How Many Asymmetric Centers Does Cholesterol Have
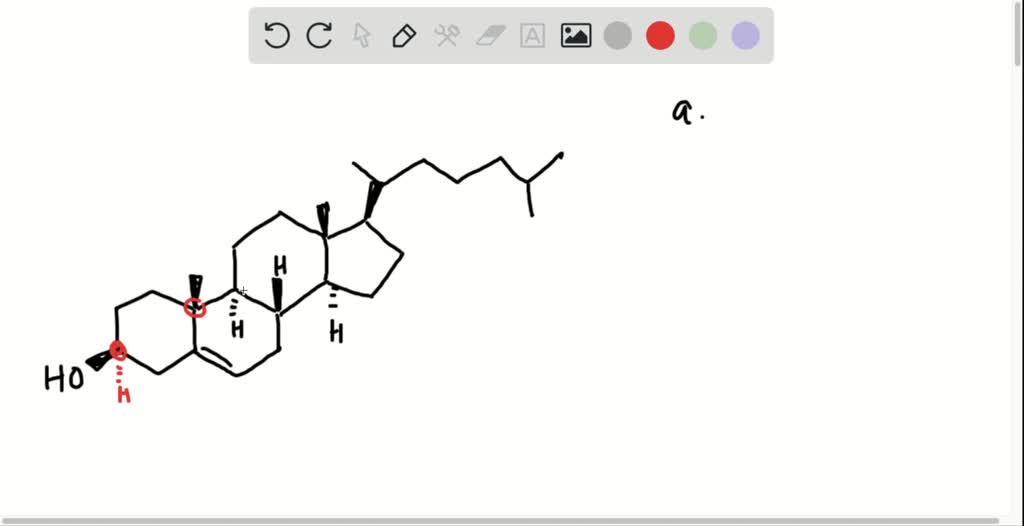
Ah, cholesterol. The word itself conjures up images of greasy fries and maybe a stern-faced doctor. But beneath that sometimes-shady reputation lies a molecule that’s actually pretty darn important. It’s like the bouncer of your cells, making sure they’re not too flimsy. And it’s got a secret life, a little game of “spot the difference” happening within its structure. We’re talking about something called asymmetric centers. Sounds fancy, right? Like something you’d find in a chemistry textbook with a lot of intimidating diagrams. But let’s break it down, the fun way.
Imagine a molecule as a little LEGO creation. Most of the time, the pieces are pretty straightforward. But then, sometimes, you have a special LEGO brick. This brick is like a celebrity. It has four different “friends” attached to it. Not just two or three, but a full quartet of distinct buddies. This makes our special brick – or in chemistry terms, our chiral carbon atom – a bit of a diva. It can’t be perfectly mirrored. Flip it over, and it’s like looking in a funhouse mirror. It’s a unique configuration. These are our asymmetric centers.
Basically, it’s like having a four-sided die where each side is a different color. You can’t just flip it and make it look the same. It’s always going to show a different face.
Now, cholesterol, our cell-bouncer extraordinaire, is a pretty big and complex molecule. It’s not just a simple ring. It’s got a whole bunch of appendages and bits and bobs sticking out. And in this rather substantial structure, it’s hiding some of these special, four-armed LEGO bricks. It’s got quite a few of them, actually. We’re not talking one or two. We’re talking a respectable number that makes it a rather sophisticated piece of molecular architecture.
So, how many of these asymmetric centers does this famously misunderstood molecule possess? Drumroll, please… it’s eight! Yes, a whopping eight. That means there are eight different carbon atoms in the cholesterol molecule that are each holding onto four completely different groups of atoms. Think of it as eight little decision-making points within the molecule. Each one is like a tiny crossroads where the path could go in four different directions, and each direction is unique.
Now, I know what you might be thinking. “Eight? That’s a lot. How do they even keep track?” Well, that’s the magic of chemistry, I suppose. These asymmetric centers are what give cholesterol its specific three-dimensional shape. And its shape is super important. It dictates how it interacts with other molecules, like fitting into cell membranes or being recognized by enzymes. It’s the reason why, when your body needs to build something or signal something, it uses this specific arrangement of atoms, not a slightly different one.
It’s kind of like having a set of keys. You have a bunch of keys, and they all look vaguely similar, but only one will open your front door. The tiny differences in their cuts, like the subtle twists and turns of those asymmetric centers in cholesterol, are what make them unique and functional. If one of those asymmetric centers were different, the whole key wouldn't fit. The whole molecule might not work properly.
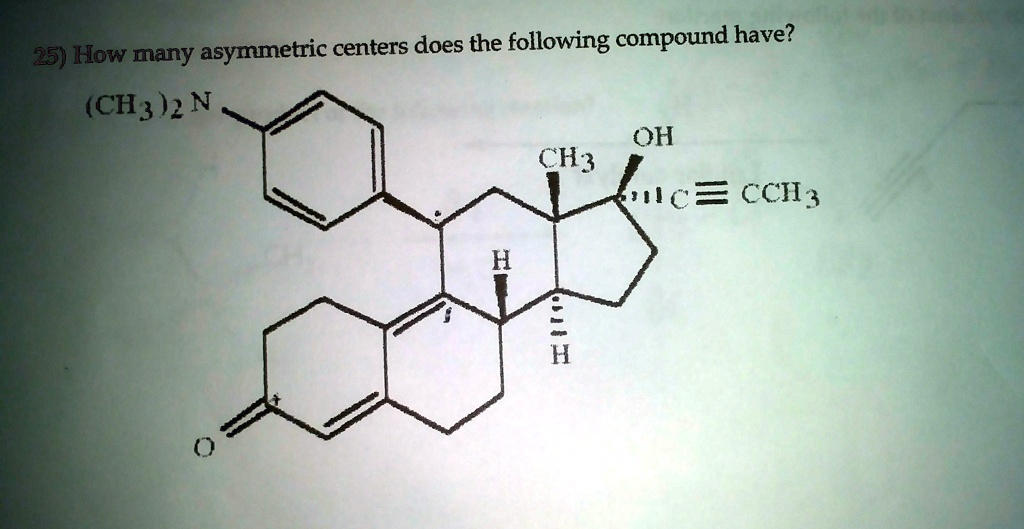
And here’s where it gets a little bit more interesting, and perhaps a little unpopular opinion time: I kind of admire cholesterol for its complexity. We tend to blame it for all sorts of things, but at its core, it's a marvel of natural engineering. Those eight asymmetric centers aren’t there to cause trouble; they’re there to make cholesterol do its job effectively. It’s like saying a car’s engine is “bad” because it has many intricate parts. No, those parts are what make the car go!
Think about it. This one molecule, with its eight little points of asymmetry, plays a role in building cell walls, making hormones like testosterone and estrogen (yes, really!), and even helping you digest your food. It’s the unsung hero, the behind-the-scenes player, and it achieves all this thanks to its intricate structure, including those eight asymmetric centers. So, the next time you hear about cholesterol, maybe give it a little nod of respect. It’s a molecule with a lot going on, and those eight asymmetric centers are a big part of its fascinating story.
It’s just a molecule, folks. A rather busy, multi-talented molecule with a lot of unique twists and turns. And I, for one, am not mad at it. In fact, I think it’s pretty cool that something that gets such a bad rap is actually so structurally… interesting. Those eight little chiral carbons are like the molecule’s quirky personality traits, making it distinct and, dare I say, rather elegant in its own molecular way.
