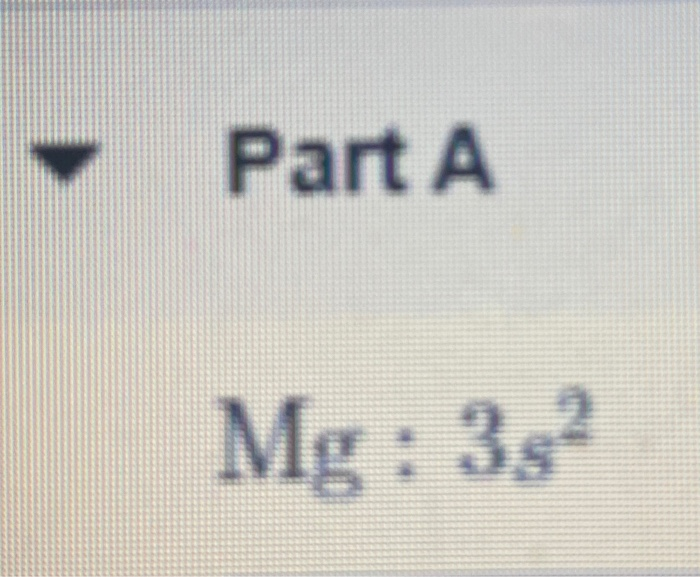
How Many Bonds Can N Make Without Hybridization
Okay, so picture this: it was a particularly dreary Tuesday. I was wrestling with a particularly stubborn chemistry problem, the kind that makes you question all your life choices up to that point. My whiteboard was a chaotic mess of arrows, lone pairs, and molecular structures that looked suspiciously like a toddler’s abstract art. I was trying to figure out the bonding in some weird nitrogen-containing molecule, and for the life of me, I couldn't make the electrons add up in a way that made sense. It was like trying to fit a square peg into a round hole, but with atoms. Frustrating, to say the least.
Then, my cat, Archimedes (yes, that’s his name, don't judge), decided it was the perfect moment to perform a death-defying leap from the bookshelf onto my desk, scattering my notes and nearly taking out my coffee mug. As I was picking up the scattered papers, a little diagram of a simple ammonia molecule caught my eye. NH₃. Nitrogen bonded to three hydrogens. Simple, right? But then my brain, still buzzing from the aforementioned electron-counting struggle, went, “Wait a minute. How does nitrogen do that?”
This seemingly innocent ammonia molecule was the catalyst for a much deeper dive. It got me thinking about the fundamental capabilities of an element like nitrogen. Specifically, how many of its tiny electron friends can it actually invite to a bonding party without needing some fancy, room-clearing dance moves like hybridization? Let’s unpack this, shall we?
The Humble Beginnings: Nitrogen's Electron Wallet
So, let’s start with the star of our show: Nitrogen. Atomic number 7. What does that mean? It means a neutral nitrogen atom has 7 protons and, crucially for bonding, 7 electrons. Now, these electrons aren't just floating around aimlessly; they’re organized in shells, like a cosmic set of Russian dolls. The first shell, the one closest to the nucleus, can hold a maximum of 2 electrons. The second shell, the one that matters most for bonding, can hold up to 8.
Nitrogen’s electron configuration is 1s²2s²2p³. The ‘1s²’ part is all tucked up in the first shell, happy and contained. The real action is in the second shell, the valence shell. Here, we have 2 electrons in the 2s orbital and 3 electrons in the 2p orbitals. So, in total, nitrogen has 5 valence electrons. This is our starting point, our electron budget, if you will.
These 5 valence electrons are key to understanding how nitrogen forms bonds. Remember, atoms are generally a bit greedy. They want to achieve a stable electron configuration, usually by having a full outer shell, which for most elements means 8 valence electrons (the octet rule). It's like they're trying to get to that magical, happy number.
