How Many Bonds Can Each Atom Make Without Hybridization
So, let's talk about atoms. Little guys, right? Tiny building blocks of everything. But they're not just sitting there. Oh no. They're busy little bees, always looking to make friends. And how do they make friends? By forming chemical bonds!
Now, you might think all atoms are the same when it comes to making bonds. Like, everyone gets two hands and that's it. But nope! It's way more interesting than that. Some atoms are super popular, others are a bit more selective. And it all depends on how many friends they can make, even before we get all fancy with science-y stuff like hybridization.
Think of it like this: before hybridization, an atom is just showing up to the party with its natural, unadorned orbitals. No fancy costume changes yet! It's all about what it's got inherently. So, how many buddies can each element snag without any of that high-tech orbital mixing?
The Usual Suspects: Singles and Doubles
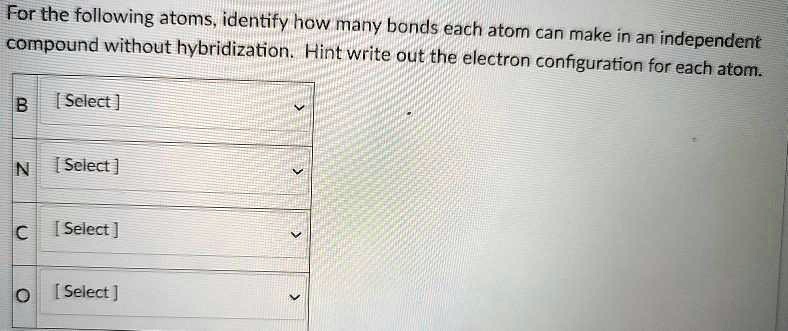
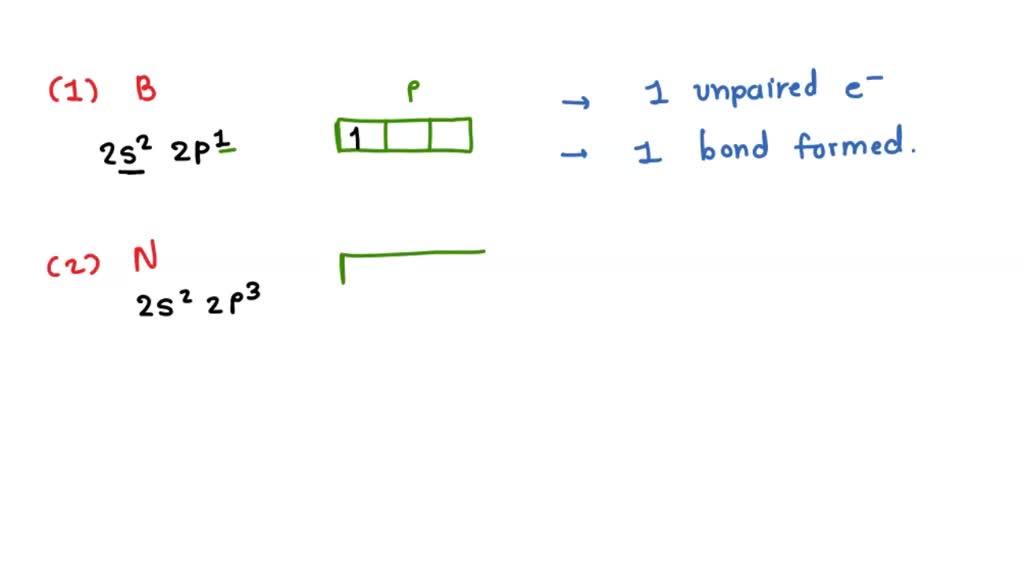
Let's start with the super common elements. You know, the ones that are literally everywhere. Like hydrogen. This little guy has just one electron in its outermost shell, the 1s orbital. That's like having one free hand. So, hydrogen can only make one bond. Pretty straightforward, right? It just wants one buddy to complete its little happy shell.
Then we have oxygen. Oxygen is a bit more of a social butterfly. It's got two unpaired electrons. That means it's ready to make two bonds. Think of water (H₂O). One oxygen, two hydrogens. Perfect! Oxygen is often the glue that holds things together, making two connections to satisfy its bonding urges.
Nitrogen is another big player. It’s got three unpaired electrons. So, guess what? It can make three bonds. The classic example is ammonia (NH₃). Three hydrogens clinging to that central nitrogen. It’s like nitrogen is saying, "Come on over, I've got room for three!"
And then there's carbon. Ah, carbon. The star of the show! Carbon is the backbone of life, literally. On paper, without hybridization, carbon has two unpaired electrons. So, it should only make two bonds, right? But here's where things get really interesting. Carbon is like the chameleon of the chemical world. It usually makes four bonds, and that's thanks to hybridization. But if we're sticking to the no hybridization rule, it's a bit more reserved. Still, it can handle two bonds in some basic scenarios, especially if it's paired with a super electronegative atom that pulls electrons strongly.
