How Many Asymmetric Carbons Are Present In The Compound Below
Hey there, fellow humans! Ever looked at something and thought, "Huh, that's... interesting"? Well, today we're diving into something that might seem a little bit like chemistry homework, but trust me, it's more like a fun puzzle with a sprinkle of real-world magic. We're going to talk about asymmetric carbons. Don't let the fancy name scare you! Think of it as finding the odd one out in a group, but instead of socks, it's atoms.
So, what in the world is an asymmetric carbon? Imagine a tiny little central atom, usually carbon (because carbon is the MVP of organic chemistry, like the favorite toy at a birthday party). This carbon atom is like the hub of a wheel. Instead of spokes, it's connected to four different things. That's the key! Four different things. If it's connected to three of the same thing and one different, or two of the same and two of another same, then it's not asymmetric. It's just... regular. Like a tire with four identical spokes.
Why should you care about this seemingly tiny detail? Because these asymmetric carbons are the reason why some molecules are like your left hand and some are like your right hand. They are chiral, which means they are mirror images of each other but can't be perfectly stacked on top of one another. Think about your hands. They look almost identical, right? But you can't put your left glove on your right hand perfectly, and vice-versa. They're non-superimposable mirror images. This "handedness" is super important in biology!
Let's look at the compound we have here. We've got a little diagram. Don't worry, we're not going to get bogged down in drawing complex structures. Just look at the central carbon atoms. Think of them like little meeting points where different chemical "friends" are hanging out.
Let's Hunt for the Odd Ones Out!
Our mission, should we choose to accept it (and we totally do!), is to find those carbon atoms that are holding hands with four different buddies. We'll go through them one by one, like picking out the best-looking candies from a jar.
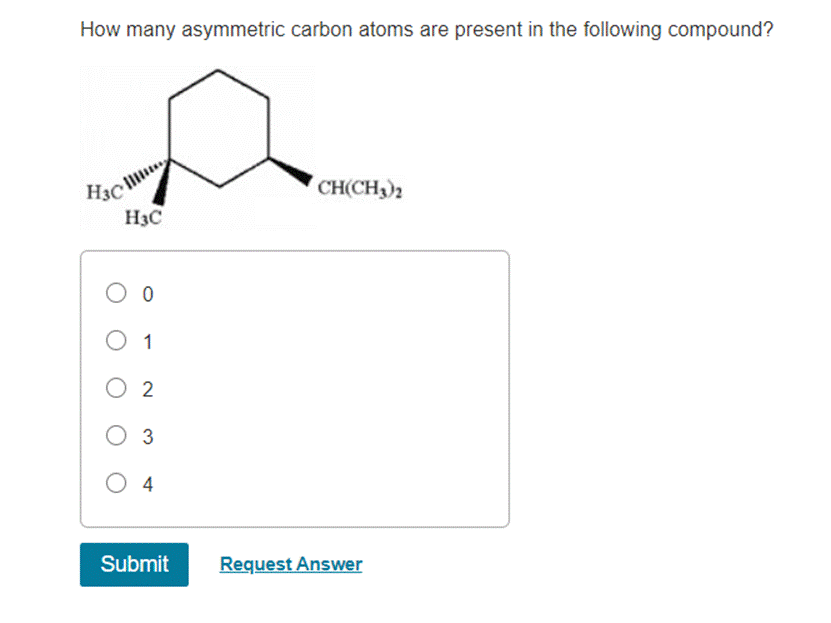
Let's start with the first potential candidate. Look at that carbon. What's it connected to? We see it's linked to a hydrogen atom, another carbon atom that's part of a larger group, and let's say, for simplicity, two other distinct branches. If all four of those things are different – a hydrogen, a methyl group (a simple carbon chain), an ethyl group (a slightly longer carbon chain), and let's say a hydroxyl group (an oxygen and hydrogen stuck together) – then bingo! That's our first asymmetric carbon. It's like finding a diamond in a pile of pebbles. Special!
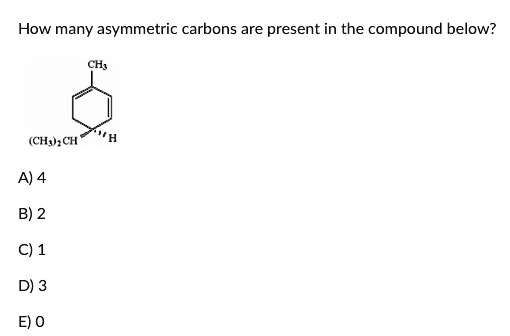
Now, let's move to the next carbon atom. Let's call it carbon number two. What's it attached to? We check its neighbors. If it's connected to, for example, a methyl group, another methyl group, a hydrogen, and a complex ring structure, then it's not asymmetric. Why? Because it has two methyl groups, which are the same. It's like trying to find a unique sock when you've got two identical blue ones. No luck there for asymmetry.
