How Many Electrons Are Necessary To Produce A Charge Of
Ever stopped mid-awe at a lightning strike, or perhaps just pondered why your socks cling together after a tumble dry? It’s all about the fascinating world of tiny, invisible particles called electrons! And today, we’re going to have some fun diving into a question that might sound a tad technical, but trust me, it’s got some surprisingly cool implications: How many electrons are necessary to produce a charge?
Now, before you start picturing complex equations and lab coats (though those can be fun too!), let’s keep it light. Think of it this way: you know how a single brick can be part of a giant wall? Well, electrons are like those fundamental bricks of electricity. They are the tiny building blocks that make up what we call an electric charge.
So, to get a measurable charge, do you need a million? A billion? A gazillion? The answer is… just one! Well, sort of. You see, it’s not really about having electrons, but about the imbalance of them. It’s like having a seesaw – when it’s perfectly balanced, nothing much happens. But tip one side up, and you’ve got yourself some action!
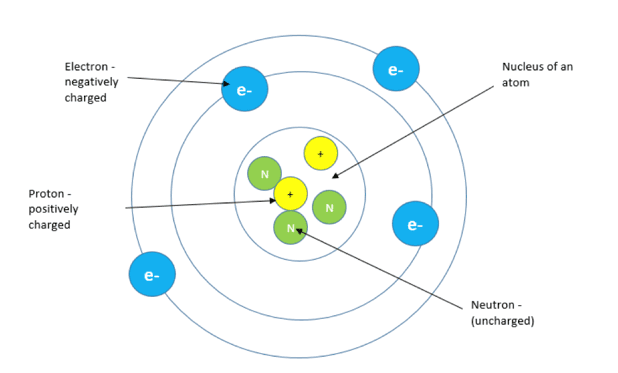
Let’s break it down. Everything around us, from your trusty smartphone to that fluffy cloud outside, is made of atoms. And atoms, bless their little hearts, usually have a nice, tidy balance of positive and negative charges. The positive bits live in the nucleus (think of it as the atom’s cozy home), and the negative bits, our star players, the electrons, zip around the outside.
When an atom has an equal number of positive and negative charges, it’s electrically neutral. No charge, no fuss. But, and this is where the magic happens, if an atom *gains an electron, it becomes negatively charged. It’s like getting an extra friend at a party – suddenly the vibe shifts! Conversely, if an atom *loses an electron, it’s left with more positive bits than negative, making it positively charged. It’s like a friend leaving the party – the overall energy changes.

So, the fundamental unit of charge, the teeny-tiny smallest amount of electrical charge you can have, is the charge of a single electron. Isn’t that neat? A single, almost impossibly small particle holds the key to all the electrical phenomena we experience!
Now, let’s get a bit more specific. The charge of a single electron is a fixed, incredibly small number. It’s approximately -1.602 x 10-19 coulombs. Woah, right? That exponent (-19) tells you just how minuscule that charge is. If you wrote it out, it would be a decimal point followed by 18 zeros and then 1602! You’d need a staggering number of electrons to create a charge that’s easily noticeable in our everyday lives.
So, how many exactly?
To produce a charge of one coulomb – the standard unit of electric charge – you’d need approximately 6.24 x 1018 electrons! That’s 6.24 followed by 18 zeros! Imagine trying to count that many grains of sand. It’s a number so astronomically large it’s almost impossible to truly grasp. It’s like trying to count all the stars in the universe – and then some!
This is why we don’t typically talk about the charge of a single electron in our daily electrical conversations. When we talk about powering our devices, we're dealing with a massive flow of these tiny charge carriers. Think about the electricity that flows through the wires in your home. That’s not just a few electrons; it’s a superhighway of them, zipping along at incredible speeds.
The concept of charge and how it’s produced by electrons is what makes so many everyday things possible and frankly, quite fun! Take static electricity, for instance. When you walk across a carpet in your socks, you’re essentially rubbing electrons from the carpet onto your socks. Your socks then have an excess of electrons, making them negatively charged. And when you get close to something else, like your metal doorknob, those extra electrons want to jump to a place where they are less crowded – hence the little zap!
It’s this tiny imbalance, this slight shift in the electron population, that can lead to such noticeable effects. It’s a reminder that even the smallest things can have a big impact. And isn't that an inspiring thought?
This principle is also at the heart of how batteries work. They are essentially cleverly designed chemical systems that encourage electrons to move from one place to another, creating a flow of charge that we can harness to power our gadgets. Every time you plug in your phone, you're relying on the controlled movement of countless electrons.
Even something as simple as a light bulb glowing is a testament to the power of electrons. When electricity flows through the filament, the electrons collide with the atoms in the filament, making them hotter and hotter until they glow, producing light. It’s a beautiful dance of energy transfer, all thanks to these fundamental particles.

Understanding that it takes an unfathomable number of electrons to make a readily usable charge can actually be quite empowering. It highlights the incredible efficiency and power of the natural world. It makes you appreciate the intricate workings of the universe at its most fundamental level.
So, the next time you marvel at a technological wonder, or even just enjoy the comfort of a light switch, remember the unsung heroes: the electrons. Remember that it takes an immense collective effort of these tiny particles to create the electric world we live in. It’s a testament to the power of numbers, and the profound impact that even the smallest components can have when they work together.
Isn’t it amazing to think that the entire foundation of our electrical world rests on such incredibly small, yet fundamentally important, entities? The universe is full of these hidden wonders, just waiting for us to explore and appreciate them. So go ahead, be curious! The more you learn about the science around you, the more you’ll realize how much magic is packed into the mundane. And who knows, maybe this little dive into electron counts will spark a lifelong fascination with the incredible world of physics!
