How Do The Values Of Successive Ionization Energies Compare

Hey, you! Yeah, you, the one with the curious mind. Let's talk atoms. Not in a boring textbook way, okay? More like, we're dissecting a tiny, energetic universe. And today's special guest? Successive Ionization Energies. Sounds fancy, right? Don't worry, it's actually super cool and a little bit wild.
So, what's the deal? Think of an atom like a little family. It's got a nucleus, the bossy parent in the middle, and electrons, the energetic kids whizzing around. Ionization energy is basically the energy you need to snag one of those electrons. Like, "Gimme that electron, you little rebel!"
The First Kid's Easy (Usually)
The first ionization energy is the energy to pull off the outermost electron. It's like the first kid to leave home. They might miss the Wi-Fi, but they're ready to spread their wings. For most elements, this is the easiest electron to grab.
Imagine an atom is a house party. The first electron? That's the one chilling closest to the door, easy to nudge out so a new guest can come in. Not a big fuss.
The Second Kid? Uh Oh.
Now, here's where it gets juicy. When you want to take away a second electron – the second ionization energy – things get… more interesting. That's because the remaining electrons are now feeling the pull of the nucleus a lot more. The nucleus is like, "Okay, you little punks, you're all mine now!"
So, the second ionization energy is always going to be higher than the first. Always. It's like the remaining kids in the house party are now huddled together, giving you the stink eye. You gotta work harder to get them to leave.

The Jumps! The Huge Leaps!
And then… BAM! Sometimes, the ionization energy takes a massive leap. We're talking a giant, unbelievable jump. This is the fun part! It's like, you're trying to get kids to leave the party, and one of them is the absolute life of the party, practically glued to the DJ. You need way, way, way more energy to get them to budge.
Why does this happen? It all comes down to electron shells. Electrons hang out in different energy levels, or "shells." The outermost shells are easier to reach. But when you start pulling off electrons from a more stable, inner shell – one that's happy and complete – it's like trying to steal the family jewels. The atom really doesn't want you to.
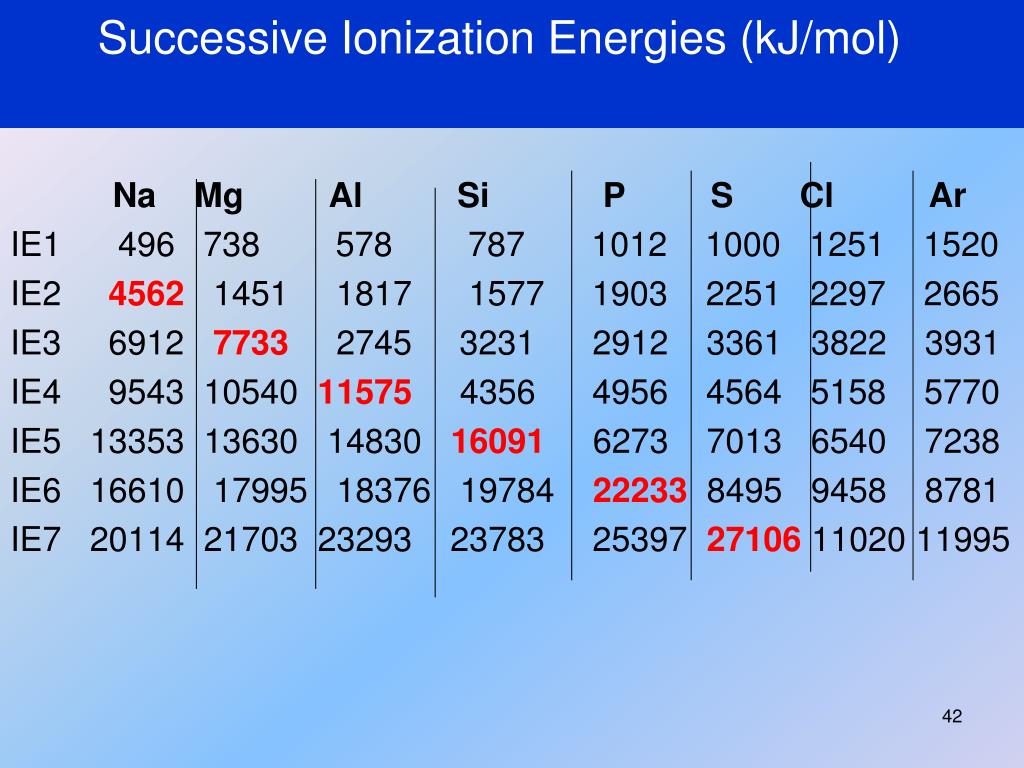
A Quirky Example: Sodium
Let's talk about sodium. It's that common stuff in table salt. Sodium has 11 electrons. The first electron? Pretty easy to take off. It's hanging out all by itself in the outer shell, basically waving a white flag.

But the second ionization energy for sodium? Whoa, Nelly! It's like asking for a kidney. Suddenly, you're trying to pull an electron from a really stable, "full" inner shell. The energy required is gargantuan! The difference between the first and second ionization energy for sodium is HUGE. It's the difference between asking for a spare pen and demanding their firstborn.
This massive jump tells us something important: Sodium really likes to lose just one electron. It's its happy place. That's why sodium is so reactive, always looking to shed that one electron to achieve stability.
Why Is This So Darn Fun?
Because it's like a detective story! Each element has its own unique pattern of ionization energies. By looking at these numbers, scientists can figure out how many electrons an element is likely to lose or gain. It's a peek into the atom's personality.
Think of it like this: You're trying to figure out how stubborn your friends are. Some will leave a party if you just text them. Others need a full-on intervention. Ionization energies are the atomic version of that.
The "Noble Gas" Moment
And don't even get me started on the noble gases! Helium, neon, argon… these guys are the ultimate introverts. They have completely full outer electron shells. They are so stable. Their ionization energies are sky-high. It's like trying to convince a cat to enjoy a bath.
The first ionization energy for a noble gas is already pretty big. But there's no second big jump because there's no easily accessible inner shell to mess with. They are content in their electron-filled bubbles. They've already won the electron game.
This stability is why noble gases are, well, noble. They don't readily react with anything. They're the lone wolves of the periodic table. Pretty cool, right?
It's All About Stability
Ultimately, these ionization energy jumps are all about achieving a stable electron configuration. Atoms, like us, want to be comfortable. They want to be in a state of low energy. And for most elements, losing or gaining specific numbers of electrons is the easiest way to get there.
The values of successive ionization energies tell us exactly how much effort it takes to mess with an atom's electron arrangement. It's a quantitative measure of how tightly those electrons are held.
So, the next time you see a chemical reaction happening, remember the dance of the electrons and the energy required to make them move. It's a little bit of magic, a little bit of physics, and a whole lot of fun. You're basically looking at the atomic motivation behind everything!
It’s like unlocking secret codes within elements. Each jump in ionization energy is a clue, a whisper about the atom’s inner workings. And that, my friend, is pretty darn exciting.
