How Many Diastereomers Are There Of The Molecule Shown Below

Ever looked at a complex molecular structure and wondered, "How many ways can this thing twist and turn while still being, well, this molecule?" If so, you've stumbled into the wonderfully intriguing world of stereoisomers! It's a topic that might sound a bit intimidating at first, like trying to decipher a secret code. But trust me, understanding how molecules can have different 3D arrangements is not only super cool, it's also incredibly relevant in fields like medicine, materials science, and even flavor chemistry. Think about it: sometimes, a tiny change in how atoms are positioned in space can completely alter a molecule's behavior – making one version a life-saving drug and another, well, not so much! Today, we're going to dive into a specific puzzle: figuring out the number of diastereomers for a particular molecule. It’s like solving a 3D puzzle, and we're going to break it down in a way that’s fun and accessible for everyone.
The purpose of this exploration is to demystify a fundamental concept in organic chemistry: the identification and enumeration of stereoisomers, specifically diastereomers. We’ll be focusing on a given molecular structure and applying a straightforward method to determine how many different spatial arrangements, or stereoisomers, it can have. The benefits of grasping this concept are numerous. For students of chemistry, it's a crucial stepping stone to understanding more advanced topics and tackling complex problem-solving. For anyone with a curious mind, it’s a peek into the intricate, three-dimensional world that governs much of our reality, from the subtle scents of flowers to the efficacy of the medicines we take. Knowing how to identify and count these different molecular forms helps chemists design new compounds with specific properties and ensures the quality and safety of existing ones. It’s about recognizing that even seemingly identical building blocks can be assembled in subtly different ways, leading to dramatically different outcomes. So, buckle up, and let's get our molecular puzzle-solving hats on!
Unlocking the Secrets of Diastereomers
Before we get to our specific molecule, let’s quickly clarify what we’re talking about. Molecules, as you probably know, are made of atoms bonded together. But the way these atoms are arranged in 3D space is what we call stereochemistry. Stereoisomers are molecules that have the same molecular formula and the same connectivity (meaning the same atoms are bonded to each other), but they differ in the 3D arrangement of their atoms. Think of it like having the same Lego bricks but building two slightly different structures.
Within the realm of stereoisomers, there are two main categories: enantiomers and diastereomers. Enantiomers are like mirror images of each other that cannot be superimposed – imagine your left and right hands. They have opposite configurations at all their chiral centers. A chiral center is typically a carbon atom bonded to four different groups. Molecules with chiral centers are like the stars of our stereochemistry show!
Now, diastereomers are stereoisomers that are not mirror images of each other. They are stereoisomers that differ in the configuration at one or more, but not all, of their chiral centers. This is the key difference! If you have two chiral centers, say at positions A and B, enantiomers would have opposite configurations at both A and B (e.g., R at A, S at B vs. S at A, R at B). Diastereomers, on the other hand, might have the same configuration at A but opposite at B (e.g., R at A, S at B vs. R at A, R at B), or vice versa.
The total number of possible stereoisomers for a molecule with n chiral centers is generally 2n. However, this formula can sometimes be tricky due to internal symmetry. But the number of diastereomers is a bit more nuanced. For a molecule with n chiral centers that doesn't have any internal symmetry leading to meso compounds (which are achiral despite having chiral centers), there will be 2n total stereoisomers. Of these, there will be 2n-1 pairs of enantiomers, and the rest will be diastereomers of each other. A simpler way to think about it is to consider the number of chiral centers and systematically draw out the possible configurations.
The Molecule in Question and Our Strategy
Let's look at the molecule we have here:
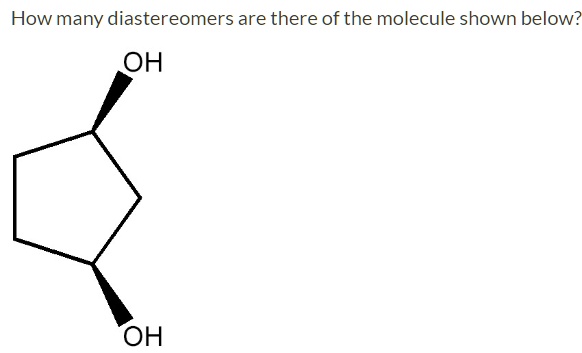
Imagine a molecule with four carbon atoms in a chain. Let's call them C1, C2, C3, and C4 from left to right.
