How Many Atoms Are Equal To 1.5 Moles Of Helium
You know, the other day I was staring at a balloon. Just a regular, helium-filled party balloon, the kind that makes your voice sound all squeaky and silly. My nephew had his birthday, and the room was just swimming with them. And as I watched one of them bobbing around, a thought, as random as a rogue confetti bit, popped into my head: how many tiny little things are actually in there? It’s not just empty space and a bit of gas, right? There has to be something making it float, something…atomic.
It got me thinking about how we quantify things in science. We talk about grams, liters, and then, when things get really, really small, we use these weird units like moles. It’s a bit like trying to count grains of sand on a beach – you can’t just say "a handful," you need a more precise way to measure. And that’s where the mole comes in. It’s basically a chemist’s magic number for grouping tiny particles.
So, if I have a bunch of these helium balloons, and I’m told I have, say, 1.5 moles of helium, what does that really mean in terms of the actual, physical atoms zipping around inside? It feels like a question that’s just begging to be answered, doesn't it? Like unlocking a secret code to the universe, but on a slightly smaller, more manageable scale. We’re not talking about galaxies here, just a bit of gas. But even a bit of gas is made of stuff!
The Humble Mole: Our Tiny-Particle Superhero
Before we dive into the glorious world of helium atoms, let’s have a quick chat about the star of our show: the mole. Honestly, I used to find the term "mole" incredibly confusing. It sounds like that little velvety creature that digs tunnels, or a suspicious mark on your skin. Turns out, in chemistry, it’s neither. It's just a number. A really, really big number. Think of it like a baker’s dozen. A dozen is 12. A baker’s dozen is 13. A mole is… well, it’s a lot more than that.
This number is called Avogadro's number, named after an Italian scientist who, thankfully, wasn't obsessed with digging. Avogadro’s number is approximately 6.022 x 1023. Let that sink in for a second. That’s a 6, followed by 23 zeros. It’s a number so astronomically huge, it makes the national debt look like pocket change. It's the kind of number that makes you question the sanity of scientists, but also marvel at their dedication to counting the uncountably small.
So, what does this massive number represent? It represents the number of elementary entities – usually atoms, molecules, ions, or electrons – in one mole of a substance. It's our way of taking a manageable chunk of incredibly tiny things and giving it a name. It’s like saying, "Okay, instead of trying to count every single grain of sand, let’s just agree that a 'heap' is this many grains." Except, in chemistry, that "heap" is a mind-bogglingly precise quantity.
Why did scientists even invent this? Because dealing with individual atoms is practically impossible. Imagine trying to pick up one single atom of helium to weigh it. You'd need microscopic tweezers the size of an atom itself, and even then, how would you know you only picked up one? It’s simply not feasible. So, they needed a way to work with amounts of substances that are large enough to be measured by standard lab equipment, but still correspond to a specific, countable number of particles. Enter the mole!

