How Many Chiral Carbons Are Contained In Ribose
Picture this: it’s late, you’ve had way too much coffee, and you’re staring at a textbook that looks suspiciously like it was written in ancient Sumerian. You’re trying to understand these molecules, these building blocks of life, and suddenly you hit a word that throws you for a loop: chiral. What on earth is a chiral carbon, and why should you care? I’ve been there, my friends. Staring blankly, wondering if my brain had just decided to take a spontaneous vacation. But don't worry, we're going to unravel this mystery together, one molecule at a time. Today, we're focusing on a particularly important one: ribose. Ever heard of it? If you’ve ever paid attention in biology class, you probably have.
So, what’s the big deal with ribose anyway? Well, it’s a sugar, a pentose sugar to be precise, meaning it has five carbon atoms. And it’s not just any sugar; it's a fundamental component of RNA (ribonucleic acid) and also plays a crucial role in energy production as part of ATP (adenosine triphosphate). Think of it as a tiny, five-carbon architect that helps build the very blueprint of life and keeps our cells humming with energy. Pretty cool, right?
Now, back to our mystery word: chiral. Imagine you have a glove. You can have a left glove and a right glove, right? They look incredibly similar, almost identical, but they are not the same. You can’t wear a left glove on your right hand (well, you could, but it would be a terrible fit, and let’s be honest, a little awkward). This "handedness" is what we mean when we talk about chirality in molecules. A chiral molecule is one that is non-superimposable on its mirror image. Just like your left and right hands!
And what makes a molecule chiral? Typically, it’s a chiral carbon atom. This is a carbon atom that is bonded to four different atoms or groups of atoms. Think of it as the carbon atom having four unique dance partners. If any two of those partners are the same, the carbon is not chiral, and the molecule might not have that interesting "handedness." This is where things start to get interesting, especially when we look at sugars like ribose.
So, how many of these "dance-happy" carbons does ribose have?
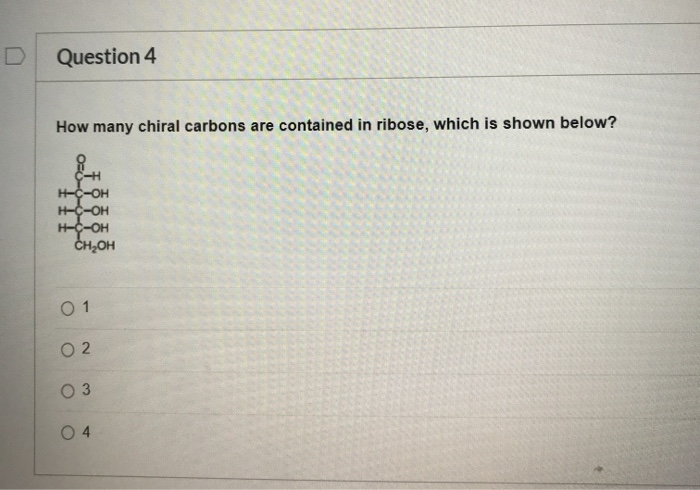
Let’s break down the structure of ribose. As we mentioned, it’s a pentose sugar, meaning it has a backbone of five carbon atoms. We usually number these carbons from 1 to 5, starting from the end that has the aldehyde or ketone group (in ribose’s case, it's an aldehyde). So, we have C1, C2, C3, C4, and C5.
To figure out how many chiral carbons ribose has, we need to examine each carbon atom individually and see if it’s attached to four different things. Let’s go through them, shall we? Prepare for some molecular detective work!

Carbon 1 (C1): The Aldehyde Group
In ribose, the first carbon atom, C1, is part of an aldehyde group. This means it’s double-bonded to an oxygen atom (C=O) and also bonded to a hydrogen atom (C-H). Because it has a double bond, it can only be bonded to three different entities: the double-bonded oxygen, the hydrogen, and the rest of the carbon chain (C2). Therefore, C1 is not a chiral carbon. It’s kind of like a carbon that’s just trying to be friendly with two atoms at once, so it doesn't have the space or the distinct partners to be chiral.
Carbon 2 (C2): The First Potential Chiral Center
Now, let’s look at C2. What is this carbon attached to?
- It's bonded to C1 (the aldehyde end).
- It's bonded to C3 (the next carbon in the chain).
- It’s bonded to a hydrogen atom (-H).
- And it’s bonded to a hydroxyl group (-OH).