- C1 is bonded to one hydrogen (H) and the rest of the chain.
- C2 is bonded to a methyl group (CH3), a hydrogen (H), a chlorine atom (Cl), and the rest of the chain. This makes C2 a chiral center.
- C3 is bonded to a bromine atom (Br), a hydrogen (H), an ethyl group (CH2CH3), and the rest of the chain. This makes C3 another chiral center.
- C4 is the end of the chain and has its usual bonding (likely to hydrogens to satisfy valency).
So, our molecule is 3-bromo-2-chloropentane.
This molecule has two chiral centers: C2 (bonded to CH3, H, Cl, and the C3-C4 part) and C3 (bonded to Br, H, CH2CH3, and the C1-C2 part). Since we have two chiral centers, let's call them C2 and C3, we can predict the maximum number of stereoisomers.
The general rule for molecules with n chiral centers is that there can be up to 2n stereoisomers. In our case, with n = 2 chiral centers, we can have up to 22 = 4 stereoisomers.
These 4 stereoisomers will consist of two pairs of enantiomers. Each pair will be diastereomers of the other pair.
Let's denote the configuration at C2 as either R or S, and the configuration at C3 as either R or S. We can list all the possible combinations:
- Stereoisomer 1: (2R, 3R)
- Stereoisomer 2: (2S, 3S)
These two are enantiomers because they are mirror images and have opposite configurations at both chiral centers.
- Stereoisomer 3: (2R, 3S)
- Stereoisomer 4: (2S, 3R)
These two are also enantiomers because they are mirror images and have opposite configurations at both chiral centers.
Now, let's consider the relationship between these pairs.
- (2R, 3R) and (2R, 3S) are diastereomers. They have the same configuration at C2 but different configurations at C3.
- (2R, 3R) and (2S, 3R) are also diastereomers. They have different configurations at C2 but the same configuration at C3.
- (2S, 3S) and (2R, 3S) are diastereomers (different at C2, same at C3).
- (2S, 3S) and (2S, 3R) are diastereomers (same at C2, different at C3).
So, within our set of 4 stereoisomers:
- We have one pair of enantiomers: (2R, 3R) and (2S, 3S).
- We have another pair of enantiomers: (2R, 3S) and (2S, 3R).
The members of one enantiomeric pair are diastereomers of the members of the other enantiomeric pair. For example, (2R, 3R) is a diastereomer of (2R, 3S) and (2S, 3R). Similarly, (2S, 3S) is a diastereomer of (2R, 3S) and (2S, 3R).
Therefore, for the molecule 3-bromo-2-chloropentane, there are a total of four stereoisomers. These four stereoisomers are made up of two distinct pairs of enantiomers. The members of these two pairs are, by definition, diastereomers of each other.

So, if the question is "How many diastereomers are there of the molecule shown below?" referring to the set of all possible stereoisomers, then each stereoisomer is a diastereomer of three other stereoisomers. However, usually the question implies distinct structural isomers that are diastereomers. In this context, we have two distinct sets of enantiomers, and the members of one set are diastereomers of the members of the other set.
The number of unique diastereomeric relationships is often what’s implied. If we consider the four stereoisomers, each one has three other stereoisomers that are its diastereomers. But the question is about the number of distinct diastereomers. We have two pairs of enantiomers. The members of one pair are diastereomers to the members of the other pair. Thus, there are four stereoisomers in total, and they exist as two pairs of enantiomers, where each member of one pair is a diastereomer to both members of the other pair.
Let's rephrase to be super clear. We have 4 total stereoisomers.
- (2R, 3R) is a diastereomer of (2R, 3S) and (2S, 3R).
- (2S, 3S) is a diastereomer of (2R, 3S) and (2S, 3R).
- (2R, 3S) is a diastereomer of (2R, 3R) and (2S, 3S).
- (2S, 3R) is a diastereomer of (2R, 3R) and (2S, 3S).
The final answer, in terms of distinct molecules that are diastereomers of one another, is that there are four stereoisomers, and each of these is a diastereomer to two specific other stereoisomers within that set of four.