The Lone Pair Powerhouse
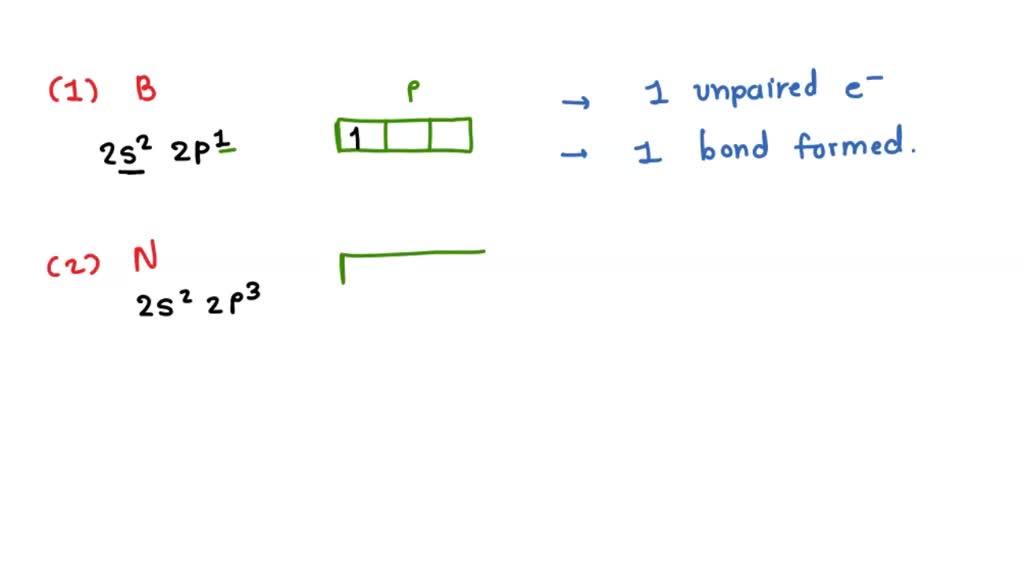
Looking at that 2s²2p³ configuration, you might notice something interesting. The 2s orbital is completely filled with its two electrons. These two electrons are paired up, snug as a bug in a rug. They're not readily available for forming new, covalent bonds. Think of them as a retired couple, happy in their established relationship, not looking for new housemates.
The exciting part for bonding lies in the 2p orbitals. Nitrogen has three 2p orbitals (px, py, pz), and each of them has one electron. These are unpaired electrons. And what do unpaired electrons love to do? They love to find another electron to pair up with, forming a nice, strong covalent bond. So, inherently, nitrogen has three unpaired electrons, ready for action.
This means that, without any fancy electron shuffling, nitrogen is naturally equipped to form three covalent bonds. Each of these unpaired electrons can partner up with an electron from another atom, like hydrogen, to create a shared pair of electrons. This is the basis of what we call the normal or unhybridized bonding capacity of nitrogen.
The Ammonia Example: A Classic Case
Let’s go back to our friend, ammonia (NH₃). Nitrogen has its 5 valence electrons. Each hydrogen atom has 1 valence electron. To form NH₃, nitrogen needs to form three bonds. It uses its three unpaired electrons, one for each N-H bond. Each hydrogen contributes its single electron to form a shared pair. So, nitrogen ends up with 3 shared pairs of electrons (which count as 3 bonds) and its original lone pair from the 2s orbital. This gives it a total of 8 electrons around it – a beautiful, stable octet!
See? Three bonds, no hybridization required. The p orbitals are used directly for bonding, and the s orbital electrons remain as a lone pair. Simple, elegant, and perfectly valid. It’s like nitrogen showing up to the party with three new friends and still having its original buddy for a chat on the side. No need for a DJ or a dance floor renovation.
Beyond Ammonia: Other Unhybridized Bonds
Ammonia is a fantastic illustration, but it's not the only place where nitrogen exhibits this unhybridized bonding. Think about molecules where nitrogen is bonded to other atoms, and it only forms three single bonds. For instance, in molecules like methylamine (CH₃NH₂), the nitrogen atom is still bonded to three other atoms (one carbon and two hydrogens), and it still possesses a lone pair. The bonding in these cases often doesn't require hybridization for a fundamental explanation of the number of bonds.
Essentially, any situation where nitrogen forms three single covalent bonds, and its electronic structure can be adequately described by using its 2s and 2p orbitals directly, falls into this category. The three unpaired electrons in the p orbitals engage in sigma bonds, and the lone pair in the s orbital remains as, well, a lone pair.
The Irony of Hybridization: When Things Get Fancy
Now, here’s where things get a little ironic. Nitrogen can form more than three bonds, but when it does, it usually needs help. This help comes in the form of hybridization. Hybridization is this cool concept where atomic orbitals (like the s and p orbitals) mix and combine to form new, hybrid orbitals. These hybrid orbitals have different shapes and energies and are often better suited for forming specific types of bonds or for accommodating more bonding partners.
