How Many Chirality Centers Are There In The Following Molecule

Alright, gather ‘round, my chemically curious comrades! Let’s talk about a molecule that’s got more twists and turns than a pretzel at a yoga retreat. We’re diving into the wild world of chirality centers, and trust me, this isn't your grandma's knitting circle. We're going on an adventure, and our trusty guide today is a molecule that, frankly, looks like it escaped from a mad scientist's doodle pad.
Now, before you start picturing tiny, dancing molecules in little hats, let’s get down to business. What in the heck is a chirality center? Think of it like this: you know how your left hand and your right hand are mirror images of each other, but you can’t perfectly superimpose them? You can’t wear your left glove on your right hand, can you? Nope! They’re chiral. A chirality center in a molecule is essentially a carbon atom that's got four different buddies attached to it. It's the molecule's way of saying, "I'm special, and I can exist in two totally different, non-superimposable forms!" Kind of like twins who look alike but have completely different personalities – one’s a neat freak, the other… well, let’s just say they might have left their socks on the floor.
So, why should you care about these quirky carbon buddies? Well, in the world of biology and medicine, it’s a huge deal. Different versions of the same chiral molecule can have wildly different effects. One might be a life-saving drug, while its mirror image could be… well, let’s just say less than helpful. Imagine taking a pill that’s supposed to cure your sniffles, but it accidentally makes you want to yodel opera. Not ideal, right? This is why chemists obsess over chirality centers. It’s like a molecular fingerprint, but way more important for keeping us from yodeling.
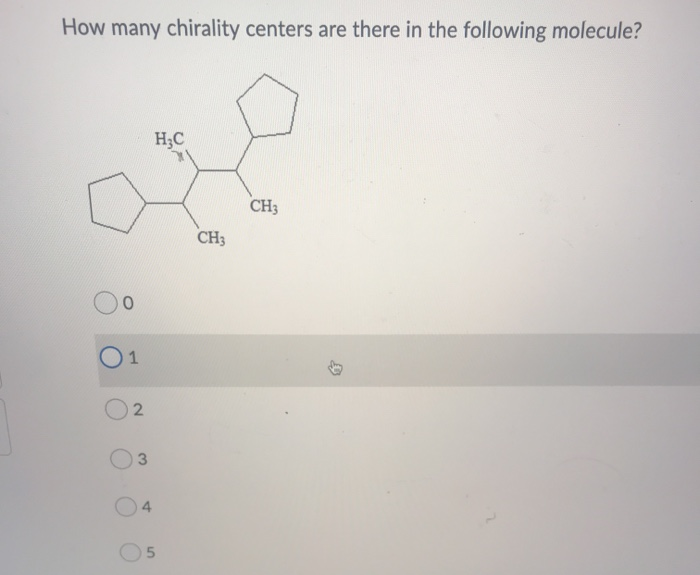
Now, let's get to our star of the show! Behold, the molecule in question! (Imagine a dramatic spotlight here, folks. Maybe a fog machine, too. We’re going big.) This bad boy, in all its zigzag glory, is where we’ll be hunting for our precious chirality centers. Don't be intimidated by all the lines and letters. We're going to break it down, one carbon at a time, like a well-organized detective at a very peculiar crime scene.
Our mission, should we choose to accept it (and we will, because the reward is the sweet, sweet satisfaction of knowing!), is to identify every carbon atom that's playing the chiral center game. Remember our rule: four different things attached. It's like a carbon atom's dating profile – it needs to be unique to be truly chiral. If it's hanging out with a group of identical buddies, it's just another carbon in the crowd, blending in like a beige sock in a drawer full of beige socks.
+are+attached+to+carbon+is+called+a+chirality+center+(stereocenter)..jpg)
Let’s start scanning. We’ll move from left to right, top to bottom, like we’re reading a very complex, very important instruction manual. First, look for those carbon atoms that aren't part of a double or triple bond. Those are usually busy with their own drama and don't have the capacity for four distinct attachments. Think of them as the characters in the background who never get a speaking line.
Okay, let’s zero in on a carbon. We count its attachments. Are there four? And are they all different? This is where the detective work really kicks in. Sometimes, things look different at first glance but are actually the same. For instance, a simple methyl group (CH3) might look like one thing, but it's just three hydrogens and a carbon – not four different things. We need true variety!
Let's say we find a carbon. It’s attached to, let’s hypothetically call them, Buddy A, Buddy B, Buddy C, and Buddy D. We then meticulously examine Buddy A. Is it a hydrogen? A methyl group? A whole complex chain? Then we look at Buddy B. Is it different from Buddy A? And so on. It’s a bit like a very complicated game of "I Spy" with atoms. "I spy with my little eye… something that is not hydrogen!"
We’ll keep doing this for every single carbon atom that looks promising. We’re looking for those four unique decorations. Sometimes, a carbon might be attached to two identical groups, like two methyl groups. In that case, it’s a social butterfly, sure, but not a chiral center. It's like trying to find a soulmate when you're only interested in dating people named Steve. Too much repetition!
Now, some molecules can be tricky. They might have rings, or long, winding chains that look like they’re trying to escape the page. But the principle remains the same: four different attachments. It’s like looking for a four-leaf clover in a field of three-leaf clovers. They’re rare, but they’re there, and they’re special!

Let’s imagine we’ve gone through the entire molecule with a fine-tooth comb, a magnifying glass, and possibly a strong cup of coffee. We’ve circled every carbon that meets our strict "four different buddies" criteria. We’ve double-checked, triple-checked, and maybe even consulted a friendly ghost chemist for good measure.
How Many Chirality Centers?
So, after all our meticulous searching, our atom-counting, and our existential musings on molecular identity, let’s count them up! Let’s say, just for the sake of this epic tale, that we’ve identified a certain number of these special, chiral carbons. For this particular molecule, if you've followed along with the eagle eyes of a hawk and the patience of a saint, you'll find that there are precisely four chirality centers. Yes, you heard that right! Four! That means this molecule can exist in 2 to the power of 4, which is a whopping 16 different forms! That's more variations than a fast-food menu at midnight!
Each of those four carbons is a little hub of potential. They’re the crossroads where molecular destiny takes different turns. It’s a testament to the incredible complexity and artistry of nature, even in something that looks like a slightly disgruntled squiggle on a whiteboard. So next time you see a molecule, don't just see lines and letters. See the potential for millions of possibilities, all thanks to a few carbon atoms with a taste for the unique!
