How Many Formula Units Are In 3.6 Grams Of Nacl

Hey there, science adventurers! Ever find yourself staring at a salt shaker and wondering, "How many tiny little salt-bits are actually in this stuff?" Well, today we're diving headfirst into the sparkly world of sodium chloride, or as you and I know it, good old NaCl. We're going to do some super-fun math, the kind that makes you feel like a chemistry wizard, and figure out just how many of these magical formula units are chilling in a measly 3.6 grams of table salt. Buckle up, because this is going to be an exciting ride!
Imagine you've got a tiny little pile of salt, about the size of a sugar cube. Seems small, right? Like, not even enough to season your entire popcorn. But get this: inside that tiny pile, there are more formula units than there are grains of sand on all the beaches in the world. Yes, you read that right! It’s like a microscopic city teeming with these little NaCl dudes, all holding hands and being, well, salty.
We're not talking about individual atoms here, oh no! We're talking about the smallest whole package of sodium chloride, like a tiny, inseparable pair of sodium and chloride buddies. Think of it as the perfect couple in the salt world, always together, always balanced. Each one of these pairs is a formula unit, and they are the fundamental building blocks of our favorite seasoning.
So, how do we actually count these invisible party-goers? Well, scientists, bless their brilliant hearts, came up with this super-cool thing called the mole. Now, a mole isn't like the furry critter that burrows in your garden. In chemistry, a mole is just a really, really, ridiculously huge number. It’s like having a gazillion of something, but way, way more. It’s a number so big, it makes a million look like pocket change. This magic number is called Avogadro's number, and it’s approximately 6.022 x 1023. Just to put that in perspective, if you had a mole of grains of sand, you could cover the entire Earth with a layer about 600 feet thick. Mind. Blown.
Now, the trick is, a mole of something always weighs a specific amount. This specific weight is called the molar mass. For NaCl, this super-special weight is about 58.44 grams. So, if you had a whole 58.44 grams of salt, you'd have exactly 6.022 x 1023 formula units of NaCl. That's like one super-sized bag of salt containing a universe of tiny salt couples!
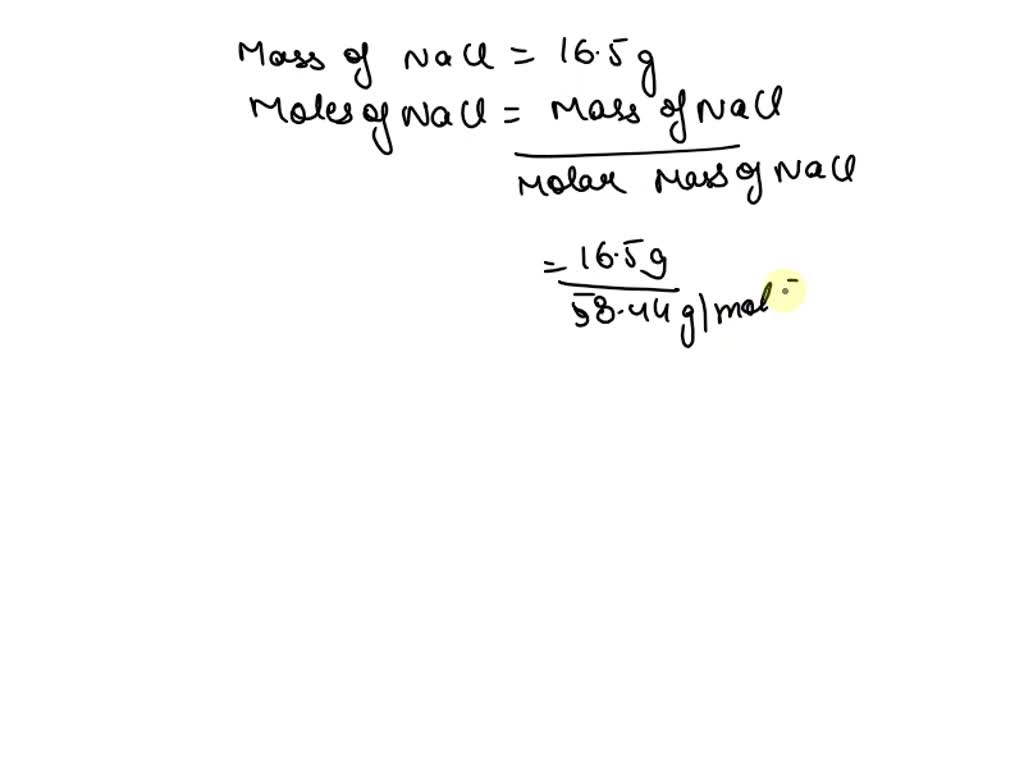
But we're not dealing with a super-sized bag, are we? We've only got a humble 3.6 grams of NaCl. That's like a little sprinkle, a tiny pinch. So, how many of our microscopic salt couples are hiding in that small amount? This is where the fun math kicks in!
First, we need to figure out how many moles we have. Since 58.44 grams is our magic number for one mole, we can find out how many moles are in 3.6 grams by doing a little division. We're essentially asking, "How many times does 58.44 fit into 3.6?" The answer, as you might guess, is not a whole lot. It’s a teeny-tiny fraction of a mole. Let’s do the calculation:
Number of moles = (Mass of NaCl) / (Molar mass of NaCl)
Number of moles = 3.6 grams / 58.44 grams/mole
When you crunch those numbers, you get a very, very small number. It’s something like 0.0616 moles. See? Just a tiny speck of a mole. It's like having just a few crumbs of a giant cake.
Now, remember that incredibly huge number, Avogadro's number (6.022 x 1023)? That's the number of formula units in one whole mole. Since we only have 0.0616 moles, we just need to multiply that tiny fraction by our giant number to find out how many formula units are actually in our 3.6 grams. Drumroll, please!

Number of formula units = (Number of moles) x (Avogadro's number)
Number of formula units = 0.0616 moles x 6.022 x 1023 formula units/mole
And the grand, spectacular, mind-boggling answer is… approximately 3.71 x 1022 formula units!

That’s a number with a 22 after it! It’s 37,100,000,000,000,000,000,000! Yes, you saw that correctly. So, that little pinch of salt on your finger? It's home to over 37 sextillion tiny, inseparable NaCl couples. It’s a microscopic salt metropolis!
So, the next time you’re sprinkling salt on your food, take a moment to appreciate the sheer, unbelievable number of tiny formula units that are making your meal taste oh-so-good. It’s a tiny amount of salt, but it’s packed with an astronomical number of these little chemical wonders. Isn't science just the coolest?