For example, in many organic molecules, nitrogen is sp³ hybridized. This means its 2s orbital mixes with all three 2p orbitals to form four equivalent sp³ hybrid orbitals. Three of these sp³ orbitals are used to form sigma bonds with other atoms, and the fourth sp³ orbital holds the lone pair. This is common in amines and amides.
Nitrogen can also be sp² hybridized, which happens when it forms one double bond and two single bonds (like in imines). In this case, one s orbital and two p orbitals mix to form three sp² hybrid orbitals, and one unhybridized p orbital remains. The sp² orbitals form sigma bonds, and the unhybridized p orbital is used to form a pi bond (as part of the double bond).
And then there's sp hybridization, where one s orbital mixes with one p orbital to form two sp hybrid orbitals, leaving two unhybridized p orbitals. This is less common for nitrogen in stable neutral molecules but can be seen in certain contexts, like in the nitrile group (-C≡N), where nitrogen forms a triple bond. Here, two sp hybrid orbitals are used for sigma bonds, and the two unhybridized p orbitals are used for the two pi bonds.
But the question is about how many bonds nitrogen can make without hybridization. And as we’ve established, that number is elegantly three.

The Limits of the Unhybridized State
So, why the emphasis on "without hybridization"? Because hybridization is a way for atoms to become more versatile. It allows them to arrange their electron clouds in ways that are more energetically favorable for forming multiple bonds or for achieving specific molecular geometries. Think of it as upgrading from basic tools to a specialized toolkit. You can build a lot with basic tools, but the specialized ones let you build more complex and stable structures.
Without hybridization, nitrogen is limited by the inherent shapes and orientations of its s and p orbitals. The s orbital is spherical, and the p orbitals are dumbbell-shaped and oriented along the x, y, and z axes. While these can form sigma and pi bonds, the geometry and the number of available, distinct bonding orientations are more constrained compared to the hybrid orbitals.
The three unpaired electrons in the p orbitals naturally lend themselves to forming three sigma bonds, often with bond angles influenced by the relative orientations of these orbitals. The lone pair in the s orbital doesn't dictate a specific directional bonding geometry in the same way a hybrid orbital does.
The "Max" Myth and the Reality of Electron Counting
It’s easy to get caught up in the idea of "maximum" bonds. Sometimes, people might think nitrogen can form, say, four bonds because they see something like the ammonium ion (NH₄⁺). In NH₄⁺, nitrogen is bonded to four hydrogens. However, this ion has a positive charge, meaning nitrogen has lost an electron or is sharing its electrons in a way that results in a net positive charge. In this case, nitrogen is using hybridization (sp³ in fact) to accommodate those four bonds.
So, when we talk about the number of bonds nitrogen can make without hybridization, we're specifically referring to its inherent electron configuration and the direct use of its valence s and p orbitals for bonding. It’s a fundamental property, not a result of orbital rearrangement for a specific, often charged, species.

The beauty of the unhybridized state is its simplicity. It’s a direct reflection of the ground-state electron distribution. Nitrogen walks into the bonding arena with three unpaired electrons and one lone pair. It can use those three unpaired electrons to form three covalent bonds. That’s its unadulterated, no-frills bonding capacity.
The Takeaway: Respect the Basics!
So, the next time you’re staring at a molecular structure and wondering how many bonds an atom can form, remember the basics. Look at its valence electrons. See how many are unpaired in its ground state. For nitrogen, that's three unpaired electrons. And those three unpaired electrons are its ticket to forming three covalent bonds without needing the sophisticated dance moves of hybridization.
It’s a reminder that even in the complex world of chemistry, understanding the fundamental building blocks and their inherent properties can unlock a lot of understanding. Sometimes, the simplest answers are hiding in plain sight, just like Archimedes plotting his next aerial maneuver from atop the bookshelf. You just have to look closely!
It's a bit like knowing how many fingers you have on one hand. You have five, plain and simple. You don't need to invent extra fingers or reconfigure your hand to count to five. Nitrogen, in its unhybridized state, has three unpaired electrons ready to bond. Three is its number, its natural limit for direct, unassisted bonding.
And hey, if you ever find yourself stuck on a chemistry problem, remember the cat. Sometimes, a little chaos (or a well-timed distraction) can lead you to the simplest, most elegant truths. Embrace the basics, appreciate the unhybridized power, and remember that even nitrogen has its limits… at least until it decides to put on its hybridization hat!
