How Many Enantiomers Are There Of The Molecule Shown Below

Hey there, curious minds! Ever looked at a molecule and wondered, "What's its deal?" Well, today we're diving into a super cool topic: enantiomers. Don't let the fancy word scare you; it's actually pretty neat and has some surprisingly relatable parallels in our everyday lives.
So, what exactly are we talking about? Imagine a molecule. It's got atoms all linked up in a specific 3D arrangement. Now, sometimes, a molecule can exist in two forms that are like mirror images of each other. Think of your left hand and your right hand. They look pretty similar, right? They both have fingers, a thumb, and a palm. But try to put your left glove on your right hand, or vice-versa. They don't quite fit perfectly, do they? That's because they are chiral, and these mirror-image forms are called enantiomers.
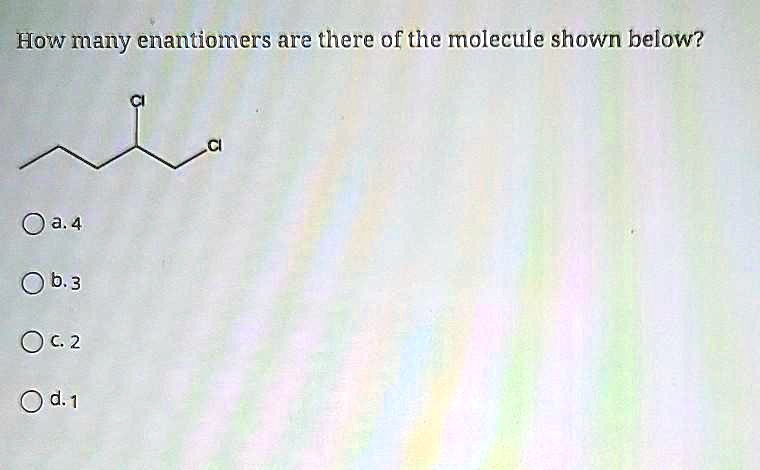
The molecule we're going to explore today is this one:
CH3
|
CH3-C-OH
|
CH2
|
CH3
Pretty straightforward, right? We've got a central carbon atom. This carbon is holding onto four different "things": a methyl group (CH3), another methyl group (CH3), a hydroxyl group (OH), and an ethyl group (CH2CH3). Wait a minute... did I just say "methyl group" twice? Let's take a closer look at that central carbon.
Let's Play "Spot the Difference" (or Not!)
Okay, so the central carbon atom is attached to a hydroxyl group (-OH), an ethyl group (-CH2CH3), and two methyl groups (-CH3). Here's the crucial bit: for a molecule to have enantiomers, it usually needs a chiral center. A chiral center is typically a carbon atom that's bonded to four different groups.
Let's re-examine our molecule. We have a carbon atom in the middle. What's it connected to? Well, it's connected to:

- A hydroxyl group (-OH)
- An ethyl group (-CH2CH3)
- A methyl group (-CH3)
- ...and another methyl group (-CH3)
Uh oh. See the problem? Two of the groups attached to the central carbon are identical: they are both methyl groups.
This is like having a pair of identical twins. They might look incredibly similar, but they are still the same person, just in duplicate. They aren't mirror images in the way that your left and right hands are fundamentally different in their spatial arrangement.
Because our central carbon atom is bonded to two identical methyl groups, it's not a chiral center. It's what we call an achiral center. And if a molecule doesn't have a chiral center, what does that mean for its enantiomers?

The Big Reveal: How Many Enantiomers?
If there's no chiral center, there's no possibility of creating those non-superimposable mirror images. It's like trying to find a left-handed version of a perfectly symmetrical object, like a sphere. You can rotate it all you want, but it will always look the same. It doesn't have that inherent "handedness."
So, for the molecule we're looking at, with its two identical methyl groups attached to the central carbon, there are no enantiomers. This molecule exists as just one single form.
It's kind of a relief, in a way, isn't it? No need to worry about which "hand" of the molecule is doing what. It's just... itself. Simple and straightforward.
Why Does This Even Matter?
Now, you might be thinking, "Okay, so it has no enantiomers. So what?" Well, the concept of enantiomers is actually super important, especially in fields like chemistry and biology. Think about medicines, for example.
Sometimes, one enantiomer of a drug might have a helpful effect, while its mirror image might be inactive or, even worse, have harmful side effects. A famous (and tragic) example is the drug thalidomide. One enantiomer was effective in treating morning sickness, but the other caused severe birth defects. This is a stark reminder of how crucial the 3D structure of molecules, and the existence of enantiomers, can be.
It's like having two different keys. One key opens your front door perfectly, but its mirror image might be just close enough to feel like it might work, but it won't actually turn the lock. Our bodies are incredibly sensitive to these subtle differences in molecular shape.
Even in nature, things are often chiral. Amino acids, the building blocks of proteins, are typically chiral. The DNA double helix itself has a specific handedness. It’s these subtle differences in molecular architecture that contribute to the complexity and wonder of life.
So, while our particular molecule today is a bit of a party pooper when it comes to enantiomers (it’s just one!), understanding why it doesn't have them is a fundamental step in appreciating the fascinating world of molecular chirality. It's all about those four different attachments around a central atom!
Keep exploring, keep asking questions, and remember, even the simplest molecules have stories to tell!
