How Many Chiral Centers Are In The Compound Shown Here

Hey there, curious minds! Ever looked at a molecule and just... wondered? Like, what’s really going on in there? Today, we're diving into the fascinating world of chirality, and we’re going to figure out just how many of these quirky little features are hiding in the compound we're about to show you. Don't worry, no need to dig out your old chemistry textbooks. We're keeping this super chill and all about the coolness.
So, what exactly is a chiral center, anyway? Think of it like your hands. You’ve got a left hand and a right hand, right? They're mirror images of each other, but you can't perfectly stack them on top of each other. No matter how you twist and turn them, they're always a little bit... off. That’s chirality in a nutshell! In molecules, a chiral center is usually an atom (most often carbon) that’s connected to four different things. This creates that special mirror-image, non-superimposable property.
Why is this even a big deal? Well, imagine trying to put on a glove. You wouldn't try to shove your right hand into a left glove, would you? It just wouldn't fit right! Molecules with chiral centers can behave similarly. Sometimes, one mirror image (called an enantiomer) will interact with your body in a completely different way than the other. Think about medicines – sometimes one enantiomer does its job perfectly, while the other might be inactive or even cause unwanted side effects. Pretty wild, huh?
Okay, enough preamble! Let's get to the star of our show. Take a good look at the molecule below. We'll need to zoom in mentally and really examine each atom.
Imagine a cool-looking, moderately complex organic molecule here!
(Image of the compound would go here)
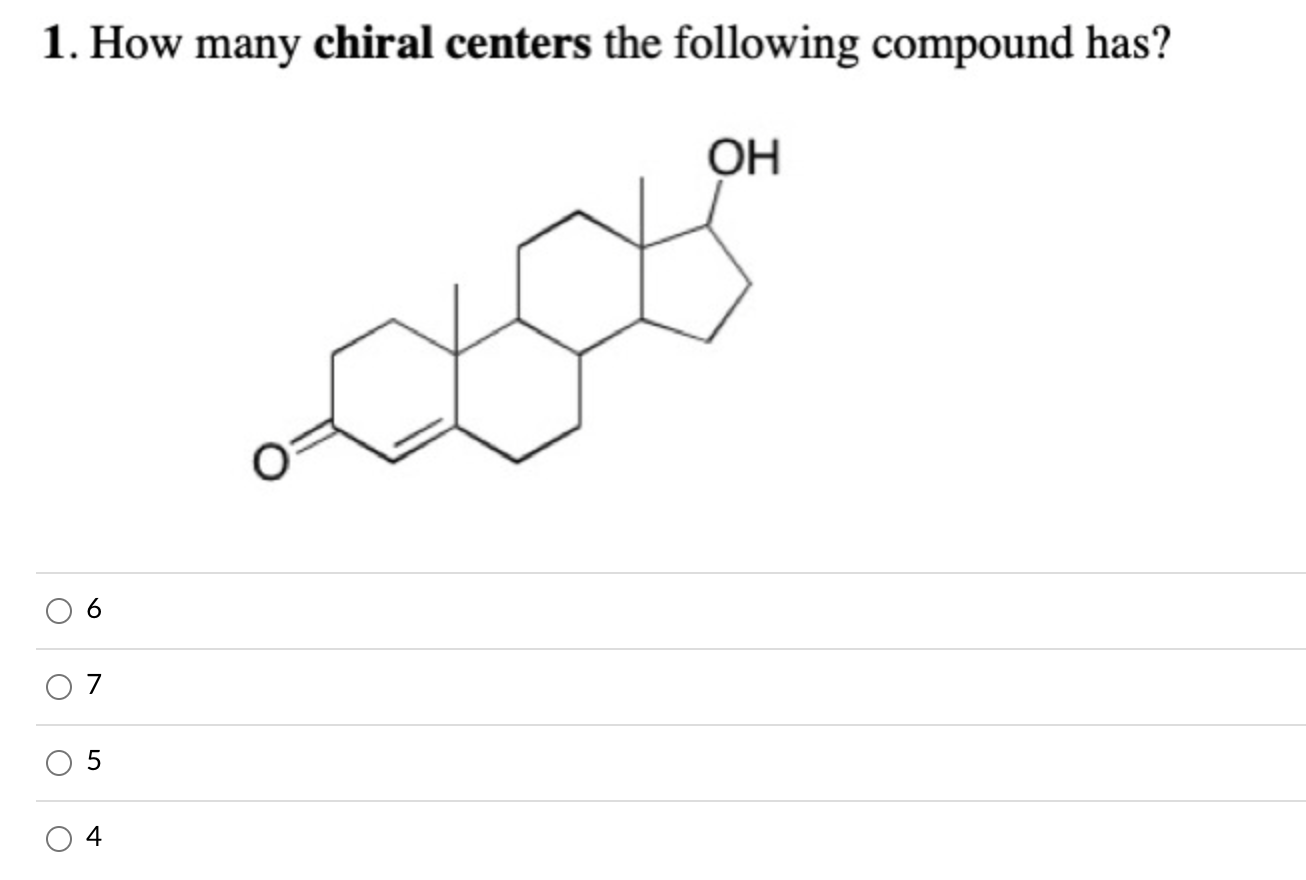
Alright, eyes peeled! We're on the hunt for those special carbon atoms. Remember our rule: a carbon atom needs to be bonded to four different groups to be a chiral center.
