How Many Atoms Are In This Carbohydrate Molecule C6h12o6
Hey there, coffee buddy! So, you wanna talk atoms, huh? Specifically, you’re curious about this groovy little molecule called C6H12O6. Sounds fancy, right? Like something you'd find in a super-science lab. But honestly, it's way more familiar than you think. Ever had a donut? A piece of fruit? A spoonful of sugar? Bingo! You've basically encountered this dude.
We're talking about sugars here, my friend. The good stuff that fuels your day. And C6H12O6? That's the universal sugar formula. Like the supermodel of the sugar world. Think glucose, fructose, galactose – they all rock this same basic atomic ensemble. Pretty neat, right? Like a secret code for sweetness.
So, the big question is: how many atoms are we even dealing with here? Let's break it down, nice and easy, just like we're figuring out the best way to dip our biscuit. No need for a calculator that glows in the dark, I promise!
Let's Count 'Em Up!
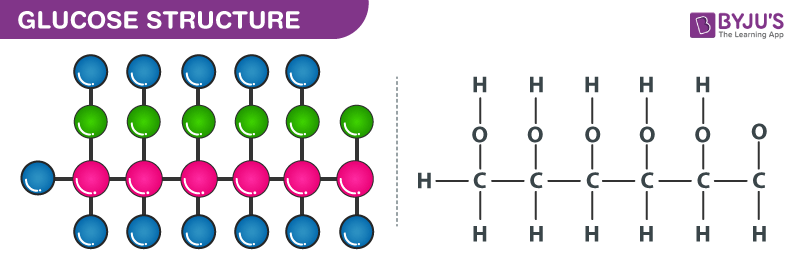
Alright, so we've got this code: C6H12O6. What does that even mean? It’s like a recipe, but for tiny building blocks. Each letter is an element, and the number tells you how many of that element are hanging out in one molecule.
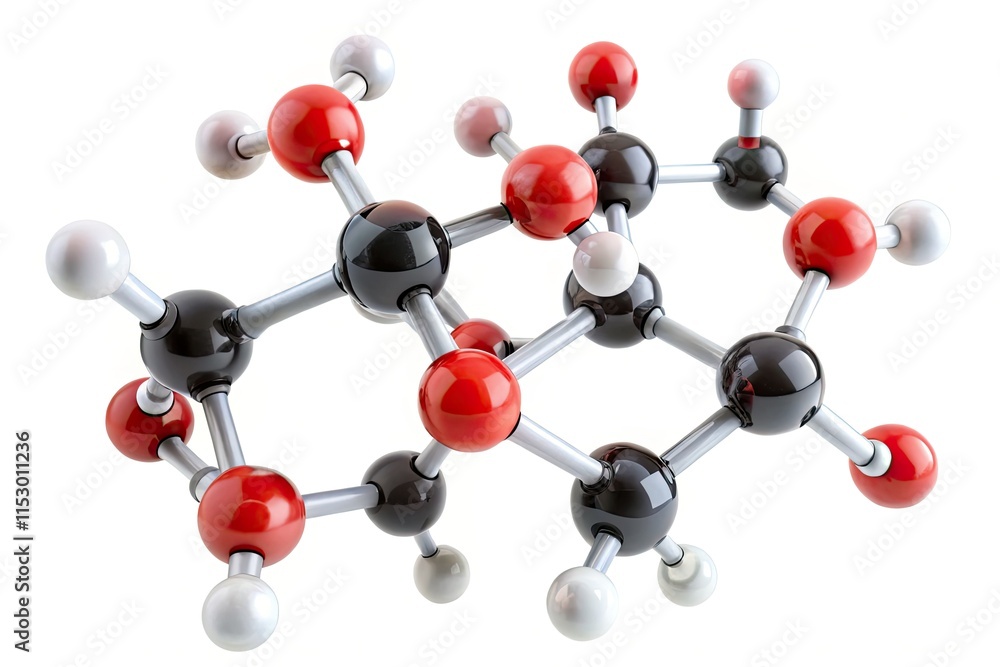
First up, we have the 'C'. That stands for Carbon. And how many carbons do we have? The little '6' right after it tells us. So, that's 6 Carbon atoms. Easy peasy, lemon squeezy. Imagine 6 little black dots. Got it?
Next, we've got the 'H'. That's for Hydrogen. And that number? A whopping 12. So, 12 Hydrogen atoms. These guys are the smallest and most common atoms in the universe, you know. Like the background extras in a blockbuster movie. Always there, but sometimes overlooked.