Helium: The Light and Lively Element
Now, let’s talk about our friend, helium. It’s element number 2 on the periodic table, which means each atom of helium has 2 protons in its nucleus. It’s a noble gas, meaning it’s super stable and doesn't like to play nicely with other elements. It’s also incredibly light, which is why it’s used to make balloons float. If you've ever inhaled a bit of helium from a balloon (don't do it too much, it can be dangerous!), you know it makes your voice go all high and funny. That’s because helium is less dense than air, so sound travels faster through it, altering the pitch of your voice.
So, we know we're dealing with helium atoms. And we've got this number, Avogadro's number, which tells us how many atoms are in one mole of any substance. It's like a universal conversion factor for tiny things. It’s a constant, a bedrock of our understanding of matter at the atomic level. Amazing, really, how such a seemingly abstract concept has such concrete implications.
When we talk about a mole of helium, we are talking about 6.022 x 1023 individual helium atoms. Just to be clear, this is an enormous number of atoms. If you could somehow line them up, they would stretch for miles and miles, probably all the way to the moon and back, several times over. It’s a scale that’s almost impossible for our brains to truly grasp. We're used to thinking in terms of dozens, hundreds, maybe thousands. But 1023? That’s a whole other ball game.
This number is fundamental. It's the bridge between the macroscopic world (the stuff we can see and touch) and the microscopic world (the atoms and molecules that make it up). Without Avogadro's number, doing calculations involving chemical reactions and the amounts of substances would be incredibly cumbersome and prone to error. It’s the secret handshake of chemists, the silent understanding that allows them to predict how much of one thing will react with another.
The Big Calculation: 1.5 Moles of Helium Atoms
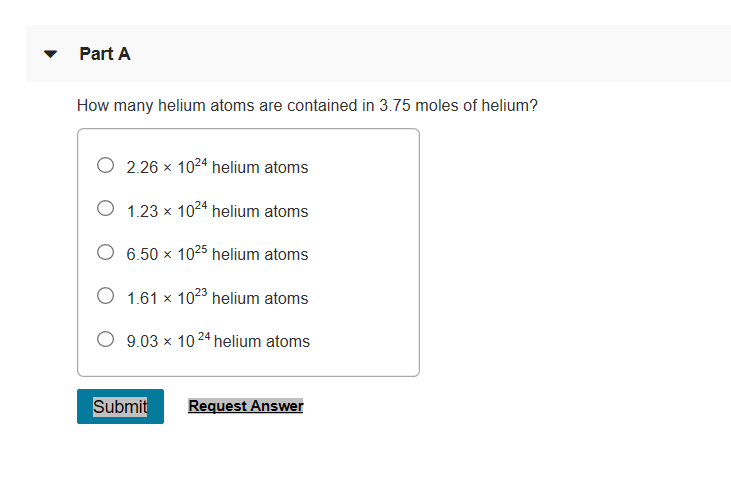
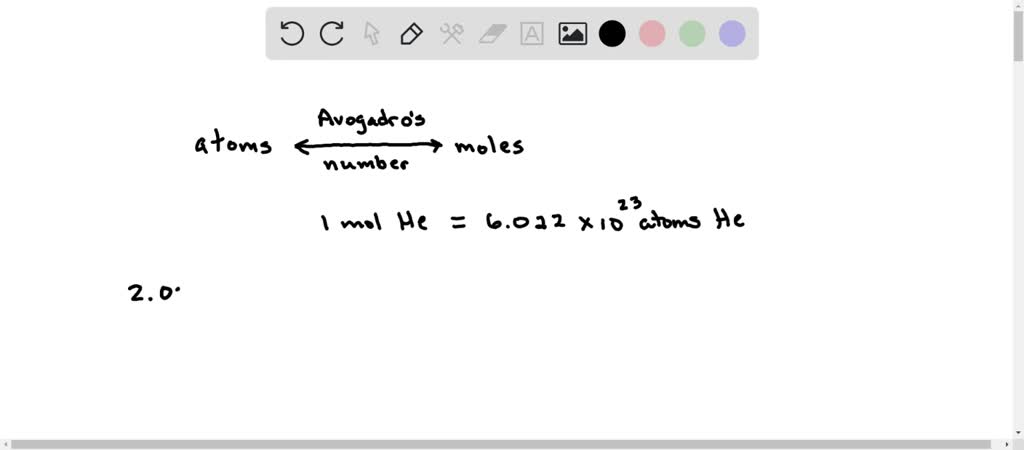
Okay, so we’re back to our original question: how many atoms are equal to 1.5 moles of helium? This is where the magic happens, or rather, the simple multiplication. We know:
- 1 mole of helium contains 6.022 x 1023 helium atoms.
- We have 1.5 moles of helium.
To find out the total number of atoms, we just need to multiply the number of moles by Avogadro's number. It's like having 1.5 bags, and each bag has 6.022 x 1023 marbles. How many marbles do you have in total? You multiply the number of bags by the number of marbles per bag.
So, the calculation is:
Number of atoms = Moles of helium x Avogadro's number
Number of atoms = 1.5 x (6.022 x 1023)

Let’s do the math. Multiplying 1.5 by 6.022 gives us 9.033.
So, the total number of helium atoms is 9.033 x 1023.
There you have it! In 1.5 moles of helium, there are approximately 9.033 x 1023 individual helium atoms. That's 903,300,000,000,000,000,000,000 atoms. That’s a nine followed by 23 zeros, with a few digits in front. It’s still a number that’s hard to wrap your head around, but it's the precise answer to our balloon-related musings. Imagine trying to count those! You'd need a lot of party guests and a very, very long time.
Think about it: that's a tiny speck of helium, probably not even enough to fill a small party balloon, yet it contains an almost incomprehensible amount of individual atoms. It really puts into perspective just how incredibly small atoms are. They are the building blocks of everything, and they exist in quantities that dwarf our everyday experience.

The Significance of Such a Huge Number
Why is this number so important? Because it allows chemists to work with matter on a practical level. When they're in the lab, they don't measure out individual atoms. They measure out grams or liters. But those measurements are directly related to the number of moles, and therefore, the number of atoms or molecules. So, when a chemist uses, say, 4 grams of helium, they know they are working with approximately 1 mole of helium atoms, which means they are working with 6.022 x 1023 atoms.
This is crucial for understanding chemical reactions. For a reaction to occur, atoms need to collide and rearrange. The rate and outcome of these reactions depend on the number of particles involved. By using moles, scientists can predict how much of a substance is needed to react completely with another substance, ensuring that experiments are efficient and predictable. It's like having a recipe where the ingredients are measured in "dozens of eggs" instead of individual eggs.
And it’s not just about chemical reactions. Understanding the number of atoms in a given amount of substance is fundamental to many areas of science, from materials science (how many atoms are in a piece of metal?) to atmospheric science (how many molecules of gas are in a volume of air?). It’s the foundation upon which much of our scientific knowledge is built. It’s the ultimate counting tool for the minuscule.
So, next time you see a helium balloon floating away, remember that it’s not just a colorful orb. It’s a container holding an astonishingly large number of incredibly tiny, incredibly important, helium atoms. And the number 1.5 moles is just a convenient way for scientists to talk about that immense quantity of matter. It’s a reminder that even the most mundane things are made of wonders on a scale that’s almost beyond our comprehension.
It’s a bit mind-boggling, isn’t it? The universe is full of these tiny, invisible building blocks, and we’ve come up with these clever ways to count them. Science can be pretty neat, even when it’s dealing with numbers that would make a supercomputer sweat. And all this from a simple party balloon and a curious mind! Who knew a bit of squeaky gas could lead to such an epic numerical journey?
