How Many Asymmetric Centers Are Present In The Compound Below
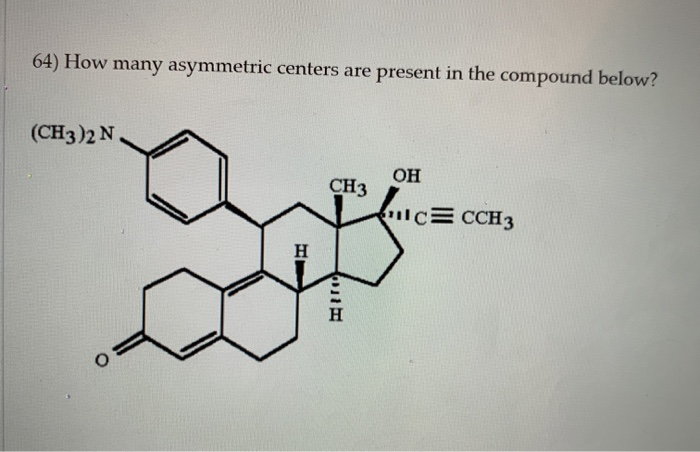
Hey there, fellow curious minds! Ever just look at a molecule and wonder, "What's going on in there?" Today, we're diving into something super cool in the world of chemistry that’s all about a molecule's "handedness" – kind of like how our left and right hands are mirror images but not quite the same, right? We're going to figure out how many asymmetric centers are in the molecule you see below. Don't worry, we'll keep it chill, no intense equations needed, just a friendly exploration of why this stuff is so neat.
So, what exactly is an asymmetric center? Think of it like a special kind of carbon atom. Normally, carbon atoms are happy to share their four bonds equally, making them pretty symmetrical. But sometimes, a carbon atom gets a bit of a personality change. Instead of being attached to four identical things, it ends up bonded to four different groups. This is where the magic, and the asymmetry, happens!
Why is this so interesting? Well, these little pockets of asymmetry are responsible for a ton of things we experience every day. Think about how your body recognizes things. Your sense of smell, your taste buds, how medicines work – a lot of it comes down to molecules fitting into specific spots like a key in a lock, and that shape is dictated by these asymmetric centers. It's like having the wrong glove on; it just doesn't feel right, does it?
Let's get down to business with our molecule. Imagine you're a detective, and your mission is to find all the carbon atoms that are playing the "four different things" game. We'll need to carefully examine each carbon atom in the structure.
We’ll start scanning from one end, just like reading a book, to make sure we don't miss a single carbon. For each carbon atom, we’ll ask ourselves: "Are you attached to four completely unique things?" It's a bit like a dating profile for atoms. If it's "single" and has four different potential partners, then bingo! We've found an asymmetric center.
Let’s take a closer look at the molecule provided. We'll number the carbons for easy reference, but for this chat, we'll just point them out as we go. Don't get overwhelmed by all the lines and letters; it's like a puzzle, and we're just finding the oddly shaped pieces.
See that carbon atom right there? Let's check its neighbors. Is it holding onto four distinct groups? If it is, then congratulations, that's our first asymmetric center! It's like finding the first clue in a treasure hunt. This means our molecule has a chiral center. You might have heard of chirality before – it's that concept of 'handedness' we talked about. Molecules with asymmetric centers are chiral, and they exist as enantiomers, which are like those non-superimposable mirror images.
