are a bit more… well, let's just say they're more like a guest room that's only halfway ready. These are our half-filled orbitals.
And the question of the day, the one that keeps some scientists up at night (or maybe they're just really into caffeine), is: How many of these "almost full" rooms does our friend Bromine have?
Let's not get bogged down in the nitty-gritty of electron configurations just yet. Imagine we're peeking into Bromine's atom apartment. We're trying to count the occupants in each room.
It's kind of like trying to count how many people are at a party when some are in the living room, some are in the kitchen, and a few are just hanging out in the hallway. It can get a little chaotic.
Now, Bromine is a bit of a larger atom. It's not the smallest guy on the block, nor is it the biggest diva. It's somewhere in the middle, a solid middle-aged citizen of the periodic table.
This means it has quite a few electron rooms to manage. More rooms mean more potential for things to be… not perfectly neat. And that's where our half-filled orbitals come into play.
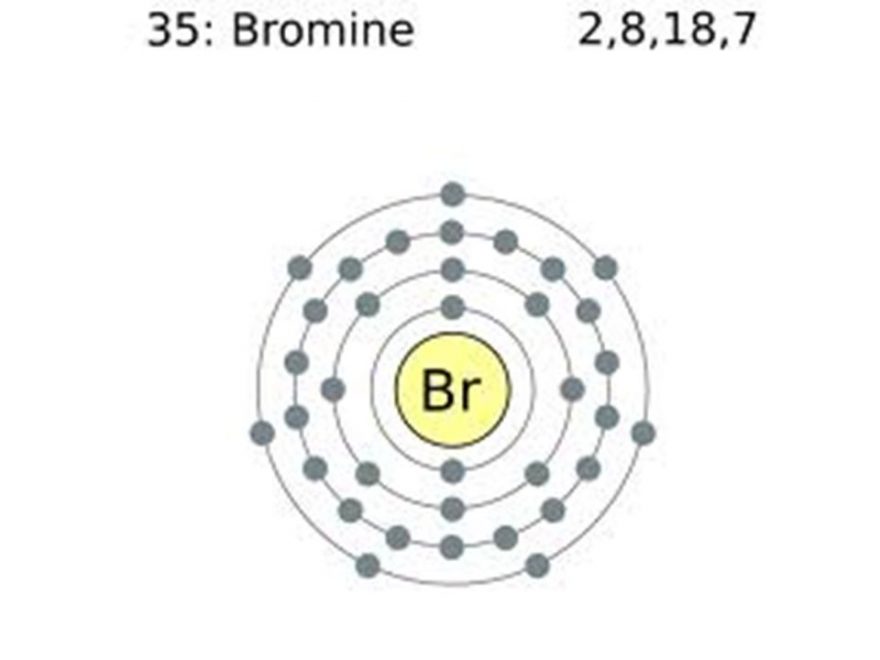
Let's just say for a moment that the orbitals are like little parking spots for electrons. Some spots are full, with two happy electrons parked side-by-side. Others have just one electron, cruising solo.
Those solo cruisers are the ones we're interested in. They're in their spots, but there's definitely room for another. They're the ones that make the atom feel a little bit… incomplete, perhaps?
And when we look at Bromine, with its unique electron personality, we discover something rather interesting. It's like finding a puzzle piece that doesn't quite fit anywhere perfectly.
So, the big reveal, the dramatic moment you've all been waiting for (or at least mildly curious about): How many half-filled orbitals does Bromine have?
Drumroll, please! (Imagine a drumroll made of tiny bouncing electrons).
Bromine, in its natural, unadulterated atomic form, boasts a grand total of one half-filled orbital.
