How Many Electrons Are Unpaired In The Orbitals Of Nitrogen
Hey there, science curious folks! Ever wonder about the tiny, zippy things that make up everything around us? We're talking about atoms, the building blocks of our universe. And inside these atoms are even smaller guys called electrons. Today, we're going to have a little chat, a super chill exploration, about one particular atom: nitrogen. You know, the stuff that makes up a big chunk of the air we breathe! We're going to peek inside its atomic house and see how many of its electrons are feeling a bit… independent. Yeah, we're talking about unpaired electrons!
Now, before you start picturing tiny electrons with little backpacks, heading off on solo adventures, let's clarify. Electrons are always on the move, buzzing around the atom's nucleus like tiny hummingbirds around a bright flower. They don't exactly "pair up" like socks in a drawer, but they do have preferred living spaces within the atom, called orbitals. Think of these orbitals as cozy little rooms within the atomic apartment complex.
Each of these "rooms" or orbitals can hold a maximum of two electrons. And here's the fun part: electrons are a bit like shy people at a party. If there's only one electron in a room, it's a bit lonely, or in science-speak, it's unpaired. If two electrons are in the same orbital, they're considered paired. They're like dance partners, spinning around together!
So, why should you, a perfectly normal human being, care about unpaired electrons in nitrogen? Well, it's actually pretty neat! These unpaired electrons are like the wild cards of the atomic world. They're the ones that are most likely to get involved in chemical reactions, to form bonds with other atoms, and to create all the amazing molecules that make up our world – from the water you drink to the food you eat, to the very air you're breathing right now.
Imagine you're at a potluck. You've got your guests (electrons) and your tables (orbitals). Some tables might have two guests happily chatting away (paired electrons). But then there are those tables with just one guest, looking around, maybe a little bored, definitely open to striking up a conversation with someone new (unpaired electrons!). These solo guests are the ones who are most likely to grab a plate of that delicious casserole you brought or to introduce themselves to the person at the next table. They're the ones driving the social interaction, the ones making the party happen!

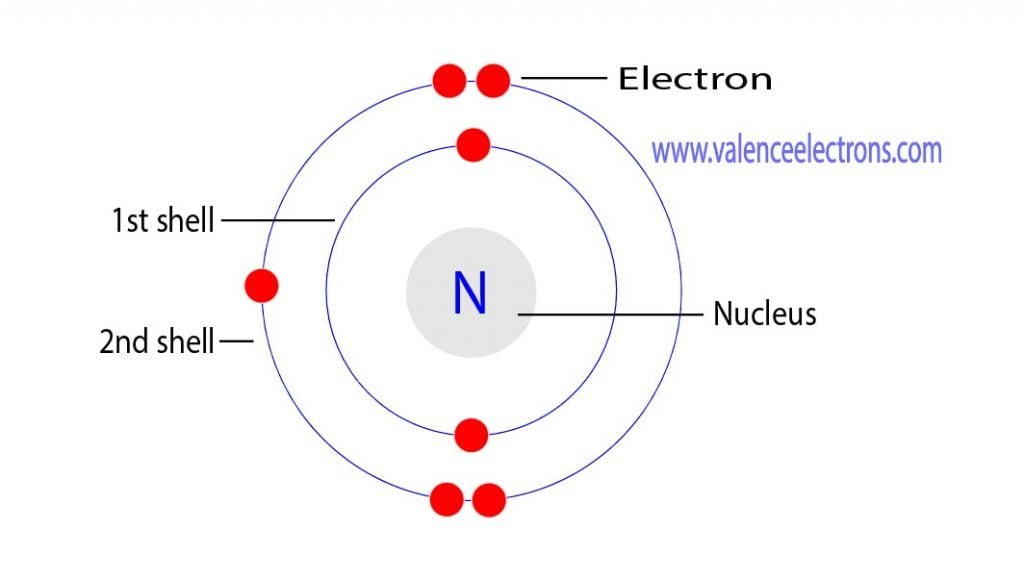
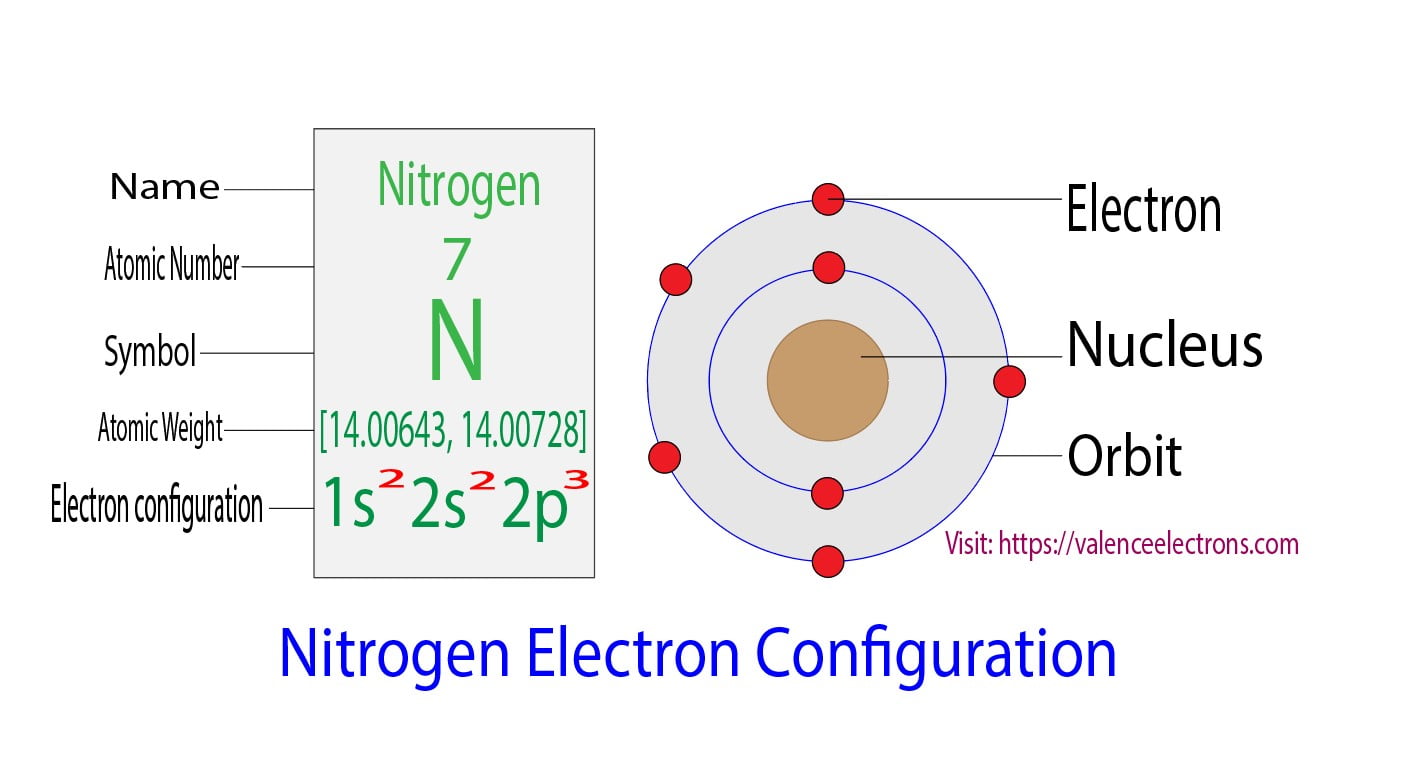
Let's get a little more specific about nitrogen. A nitrogen atom has a total of seven electrons. These electrons don't just float around randomly; they fill up these orbital rooms in a specific order, kind of like how you might fill up your house room by room, starting with the most accessible ones.
For nitrogen, the first two electrons are super tidy and go into the lowest energy orbital, called the 1s orbital. Think of this as the tiny starter studio apartment in the building – it's small but it's the first one available. These two electrons are happy campers, perfectly paired up. They're like a couple who have their own little bubble.

