How Many Electrons Does Barium Need To Be Stable
You know, sometimes I look at the periodic table and just think, "Wow, these elements are so dramatic." They're always trying to reach this perfect state of stability. It's like they're constantly vying for that ultimate peace and quiet.
And then there's Barium. Oh, Barium. This element, this… personality. It’s got a whole story to tell, and it’s all about fitting in.
My unpopular opinion? Barium doesn't need a whole lot. It just wants to be just right. Not too much, not too little. You know, like Goldilocks, but with electrons.
So, how many electrons does our friend Barium need to achieve this blissful state of electron zen? Let’s break it down, no fancy science jargon, promise.
Imagine electrons as little friends, right? Atoms are always trying to have the perfect number of these friends to feel complete and secure. It's like having a full friend group where everyone feels included.
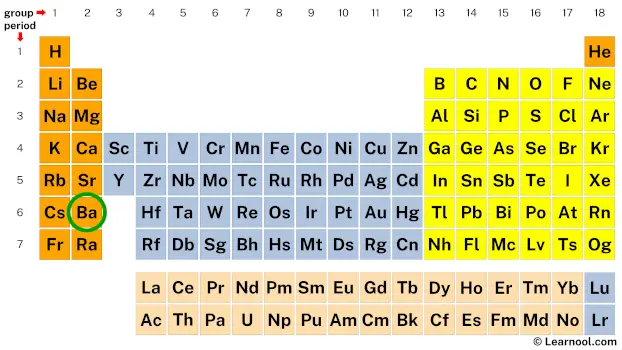
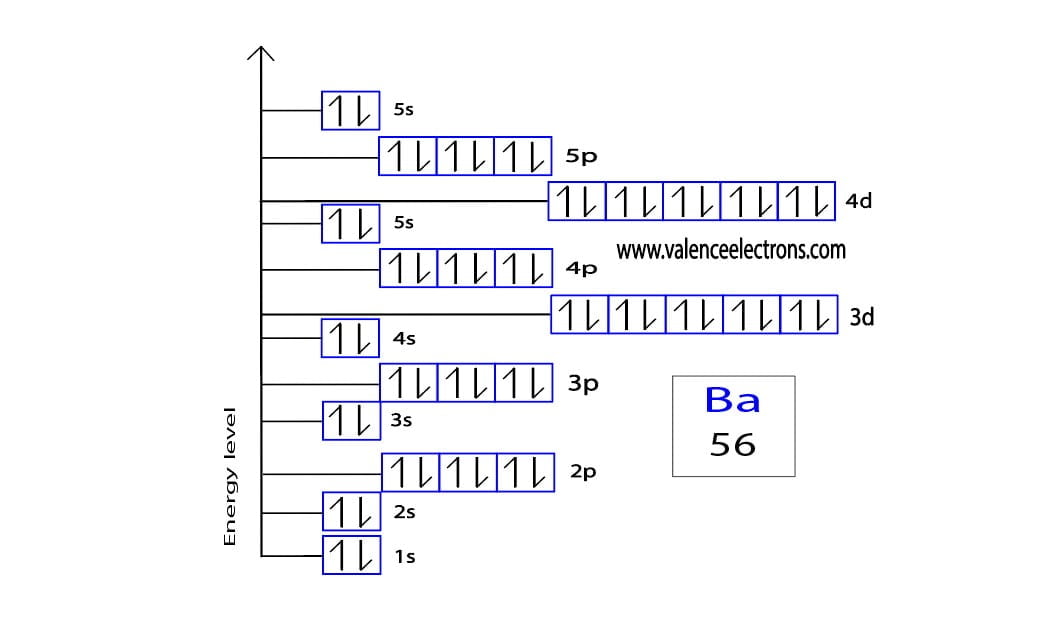
Barium, bless its metallic heart, is element number 56 on the periodic table. That number tells us something important. It's like its unique ID number in the grand scheme of things.
This number, 56, is the total number of electrons Barium has when it's just chilling, minding its own business. Think of it as its starting wardrobe, its full ensemble.
But here's the kicker: most atoms, including Barium, don't like being the odd one out. They want to have a specific, very neat arrangement of these electron friends. It's all about filling up outer shells, you see.
These outer shells are like the popular cliques in high school. Everyone wants to be in the most desirable clique. For atoms, this means having a full outer shell.