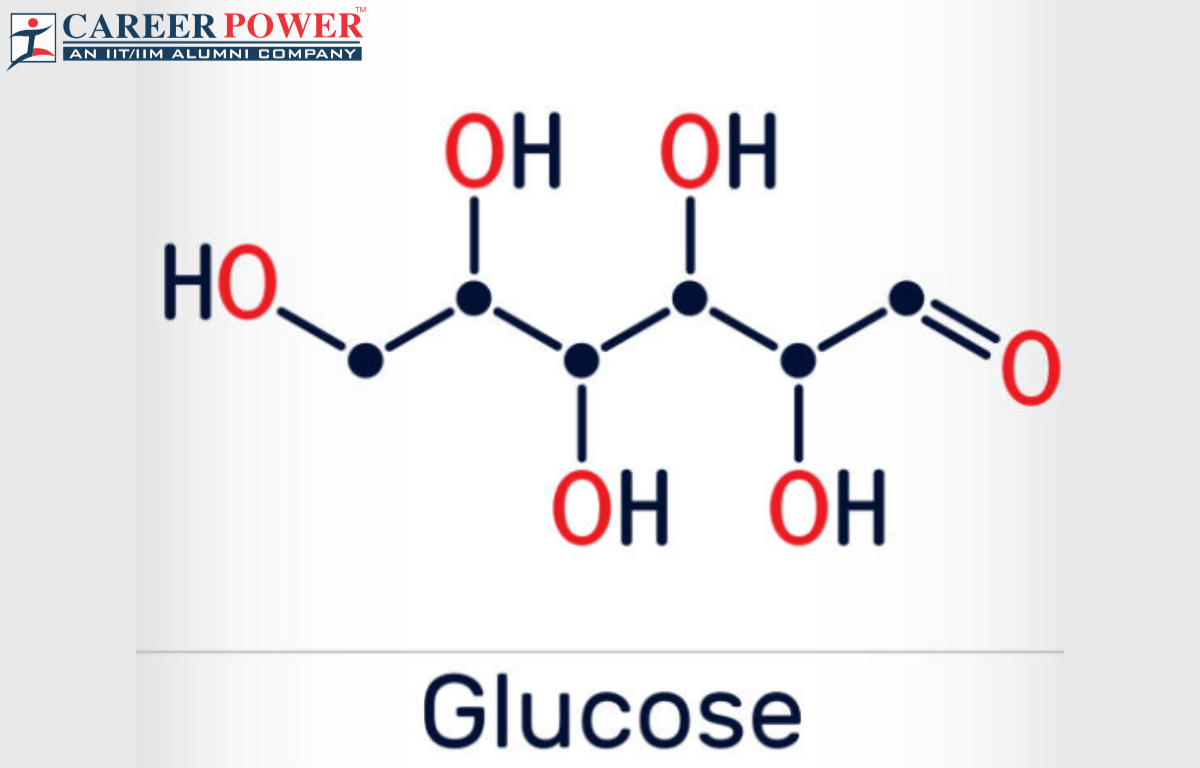
And finally, the 'O'. That's for Oxygen. And the number here is 6. So, 6 Oxygen atoms. Oxygen, the stuff we breathe! Essential for life, and apparently, for sugar too. Makes sense, right? We need both to keep going.
Putting It All Together, Atom-Style!
So, we've got our little groups: 6 Carbons, 12 Hydrogens, and 6 Oxygens. To find the total number of atoms in this C6H12O6 molecule, we just gotta add them all up. It’s like counting the number of sprinkles on your cupcake. Gotta get the whole picture!
Let's do the math, because even though we're casual, we can still be smart. It's 6 + 12 + 6. Drumroll please…
… that equals a grand total of 24 atoms! 🎉
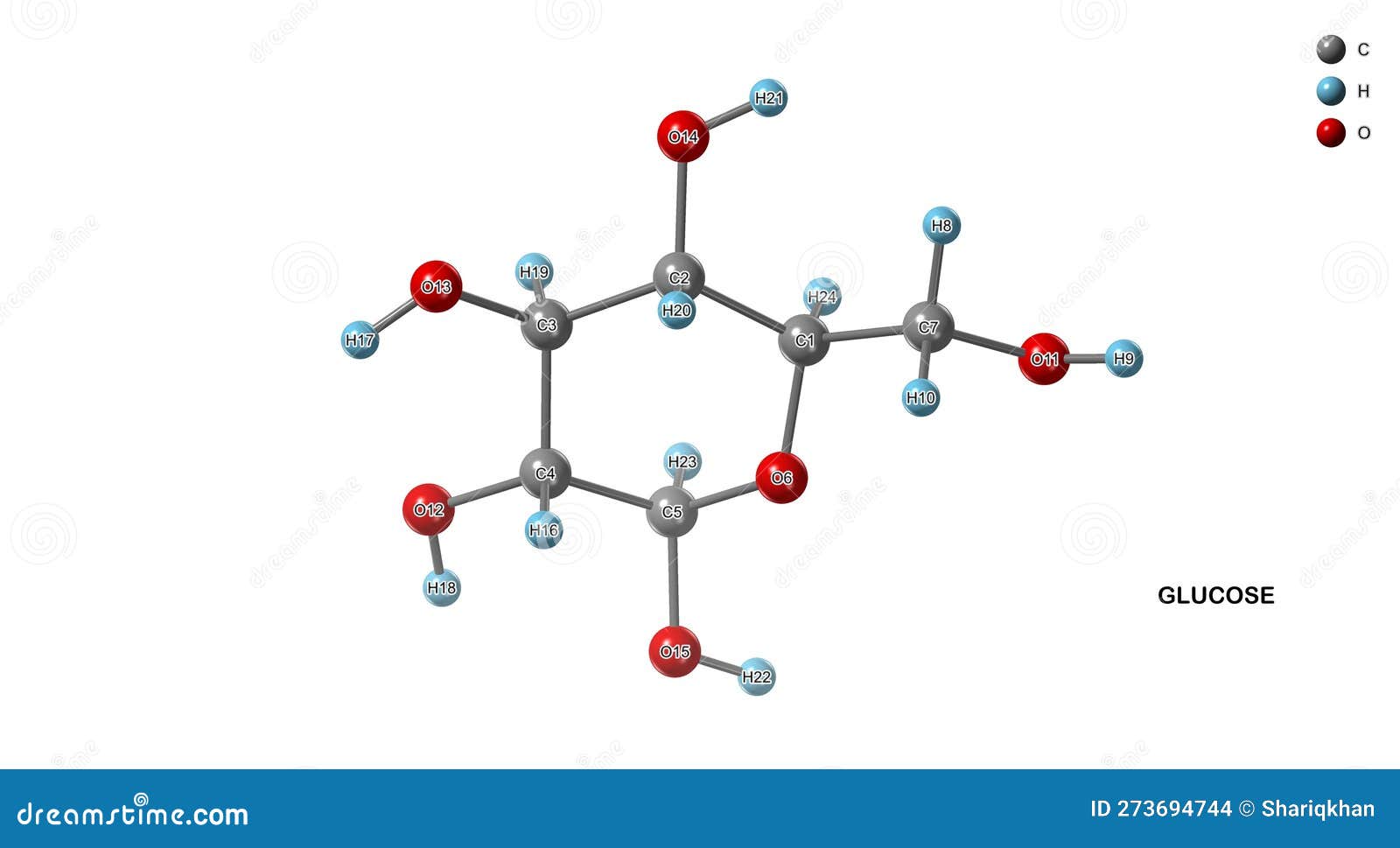
Yep, just 24 little guys, all holding hands (or, you know, forming chemical bonds) to create one single molecule of this fundamental sugar. Isn't that wild? So much happening in something so small. It’s like a miniature universe of its own. Mind. Blown. Or at least, slightly impressed, right?
Why Does This Even Matter? (Besides Sweetness!)
Okay, so counting atoms is fun, but you might be thinking, "Why do I care about 24 atoms in a sugar molecule?" Great question, my friend! It’s like asking why we need to know how many ingredients are in our favorite cookie recipe. If you want to bake it perfectly, you kinda need to know the proportions, right?
Understanding the atomic makeup of molecules like C6H12O6 is the foundation of a lot of things. Think about it: this molecule is the primary fuel source for your cells. It’s what gives you that burst of energy when you eat something sweet. Without this structure, your body wouldn't know how to break it down and use it for power.
