How Does Naoh Affect The Solubility Of Benzoic Acid Why

Hey there, science curious folks! Ever wonder what happens when you mix some bubbly stuff with a common acid? Today, we're diving into a little kitchen chemistry adventure. We're talking about sodium hydroxide, or NaOH for short, and its wacky relationship with benzoic acid. Sounds fancy, right? But trust me, it's way more fun than it sounds.
So, what's the deal? Basically, NaOH is a super strong base. Think of it as the ultimate party guest that loves to neutralize things. Benzoic acid, on the other hand, is… well, an acid. It's found in things like cranberries and cinnamon. Kinda yummy, kinda scientific!
The Solubility Shake-Up
Now, here's where the magic happens. Benzoic acid isn't exactly a water-loving kind of molecule. It's more of a "chill by itself" kind of guy. If you try to dissolve pure benzoic acid in plain old water, it's like trying to get a cat to wear a tiny hat. Not gonna happen easily. It just doesn't dissolve very well. Bummer.
But when you introduce our friendly neighborhood NaOH, things get wild. The water suddenly becomes a lot more inviting for benzoic acid. It's like NaOH throws a huge party and invites everyone, including the shy benzoic acid. And guess what? Benzoic acid loves the party. It dissolves! Poof!
Why the Big Change? It's All About the Ions!
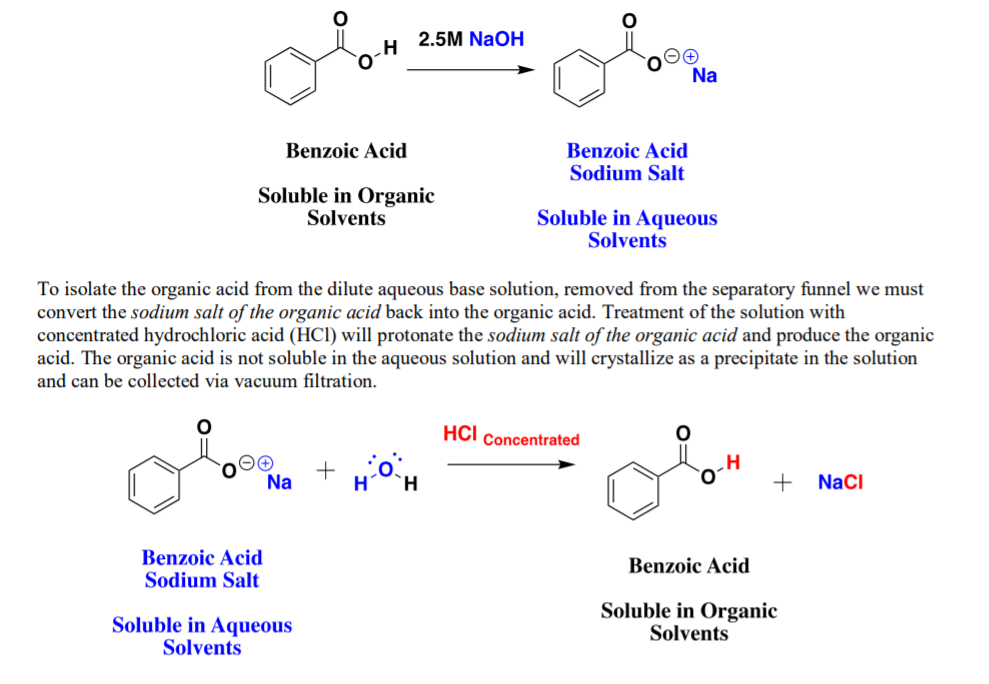
So, why does this happen? It all boils down to some seriously cool chemistry. NaOH is an ionic compound. That means it's made of charged particles, like tiny little magnets. When you dissolve NaOH in water, it breaks apart into its happy little ions: a sodium ion (Na+) and a hydroxide ion (OH-).
Now, benzoic acid has a special part called a carboxyl group. This group has a hydrogen atom attached to it. Acids, as you might know, are all about giving away hydrogen ions. Benzoic acid is no different. It can donate its hydrogen ion, leaving behind a negatively charged benzoate ion.

Here's the kicker: The hydroxide ions (OH-) from NaOH are like super magnets for those hydrogen ions that benzoic acid wants to ditch. When they meet, they team up to form water (H2O). This is a classic acid-base neutralization reaction. It's like a chemical high-five!
The Secret Ingredient: A New Form!
But the real reason benzoic acid suddenly becomes so soluble is because it's transformed! When the benzoic acid loses its hydrogen ion and the hydroxide ion from NaOH swoops in, they form a new, totally different molecule. This new molecule is called sodium benzoate.
And guess what? Sodium benzoate is a champ at dissolving in water. It's like it went from being a lone wolf to joining a popular club. It's now a salt, and most salts love water. They're all about getting hydrated and mingling.

So, instead of the original benzoic acid molecule floating around awkwardly, you now have these happy, water-loving sodium benzoate molecules chilling in the solution. They've traded their shy persona for a bubbly, dissolved existence.
It's Like Giving Your Acid a Makeover!
Think of it this way: Benzoic acid, in its original form, is like a fancy, slightly hydrophobic guest at a pool party. It's just not feeling the water. But add NaOH, and you're essentially giving benzoic acid a tiny, chemical makeover. You're turning it into sodium benzoate, its more social, water-loving alter ego.
This is why the solubility changes so dramatically. It's not just about the acid being *near the base. It's about the acid becoming something else entirely. A brand new, water-friendly compound.
Quirky Facts and Fun Tidbits
Did you know that this whole process is super important in a bunch of real-world applications? Sodium benzoate, our newly minted soluble star, is a common food preservative. It helps keep things like soft drinks, jams, and pickles from going bad too quickly. So, next time you're enjoying a fizzy drink, you can thank NaOH and benzoic acid for its shelf life!

It's also used in some medicines. Pretty neat, huh? This seemingly simple interaction has a big impact. It’s a testament to how fundamental chemistry can be.
And get this, the reaction is pretty quick! You don't have to wait ages to see the benzoic acid dissolve once you add the NaOH. It's like a science magic trick happening right before your eyes. Abracadabra, dissolved!
Why is This Fun to Talk About?
Because it’s like uncovering a secret superpower! Benzoic acid, this ordinary-looking molecule, can be transformed into something completely different, something that behaves in a totally new way. It’s a reminder that even everyday substances have hidden talents.
Plus, the idea of a base "neutralizing" an acid sounds so dramatic, doesn't it? Like a chemical showdown where everything ends up in a peaceful, dissolved state. It’s the ultimate chemical reconciliation!
It also shows how a little bit of a different chemical can completely change the rules of the game. NaOH isn't just adding something to the water; it's actively changing the benzoic acid itself.
The Takeaway: More Than Just Mixing
So, the next time you hear about NaOH and benzoic acid, you'll know the secret. It's not just about mixing; it's about transformation. The strong base NaOH reacts with the acid benzoic acid, creating the water-soluble salt, sodium benzoate. This new form of benzoic acid is super happy to hang out in water, making it much more soluble.
It’s a small chemical reaction with a big impact, from preserving our food to making chemistry class a little more interesting. So go forth and impress your friends with your newfound knowledge of benzoic acid's solubility secrets. You're basically a chemical whisperer now! You're welcome.