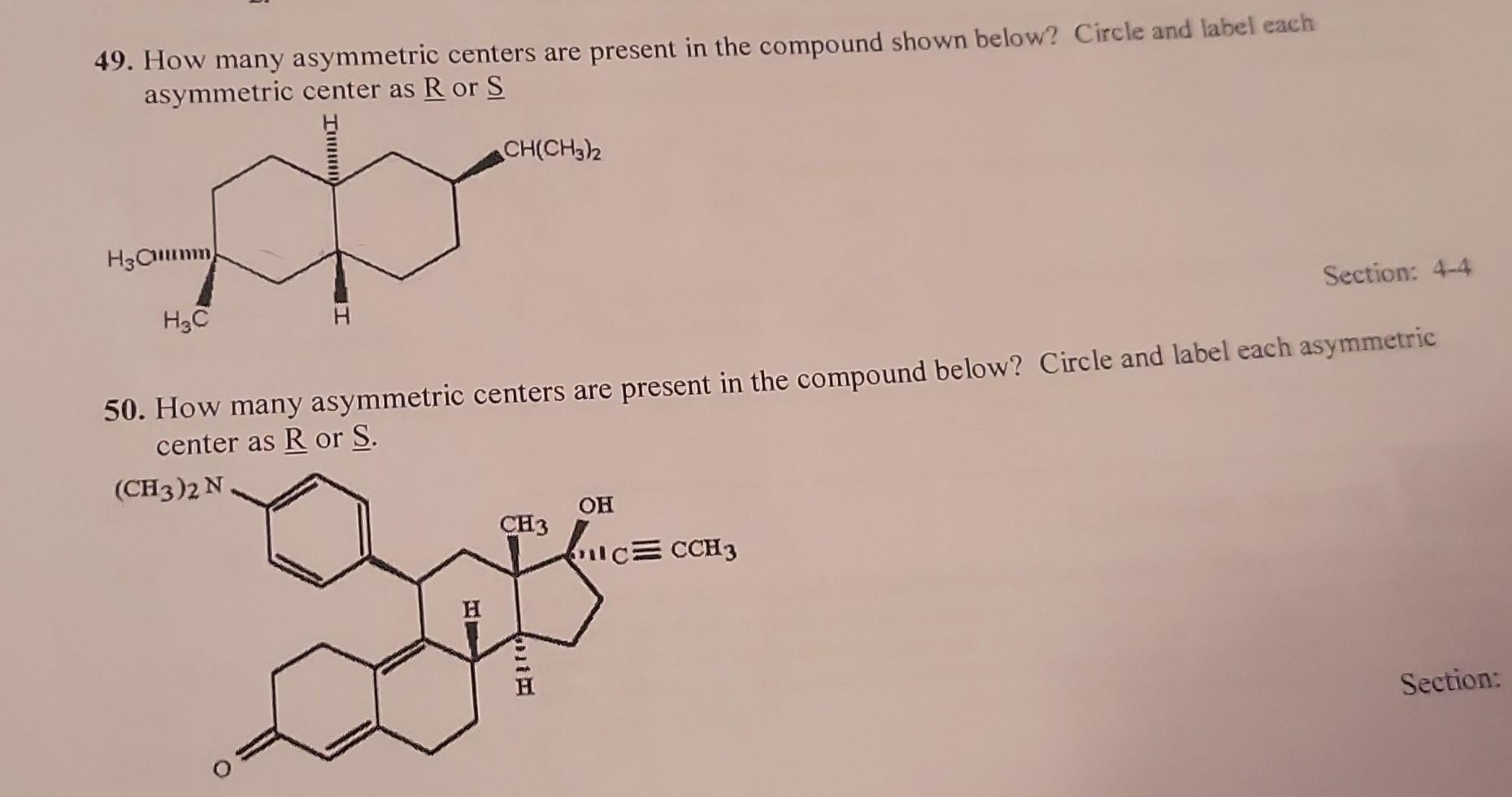
Now, let’s systematically go through each carbon atom. We need to be super diligent. Imagine you're a meticulous baker, ensuring every ingredient is perfectly measured. We have to be just as careful here.
We'll look at the carbon that’s part of the main chain. Is it connected to… (let’s list them out broadly) …a hydrogen, an alkyl group (that's just a chain of carbons and hydrogens), and then two different directions down the molecule?
If the answer is a resounding "yes!" for a particular carbon, we tick that box. It’s like finding another gem in our treasure chest. Remember, the key is four different substituents. If two of them are the same, then that carbon doesn't count as asymmetric. It's like trying to match socks; if you have two identical red socks, they don't contribute to an interesting pattern of asymmetry, do they?
Let's trace our way through the molecule. We're looking for those carbons that are truly unique in their neighborhood. It's not just about what's immediately attached, but what those attachments lead to further down the line as well. Sometimes, you have to look a little further to see the difference, like noticing the subtle variations in a forest.
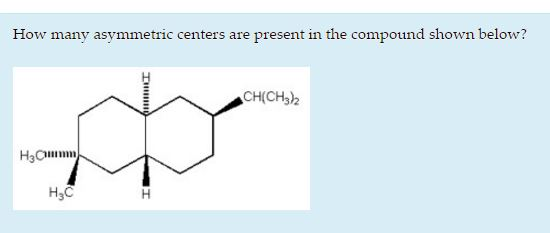
So, for carbon number X (hypothetically speaking, of course!), we see it’s bonded to a hydrogen atom. Good start. Then, it's bonded to a methyl group (CH3). That's two. Now, look at the rest of the molecule branching off. On one side, it connects to a different part of the chain. On the other side, it connects to a yet another different part. Are these two remaining branches different from each other? If they are, and the hydrogen and methyl group are also distinct, then boom! That carbon is an asymmetric center.
We’ll continue this process for every single carbon atom in the molecule. It's a systematic approach, like a crossword puzzle solver. We’re not guessing; we’re verifying.