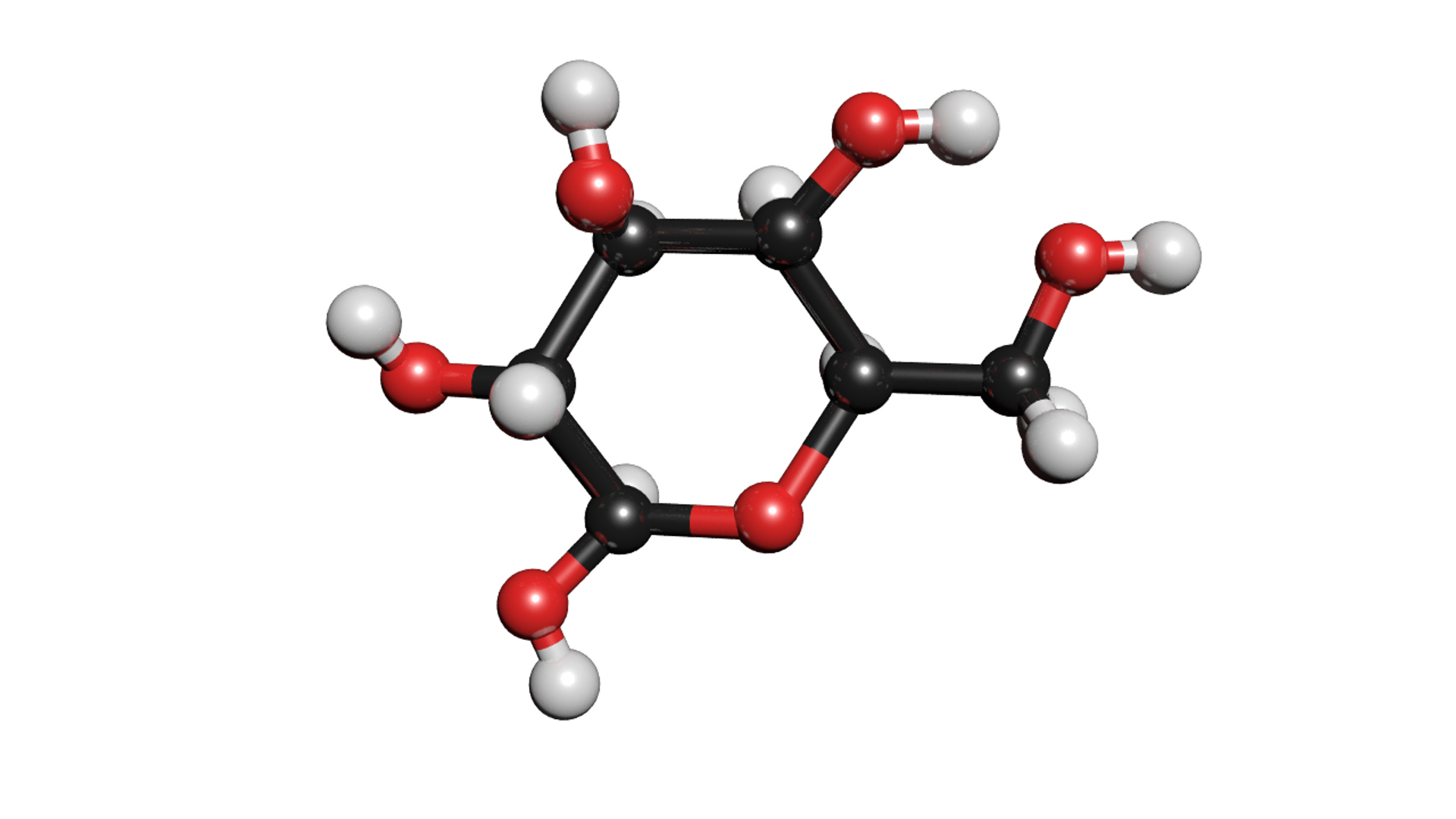
And it’s not just about eating. It’s about how your body works. How plants make their food through photosynthesis. How we digest things. How our muscles contract. It all comes down to these tiny molecular arrangements. So, that 24 atoms? It's the key to unlocking a whole bunch of biological mysteries.
Plus, scientists use this knowledge for all sorts of cool stuff. Developing new medicines, understanding diseases, even creating biofuels. It all starts with knowing what’s inside the ingredients, you know? It's like being a detective, but for molecules. And our clue today is 24 atoms.
So, What's the Takeaway?
Basically, the next time you enjoy something sweet, you can casually drop this fact: "You know, this deliciousness is made up of a total of 24 atoms! 6 carbons, 12 hydrogens, and 6 oxygens. Pretty wild, right?" You'll sound like a total science rockstar. Or at least, someone who had a really interesting coffee chat.
It’s a good reminder that even the simplest things in life, like a sugar molecule, are incredibly complex and fascinating when you look a little closer. It's the little things, literally atoms, that make up the big picture. And isn't that just… awesome?
So there you have it. C6H12O6: a sugar molecule with 24 atoms. Not so scary after all, right? It’s just a friendly little collection of atoms, doing its job, making life a little sweeter and a whole lot more energetic. Now, pass the sugar for my coffee… just kidding! 😉
Seriously though, it's mind-boggling when you think about how many of these molecules are in just a tiny grain of sugar. Billions? Trillions? More? The universe is full of these tiny, intricate wonders, and C6H12O6 is just one of the many stars in the molecular sky. And we’ve just counted all its members. High five!
It's like looking at a mosaic. You see the big picture, the beautiful image, but it's all made up of millions of tiny little tiles. In our case, those tiles are atoms. And for this particular, very important and very tasty mosaic, we have exactly 24 tiles per picture. Who knew counting could be so… energizing?
Think about the scale of it all. You can’t see these atoms with your naked eye, not even close. They're so incredibly tiny, it’s almost impossible to comprehend. Yet, they’re the building blocks of everything around us. Your coffee mug, your phone, me, you, this whole darn planet! It’s all just atoms hanging out, doing their thing. And this sugar molecule is a perfect, sweet little example of that. So, next time you're feeling peckish, remember the tiny universe you're about to consume. And that it’s made of exactly 24 atoms.