Next, we move up to the next set of orbital rooms. These are called the 2s orbital. Again, the next two electrons slide in here and feel quite comfortable, making themselves a paired duo. So, we've used up four electrons, and they're all happily paired.
Now, here comes the exciting part! Nitrogen has three more electrons to find homes for. These electrons go into a set of orbitals called the 2p orbitals. Now, the 2p orbitals are a bit special. Instead of just one room, there are actually three separate rooms within the 2p level. Think of them as three similar-sized bedrooms on the same floor.
And here's the rule that makes things interesting: electrons, when they have a choice of rooms at the same energy level, prefer to have their own room first! It's like kids at a hotel, each wanting their own bed before they have to share. So, the next three electrons each hop into one of the three 2p orbitals. They're all by themselves, enjoying their own space.

This means that each of these three 2p orbitals has just one electron. And when an electron is all by itself in an orbital, what do we call it? You guessed it: an unpaired electron!
So, when we count them all up for a nitrogen atom: we have two paired electrons in the 1s orbital, two paired electrons in the 2s orbital, and then three unpaired electrons, one in each of the 2p orbitals.
The big takeaway? A nitrogen atom has three unpaired electrons! These are the little electron rebels, the ones ready for action. They are the reason nitrogen is so good at forming chemical bonds. They're like the eager employees at a company, looking for projects to join. They're the reason nitrogen can form things like ammonia (NH₃), which is super important in fertilizers that help grow our food, or the nitrogen gas (N₂) that makes up so much of our atmosphere but is pretty unreactive on its own. It's the other nitrogen compounds, the ones with these handy unpaired electrons involved, that are the real workhorses.
Think about it this way: if all the electrons in nitrogen were paired, it would be a bit of a loner atom, not very interested in mixing and mingling. But because of those three unpaired electrons, nitrogen becomes a fantastic participant in the grand cosmic dance of chemistry. It's like having a bunch of single friends at a party; they're the ones most likely to strike up conversations, start dancing, and make new connections!
So, the next time you take a deep breath of air, or eat some bread, or even just look at the vast sky, remember the humble nitrogen atom and its three little, enthusiastic, unpaired electrons. They're out there, quietly making the world work, one chemical bond at a time. Pretty cool, right? It’s a little glimpse into the intricate, energetic world of atoms, and it all starts with understanding how electrons choose to arrange themselves in their orbital homes.