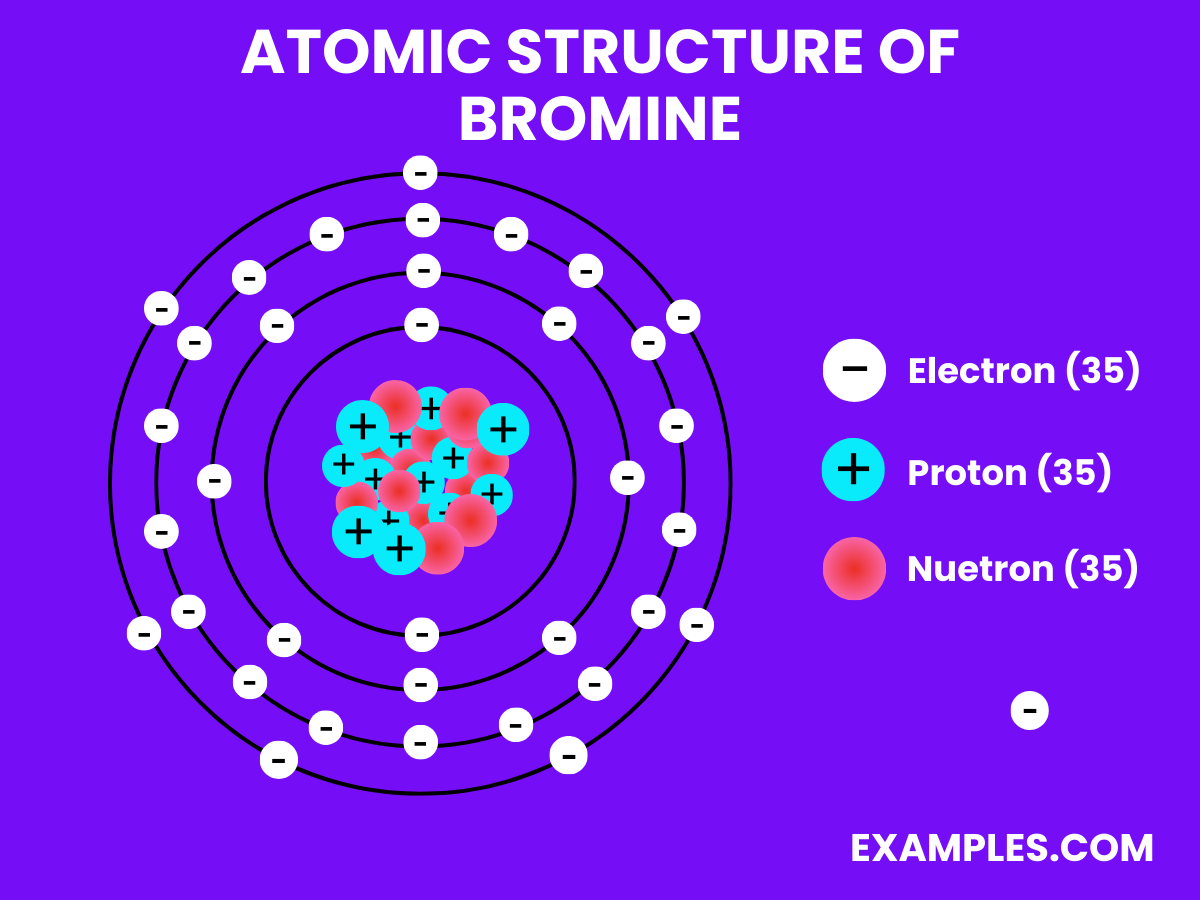
Yes, just one. One lonely little spot with a single electron, just waiting. It's like that one chair left at a table when everyone else is paired up.
It's not a lot, is it? It's not like Bromine is throwing a party and half the rooms are just half-occupied. It's more of a subtle, understated incompleteness.
This single half-filled orbital is actually a pretty big deal for Bromine. It's the key to its chemical personality. It's what makes it, well, Bromine.
Think of it as Bromine's signature move. That one special spot that allows it to interact with other atoms and form all sorts of interesting compounds.
It's like having one really good joke that you always tell. It might not be the most varied performance, but it's effective. And people remember it.
And here’s my unpopular opinion: I think it’s kind of cool. In a world of atoms that might be completely full or trying to fill all their spots, Bromine’s single half-filled orbital is a little bit of quiet rebellion.
It’s not trying to be the most complicated. It’s just doing its thing. And that one half-filled orbital? It’s like its unique little quirk.

It reminds me of that friend who always orders the same thing at a restaurant, or the one who has a signature dance move they pull out at every party. It’s predictable, yes, but it's also part of their charm.
So, next time you hear about Bromine, don't just think of it as a chemical element. Think of it as an atom with a story. An atom with a slightly quirky apartment.
It’s an atom that has one special room that’s not quite full, and that’s perfectly okay. In fact, it’s what makes it so interesting to the rest of the atomic neighborhood.
It’s this single, solitary half-filled orbital that allows Bromine to be so reactive. It's eager to share that one extra electron or to find a friend to fill that space.
It’s like Bromine is always saying, "Hey, I've got this one spot open! Anyone interested?" And a lot of other atoms are very interested indeed.
This simplicity, this focused incompleteness, is really quite elegant. It’s not over-complicated. It’s just… efficient.

And isn’t there something nice about that? In a world that often feels like it’s demanding we fill every single role and every single space, Bromine’s single half-filled orbital is a gentle reminder.
Sometimes, having just one thing that’s a little bit open is exactly what you need to make your mark.
So, there you have it. The not-so-secret secret of Bromine. A single half-filled orbital. It might not be the most exciting number in the universe, but for this particular atom, it’s everything.
It’s the reason it's a halogen, the reason it bonds the way it does. All thanks to that one slightly nudist orbital.
And honestly, who can't relate to having one area of their life that's a little bit less organized than the rest? For Bromine, that area is an orbital.
So, let’s raise a metaphorical glass (filled with electron-rich liquid, perhaps?) to Bromine. To its single, solitary, wonderfully important half-filled orbital. Cheers!
It just goes to show that sometimes, less is more. Or, in this case, one is just right.