The magic number for a full outer shell, the ultimate goal for many elements, is usually 8. It’s like the perfect number of people to play a really fun board game. Not too crowded, not too few.
Now, Barium, with its 56 electrons, is a bit of a… giver. It’s got these extra electron friends hanging around. And honestly, they’re a bit of a burden. They make Barium feel a little unstable, a little… uncool.
It's like having too many toys and not enough shelf space. You just want to get rid of the excess to make things neat and tidy. Barium feels the same way about its extra electrons.
So, what does Barium do? It’s got a couple of options, but for it, the easiest path to electron nirvana is to shed some weight. Electron weight, that is.
And how many electrons does it need to shed to get to that coveted full outer shell, that feeling of “just right”? It needs to get rid of two. Just two little guys.
It’s like saying, "You know what? These two aren't really contributing to the vibe. Let's give them away and feel so much better."
By losing these two electrons, Barium suddenly finds itself with a beautifully complete outer shell. It’s achieved electron harmony. It’s singing with joy, probably.
Think of it this way: Barium has 56 electrons. It wants to get down to a state where its outermost shell is full. And that full state is much closer if it just lets go of a couple.
It’s not about adding a lot more. It’s about simplifying. It’s about decluttering its electron life. And for Barium, that means saying goodbye to two. Simple, right?
It’s like that one friend who’s always trying to organize the group chat. "Okay, everyone, let's streamline this. Who's really necessary for this conversation?" Barium is that friend, but with electrons.
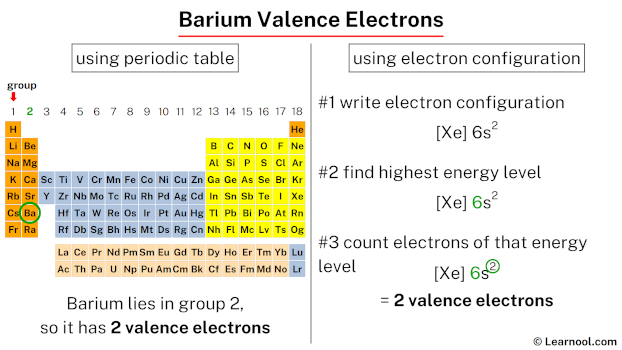
The resulting state for Barium is an ion. It’s no longer neutral Barium. It’s Barium with a positive charge. It’s like it’s earned a badge of honor for its electron generosity.
This ion is called Barium 2-plus, or Ba2+ if you’re feeling fancy. This little symbol tells the world that Barium has positively decided to part with two electrons.
And in doing so, it becomes incredibly stable. It’s found its happy place. It’s like finally getting a good night’s sleep after a long, stressful day.
So, the answer is straightforward: Barium needs to lose two electrons to become stable. It's not a complex negotiation. It’s not a lengthy debate. It’s a clear-cut decision.
It's this desire for stability that drives so much of the chemical world. Elements are always seeking that perfect electron configuration.
And Barium, with its tendency to give up two, is a prime example of this universal quest. It’s almost admirable, in a way. This willingness to simplify its electron structure for the sake of peace.
It’s like saying, “I don’t need all this baggage. I’m going to be my best self by letting go of what’s holding me back.” That’s Barium for you.
It’s a lesson for us all, really. Sometimes, to achieve stability and happiness, you just need to let go of a couple of things. Two, in Barium’s case.
It makes me chuckle, thinking about it. This huge, metallic element, just wanting to get rid of a couple of tiny electrons to feel complete. It’s so wonderfully relatable.
Who knew that the key to atomic serenity could be as simple as shedding a small number of electrons? It’s a stark contrast to our human complexities, isn’t it?
We grapple with so much, and Barium is just over there, figuring out its electron situation with a two-electron giveaway. It’s almost… enviable.
So, next time you see Barium on the periodic table, give it a nod. It’s a bit of a minimalist when it comes to its electron shells. It knows what it wants, and it goes for it.
And that’s the simple, entertaining truth. Barium needs to lose two electrons to be stable. No more, no less. Just two. It’s the most popular number for Barium's stability party, I’d wager.
It’s a small number, but for Barium, it makes all the difference. It’s the difference between electron chaos and electron calm. The difference between feeling a bit scattered and feeling perfectly content.
And that, my friends, is the magical, slightly humorous tale of Barium and its quest for electron stability. It’s a story of simplicity, of letting go, and of finding that perfect, stable state. Just two electrons at a time.