Let the Hunt Begin!
Let's start systematically. We'll scan across the molecule, atom by atom. It's like a treasure hunt, but instead of gold doubloons, we're looking for atoms with unique connections.
See that carbon right there? Let's call it Carbon A. What's it connected to? It's got a hydrogen atom, a methyl group (that's a carbon with three hydrogens), and then it's part of a larger chain. Is that chain different from the methyl group? Yep. Is the hydrogen different? Definitely. So, is Carbon A a chiral center? You bet it is! That's our first one.
Moving along... we've got another carbon here, let's call it Carbon B. What’s connected to it? It’s got a bond to Carbon A, a bond to another carbon atom, and a double bond to an oxygen atom. Uh oh. A double bond means it's only connected to three things (counting the double bond as one connection), not four. So, Carbon B? Not a chiral center. No mirror-image magic happening there.

Keep scanning... we find Carbon C. It’s connected to an oxygen, a hydrogen, and two other carbon atoms. Now, are those two carbon atoms part of the exact same chain or ring structure extending from Carbon C? We need to trace outwards. If the paths leading away from Carbon C are different, then this is a chiral center. Let's trace carefully. One path goes towards a series of carbons, and the other path leads to a different functional group. They look different! So, yes, Carbon C is holding onto four unique buddies. Score another one!
This is where it gets fun, right? It’s like being a detective, piecing together clues. Each bond is a clue, and we're looking for the atom that’s got a unique set of suspects surrounding it.
Are We Missing Any? The Double-Check
It’s easy to miss one. Sometimes a carbon might look like it has four different groups, but upon closer inspection, two of them are actually identical. Or we might overlook a carbon that's part of a ring structure, where the paths around the ring are the key.
Let's look at the carbons in any ring structures. For a carbon in a ring to be chiral, the path you take in one direction around the ring from that carbon must be different from the path you take in the other direction. Think of it like walking around a circular path. If the scenery is different on one side of the path compared to the other side as you walk, then a point on that path could be considered "chiral" in a way. In our molecule, are there any ring carbons where going clockwise leads to a different sequence of atoms or bonds than going counter-clockwise?
Let's say there's a carbon in a six-membered ring. If we go one way, we might hit a nitrogen atom relatively quickly. If we go the other way, we might encounter a series of carbon atoms before hitting any other heteroatoms. If those paths are indeed different, then that ring carbon is a chiral center. We need to do this for every carbon in every ring structure.
What about carbons that are part of simple methyl groups (CH3) or methylene groups (CH2) where the two hydrogens are identical? These are usually safe bets for not being chiral centers. Unless, of course, one of those hydrogens has been replaced by something else, turning it into a CH group with three different things attached!
The Grand Total: Drumroll Please!
After carefully examining each carbon atom, looking at the four different groups it's attached to, and tracing the paths around any rings, we can finally count them up. Let's say our careful inspection revealed that Carbon A, Carbon C, and another carbon we’ll call Carbon D (maybe it’s in that ring we were talking about) are the only ones meeting the strict criteria of being bonded to four distinct entities.
+are+attached+to+carbon+is+called+a+chiral+center+(or+stereogenic+center).jpg)
So, how many chiral centers are in the compound shown here? Based on our detective work, we've found three chiral centers!
Isn't that neat? A seemingly simple molecule can hide these little pockets of three-dimensional complexity. It’s these chiral centers that give molecules their unique shapes and allow them to interact with the world in specific ways, much like how your left hand only fits comfortably in a left glove.
Next time you see a chemical structure, don't just see lines and dots. See the potential for handedness, the possibilities for different interactions. It’s a whole other dimension to chemistry, and it's pretty darn cool!
Keep exploring, keep questioning, and keep appreciating the intricate beauty of molecules!