The Halogen Huddle: Always One and Done
Let's talk about the halogens. These are elements like fluorine, chlorine, bromine, and iodine. They're all in the same group and they're all a bit… clingy. They have seven electrons in their outer shell, just one short of a full house. This means they're always looking for that one last electron. So, naturally, they can only make one bond. They are the ultimate one-trick ponies of bonding, and they're perfectly happy with it.
Think of table salt, sodium chloride (NaCl). Sodium is happy to give away an electron, and chlorine is thrilled to take it. One bond, and they're both satisfied. It's the ultimate minimalist relationship.
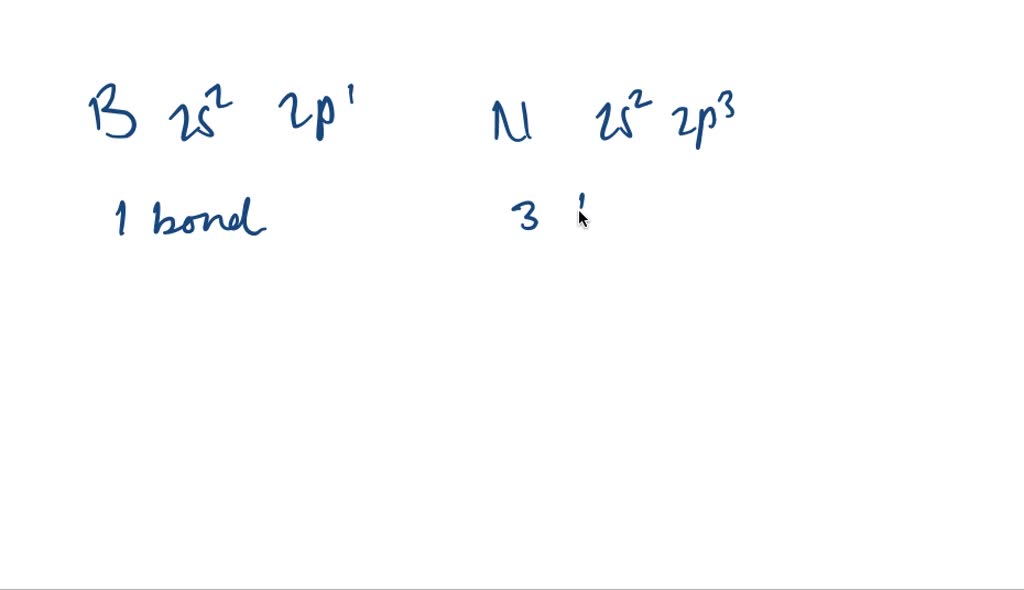
Noble Gases: The Aloof Elite
And then we have the noble gases. Helium, neon, argon… these guys are the ultimate introverts. They have full outer electron shells. Like, totally complete. They have no need for friends! So, normally, they make zero bonds. They are the loners of the periodic table, perfectly content in their own electronic bubbles. It’s like they’ve already achieved peak popularity and don't need any more connections.
Now, before you go thinking they’re completely antisocial, there are some very rare and very specific circumstances where some of the heavier noble gases can be coaxed into making a bond. But for the most part? They’re happy to stay single.
The Quirky Ones: More Than Meets the Eye
Okay, but what about the elements that don't fit neatly into the "one, two, or three" boxes? Things get a bit more… flexible.

Consider sulfur. In its elemental form, sulfur often forms rings where each sulfur atom is bonded to two other sulfur atoms. So, it can make two bonds. But when it's forming compounds, sulfur can be quite the overachiever. In sulfate ions (SO₄²⁻), for example, sulfur is surrounded by four oxygen atoms and is considered to have a formal oxidation state that implies it's involved in more than just two bonds. This is where hybridization really comes into play, allowing for more complex arrangements.
Phosphorus is another interesting case. Like nitrogen, it has five electrons in its outer shell. In its elemental form (P₄), it forms a tetrahedral structure where each phosphorus atom makes three bonds. But in many compounds, phosphorus can expand its bonding horizons. Think of phosphate (PO₄³⁻). It can participate in more than three bonds when its electron configuration gets a little… creative.
Why It Matters (and Isn't So Scary!)
So, why is this whole "bonds without hybridization" thing even a thing? Because it’s the foundation! It’s the basic blueprint before the fancy architecture starts. Understanding these natural tendencies helps us predict how atoms will initially interact.
It’s like knowing a person’s natural personality before they start dressing up for Halloween. Hydrogen is always going to be a bit of a loner, happy with one friend. Oxygen is naturally inclined to be the mediator, making two connections. It's the inherent way they are built.
And the fact that elements like carbon can break these "rules" is what makes chemistry so darn exciting! It’s not rigid. It’s adaptable. It’s full of surprises. This whole topic, even the simple version, shows us that atoms have personalities. They have preferences. They have a natural capacity for connection.
It's a little peek behind the curtain of molecular construction. No complex math, no mind-bending theories, just the raw, unadulterated bonding potential of each element. And honestly? That's pretty cool. It’s a simple concept, but it unlocks a whole universe of understanding about how everything around you is put together. So next time you see a water molecule, remember that oxygen was just naturally inclined to make two friends. Simple, elegant, and fundamental.
