How Many Electrons Does Carbon Lack In Its Outer Shell

Hey there! Ever wondered about the tiny, invisible building blocks of everything? Yep, we're talking atoms. And today, we're zooming in on a real superstar: carbon! This little guy is everywhere. Your coffee? Carbon. That comfy chair? Carbon. Even you! Totally carbon-based life forms, baby!
But what makes carbon so special? It's all about its outer shell. Think of atoms like tiny solar systems. They have a nucleus in the center, and then electrons whizzing around in different "shells" or orbits. The outermost shell is the VIP section. It determines how an atom plays with others.
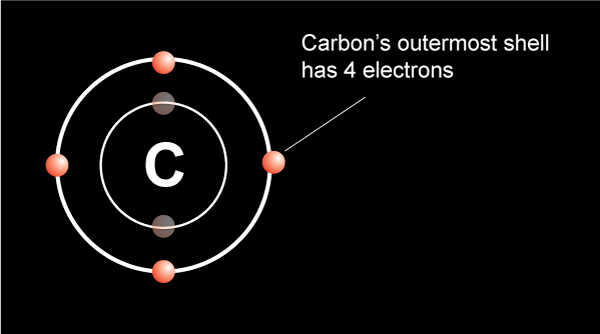
So, how many electrons does carbon want in its outer shell? This is where the fun starts. Carbon is a bit of a show-off. It’s got a nucleus with 6 protons, giving it an atomic number of 6. But in its natural, chill state, it has 6 electrons too. Two hang out in the inner shell, like the quiet kids in the back row. The other four are out in the outermost shell, the party zone!
Now, here's the kicker. Most atoms are happiest when their outer shell is completely full. It's like they've achieved peak atomic zen. For carbon, that sweet spot is having eight electrons in its outer shell. Eight is the magic number. It’s the atomic equivalent of a perfectly balanced meal or finding matching socks.
But carbon? It’s only got four electrons out there. That leaves a little… gap. A bit of an emptiness. How many electrons does carbon lack to reach that blissful state of eight? You guessed it: four more! It’s missing four electrons. Four is its number. Four is what it craves. Four is the key to its atomic happiness.
Think of it like this. Imagine carbon is at a party. It’s got four cool friends already on the dance floor (its electrons). But it knows the real fun, the ultimate dance-off, happens when there are eight people grooving. So, carbon is always looking for friends to join the party. It needs four more dancers to feel complete!

And this is why carbon is so incredibly versatile. Because it’s constantly looking for those four extra electrons, it’s super good at making friends. It can share its electrons. It can borrow electrons. It can even give and take electrons in a bit of an atomic tug-of-war. This willingness to bond is what makes carbon the backbone of organic chemistry.
Without carbon’s little deficiency, life as we know it wouldn’t exist. No complex molecules. No DNA. No pizza. (Tragic, I know). That humble, four-electron gap is the secret ingredient to the universe’s most intricate and amazing creations.
Let’s get a little quirky. Did you know carbon can form rings? Like, a bunch of carbon atoms linking hands in a circle. These are called cyclic compounds. It's like a carbon mosh pit! And some of these rings are incredibly stable, others are super reactive. It's like a carbon personality spectrum.

And then there are these things called fullerenes. Imagine a soccer ball made of carbon atoms. That's a fullerene, or a "buckyball." How cool is that? A tiny, perfect geodesic dome built by carbon’s desire to connect. It’s nature showing off its engineering skills, all thanks to carbon needing four more electrons.
And don't even get me started on diamonds. Pure carbon! Each carbon atom is bonded to four other carbon atoms in a super-strong, rigid structure. That’s why diamonds are so hard and sparkly. It’s the ultimate expression of carbon’s bonding power, all driven by that fundamental need for eight.
But carbon doesn't just form hard, shiny things. It also makes graphite. You know, the stuff in your pencil lead? That's also pure carbon! But here, the carbon atoms are arranged in flat sheets. These sheets can slide past each other easily, which is why your pencil leaves a mark. So, the same element, carbon, can be the hardest substance known or the stuff that writes your grocery list. Talk about duality!
It's all about those four missing electrons. Carbon's not picky about how it gets them. It can form single bonds, double bonds, even triple bonds with other atoms. This flexibility is mind-boggling. It allows carbon to link up with itself in endless chains, branches, and rings, creating an almost infinite variety of molecules.
Imagine a chef who can use only four basic ingredients but can create thousands of different dishes. That's carbon! Its basic "ingredients" are its four outer electrons, and its "recipes" are the bonds it forms with other atoms. The result? The incredible diversity of organic matter.
So, next time you look at a plant, a piece of wood, or even your own hand, remember the humble carbon atom. It’s out there, diligently seeking those four missing electrons. It’s reaching out, connecting, building, and shaping the world around us. It's a constant quest for balance, a fundamental drive for completeness.

It’s like a perpetual atomic dance of giving and receiving. Carbon offers its four electrons, looking for partners who can offer them more. And in return, it gains stability, and we gain a universe teeming with life and wonder. It’s a beautiful, fundamental truth of chemistry, all stemming from a simple question: How many electrons does carbon lack in its outer shell? The answer, my friends, is four. And that’s a pretty big deal.
Isn't it wild that something so small, so seemingly simple, has such a profound impact? The universe is full of these little wonders. The fact that carbon's quest for electron fulfillment is the very foundation of life is, frankly, awesome. It's a testament to how elegant and interconnected everything is.
So, there you have it. Carbon, the four-electron seeker. The ultimate builder. The star of the atomic show. And all because it’s missing just a few little things in its outer shell. Keep wondering, keep exploring, and never underestimate the power of a missing electron!