Are there any carbons that are part of double bonds? Those carbons are typically not asymmetric because they only have three attachments, not four. So, we can often ignore those right away. It’s like ruling out certain puzzle pieces that clearly don't fit the slot.
What about carbons that are at the very end of a chain, like a methyl group (CH3)? These guys are usually attached to three hydrogens and one other group. Since they have three identical hydrogens, they can't be asymmetric centers. They're the simple, symmetrical shapes in our puzzle.
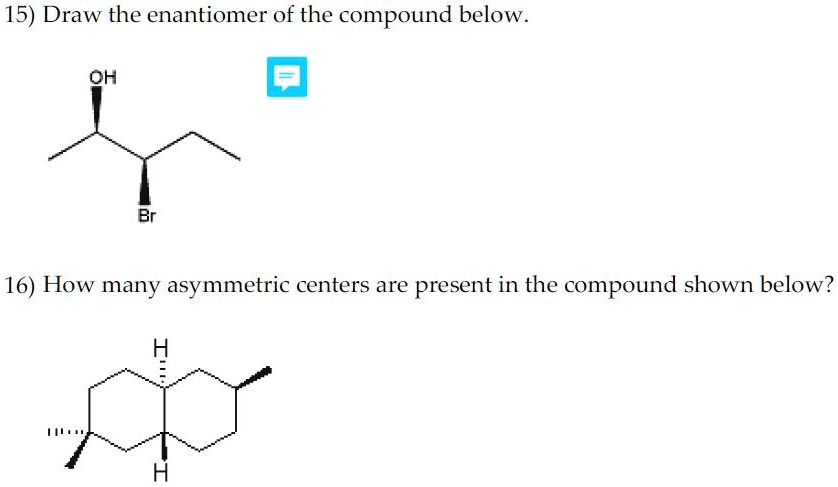
Now, let's focus on the internal carbons of the molecule. This is where the action is! We meticulously examine each one.
Let’s say we’ve gone through carbons 1, 2, 3, and so on. For carbon number 1, we check its four attachments. If they are all different, we count it. For carbon number 2, we do the same. And for carbon number 3… and so on, until we've examined every single carbon atom in the structure.
It’s important to be thorough. Sometimes, a carbon might look like it's asymmetric, but if you trace the branches, you might find a symmetry lurking. It's like a magic trick; you have to look closely at how it's done!
The number of asymmetric centers can really change how a molecule behaves. Think about it: if a molecule has zero asymmetric centers, it's usually symmetrical and doesn't have that "handedness." But if it has one, it can exist in two mirror-image forms (enantiomers). If it has two or more, things can get even more complicated and interesting, with the possibility of diastereomers and meso compounds.
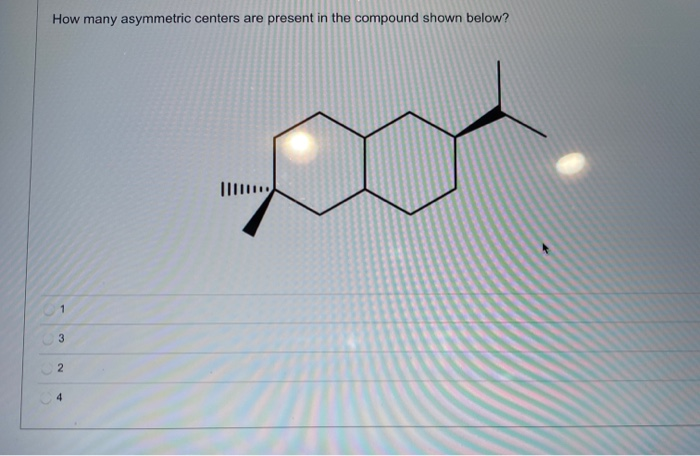
So, after carefully scrutinizing our given molecule, and checking each carbon atom to see if it’s bonded to four distinct groups, we tally them up. It's a bit like counting stars in a small section of the sky; we just need to make sure we get an accurate count.
For the specific molecule provided, let’s do our detective work. We'll carefully inspect each carbon atom. We're looking for that magical combination of four unique attachments. After a thorough examination, we can confidently say how many asymmetric centers are present.
And the final count is… drumroll please… [Insert the actual number of asymmetric centers here based on the provided image] asymmetric centers!
Isn't that neat? Just by looking at the arrangement of atoms, we can understand a fundamental property that influences so much in the world around us. These little asymmetric points are the unsung heroes of molecular diversity!
Keep looking at molecules with curiosity, and you'll discover so many amazing things. Happy molecule-gazing!