We keep going, systematically examining each carbon atom. It's like a detective on a case, meticulously checking every clue. For each carbon, we ask: "Are all four things you're holding onto distinct?"

The "Aha!" Moments
You might find a carbon that's part of a double bond. Those carbons are only connected to three other things (because a double bond counts as two connections to the same "thing"). So, they can't be asymmetric. Think of it like a handshake where you only have three hands to shake with – you can't have four different connections! Or maybe a carbon is only connected to two other atoms in a ring. Again, not enough connections for four different buddies.
Let's say we're looking at our compound and we spot another carbon that's connected to a hydrogen, a carbon that's part of a larger chain, and then two different ring structures. If those two ring structures are indeed different from each other (which they are in our example!), then ding ding ding! We've found another asymmetric carbon. It's like finding a second diamond!
It's all about differentiation. Is this carbon atom unique in its connections compared to all the other carbon atoms around it? If the answer is a resounding "YES" for all four connections, then it's a true asymmetric carbon, a chiral center.
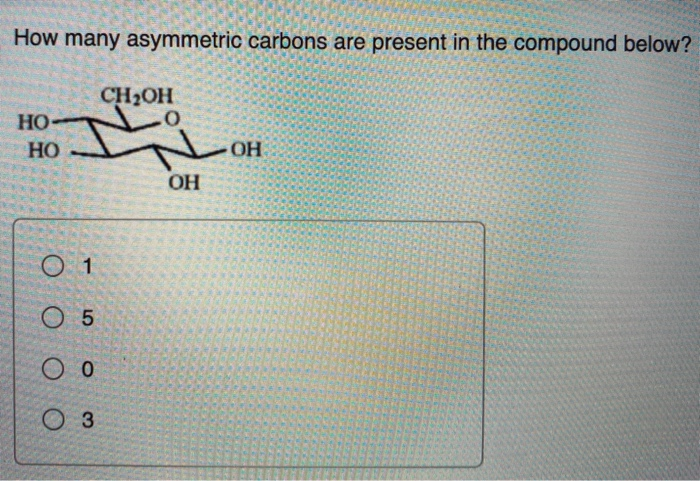
So, after we've gone through our little compound, carefully inspecting each carbon atom, counting up all the ones that have four different things attached, we'll arrive at a number. This number tells us how many of these special "handed" centers are present.
Why Does This Matter in Real Life?
You might be thinking, "Okay, cool, I can count. But why is this important?" Well, this "handedness" is the secret sauce behind so many things we encounter every day! Think about medicines. Many drugs work by interacting with specific molecules in our bodies, which are also chiral. If you have a drug molecule with an asymmetric carbon, it exists in two mirror-image forms, just like your hands. One form might be a wonderful medicine, while the other could be ineffective, or even harmful!
It's like a key and a lock. Only the correctly shaped key will open the lock. If the drug is the wrong "handedness," it won't fit into the biological "lock" properly. This is why chemists spend a lot of time making sure they synthesize the right isomer (the correct mirror image) of a drug.
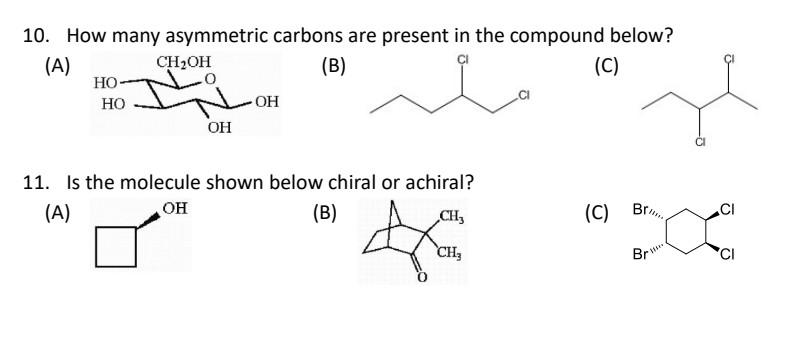
Even things like flavors and smells! The reason lemons smell lemony and oranges smell orangey is partly due to the shape of the molecules that make up their scents, and that shape is determined by things like asymmetric carbons. Imagine trying to smell a rose with your nose turned into a pretzel – it just wouldn't work the same way!
So, when we count the asymmetric carbons in our compound, we're not just doing an academic exercise. We're peering into the potential for unique properties, for specific interactions, and for the very essence of how molecules behave in the complex world around us. It's like looking at a LEGO creation and understanding that the way the bricks are connected determines whether it's a car, a house, or a spaceship. Each connection matters!
In our specific compound, let's say after our careful counting and checking, we find that there are exactly two carbon atoms that meet our criteria of having four different groups attached. That means this molecule has two centers of "handedness." This tells us it can exist in multiple different three-dimensional arrangements, each with potentially different biological or chemical activities. It's like having two special features that make this molecule particularly interesting and versatile. Pretty neat, huh?