So, C2 is bonded to C1, C3, -H, and -OH. Are these four things all different? Yes, they are! C1 and C3 are clearly different chain segments. The hydrogen atom is different from the hydroxyl group. Therefore, C2 is a chiral carbon. Woohoo! Our first chiral center found!
Carbon 3 (C3): The Second Potential Chiral Center
Moving on to C3. What’s hanging out with this carbon?
- It’s bonded to C2.
- It’s bonded to C4.
- It’s bonded to a hydrogen atom (-H).
- And it’s bonded to a hydroxyl group (-OH).
Again, let’s check: C2 and C4 are different parts of the chain. The hydrogen atom is different from the hydroxyl group. So, C3 is also bonded to four distinct groups. This means C3 is also a chiral carbon. We’re on a roll!
Carbon 4 (C4): The Third Potential Chiral Center
Almost there! Let’s examine C4. What’s connected to it?
- It’s bonded to C3.
- It’s bonded to C5 (the carbon at the end of the chain).
- It’s bonded to a hydrogen atom (-H).
- And it’s bonded to a hydroxyl group (-OH).
You guessed it! C3 and C5 are different chain segments, and -H is different from -OH. Thus, C4 is bonded to four unique substituents. Consequently, C4 is a chiral carbon. We've found another one!
Carbon 5 (C5): The End of the Line
Finally, we reach C5. This carbon is at the end of the carbon chain. What’s it attached to?
- It’s bonded to C4.
- It’s bonded to three hydrogen atoms (-H, -H, -H) because it forms a methyl group (-CH3) in the open-chain form.
Here’s the catch: C5 is bonded to C4 and three hydrogen atoms. Since three of the groups attached to C5 are identical (the hydrogen atoms), C5 is not a chiral carbon. It doesn't have the four different dance partners to qualify.
The Grand Total
So, if we tally them up:
- C1: Not chiral
- C2: Chiral
- C3: Chiral
- C4: Chiral
- C5: Not chiral
This means that ribose, in its open-chain form, contains a total of three chiral carbon atoms. Specifically, these are carbons 2, 3, and 4.
Now, you might be thinking, "Okay, three chiral carbons. So what?" Well, this is where the magic (and the complexity) really kicks in. Because ribose has these three chiral carbons, it can exist in different stereoisomeric forms. Remember our glove analogy? For a molecule with multiple chiral centers, you can have even more combinations of "left" and "right" orientations, leading to a variety of molecules that are stereoisomers of each other.

For a molecule with n chiral centers, there can be up to 2n stereoisomers. In the case of ribose, with three chiral centers, that's potentially 23 = 8 stereoisomers! However, not all of these are biologically relevant or stable. The most important form we usually talk about is D-ribose. The "D" refers to the configuration around the highest numbered chiral carbon (which is C4 in this case). It tells us the orientation of the hydroxyl group on that carbon relative to a reference molecule (glyceraldehyde). It’s a specific way of arranging those four different groups around that carbon atom.
And guess what? The other stereoisomers of ribose? Some of them are sugars too, like arabinose and xylose, but they have different arrangements of the hydroxyl groups on those chiral carbons. They are like cousins to ribose, sharing the same chemical formula but having slightly different 3D shapes. Isn't it fascinating how a tiny change in the arrangement of atoms can lead to completely different molecules with different properties and roles in biological systems?
It's also worth noting that ribose often exists in a cyclic form, especially in aqueous solutions. When the aldehyde group at C1 reacts with the hydroxyl group at C5 (or sometimes C4), it forms a five-membered ring (a furanose ring). Even in its cyclic form, the chirality of the original carbons (C2, C3, and C4) is preserved. Additionally, the cyclization process can create a new chiral center at C1 (called the anomeric carbon), but the core chiral centers of the sugar backbone remain. For D-ribose, the three chiral carbons (C2, C3, C4) continue to hold their D-configurations, dictating the molecule's fundamental identity and its interactions within biological pathways.
So, the next time you hear about RNA, ATP, or any biological process involving these vital molecules, take a moment to appreciate the humble ribose and its three chiral carbons. Those seemingly simple arrangements of atoms are the reason why life, as we know it, can exist in its wonderfully diverse and complex forms. It’s a testament to the power of molecular architecture and the subtle nuances of stereochemistry. And who knew that understanding handedness could be so important? It's like discovering that your left and right shoes aren't just variations, but essential for walking comfortably! Chemical structures are like that too; the orientation matters. So, cheers to chiral carbons and the intricate dance of molecules!
