Highest Oxidation State Of Transition Elements
Hey there, science curious folks! Ever look at a shiny piece of metal and wonder what makes it tick? Today, we're going to dive into something a little bit technical, but I promise, we'll keep it as light and breezy as a summer picnic. We're talking about the highest oxidation state of transition elements. Now, before your eyes glaze over, let's break it down with some everyday analogies.
Imagine your favorite superhero. They have their regular powers, right? But sometimes, they can tap into a supercharged version of those powers, a kind of "ultimate form" that lets them do even crazier, more amazing things. That's kind of what we're talking about with transition elements and their highest oxidation states. These are elements, like the ones in your trusty stainless steel fork or the colorful pigments in some paints, that are really good at changing their "mood" or "level of electron engagement."
What's an "Oxidation State," Anyway?
Think of oxidation state as a way to describe how many electrons an atom has decided to share, steal, or generally lend out when it's hanging out with other atoms. It's like a chemical handshake. Some atoms are really generous and like to share a lot, while others are a bit more possessive. Transition elements are the chameleons of the periodic table – they can change their "handshake style" quite a bit.
The highest oxidation state is simply the maximum number of electrons an atom of a transition element can "give away" or "lose" in a chemical reaction. It’s their absolute best effort at being "electron-donating."
Why Should You Care About This Superpower?
You might be thinking, "Okay, cool, atoms are playing electron-share-or-don't-share games. So what?" Well, this seemingly small detail has huge implications for the world around us!
Let's take a little story. Imagine you're baking. You have different ingredients, right? Flour, sugar, eggs. Each ingredient contributes something unique to the final cake. Now, the highest oxidation state of a transition element is like a particularly potent ingredient that can radically change the outcome of the "chemical bake."
It influences how these elements react, what kinds of compounds they form, and ultimately, the properties of the materials we use every single day. It's the secret sauce behind many amazing technologies and everyday objects.
The "Team Players" of the Periodic Table
Transition elements are found in the middle section of the periodic table, a bit like the exciting midfield players in a soccer team. They have a special trick up their sleeve: they can use electrons from two different energy shells. This gives them a lot of flexibility, allowing them to achieve these high oxidation states.
Think of it like having a toolbox with two different drawers of tools. You can pick and choose from both to build something truly amazing. Other elements might only have one drawer, limiting what they can construct.
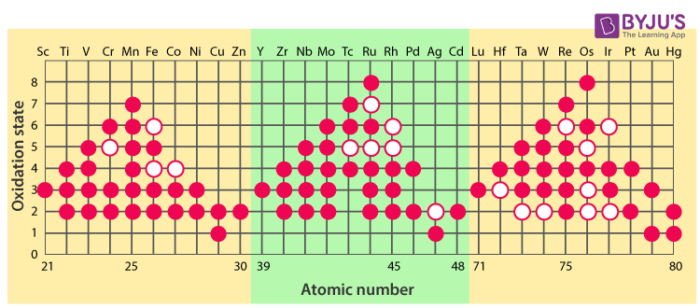
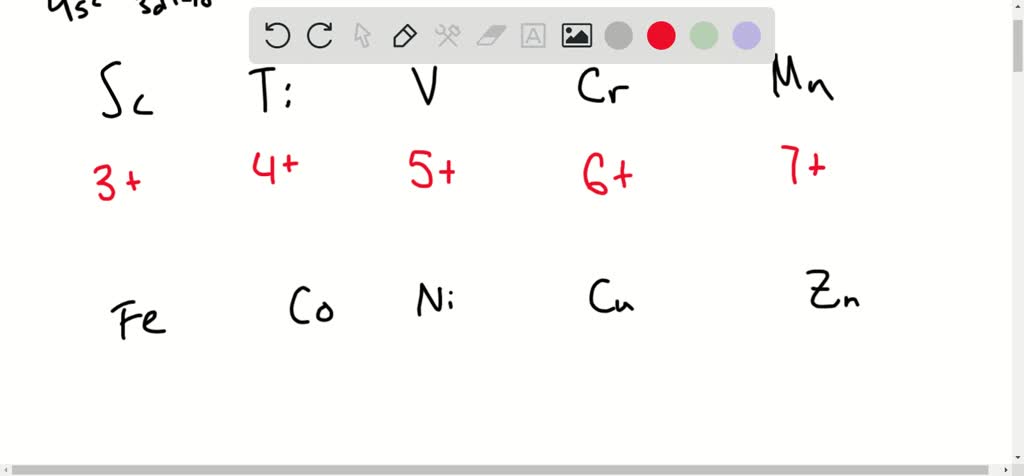
A Tale of Two Elements: Vanadium and Manganese
Let's meet a couple of these electron-sharing champions. First up, we have Vanadium. This element is known for its incredible ability to reach a +5 oxidation state. What does that mean in plain English? It can contribute a whopping five electrons to a chemical bond!

You might encounter vanadium in some super strong alloys used in aerospace, or even in the glass of your windows to tint them and block UV rays. Imagine the tiny vanadium atoms working together, each contributing their maximum electron effort to make that glass tough and protective. It's like a synchronized swimming routine, but with electrons!
Now, let's talk about Manganese. This one is a real powerhouse, capable of reaching an astonishing +7 oxidation state! That's like our superhero achieving their absolute maximum power level. Manganese in its +7 state is a very strong oxidizing agent. Think of it as a super-enthusiastic cleaner that can scrub away unwanted things. It’s used in things like batteries to help them store and release energy efficiently. Without manganese’s high oxidation state, your phone might die a lot faster!
Imagine you're trying to clean a really stubborn stain. You need a powerful cleaning agent, right? That's what manganese in its high oxidation state can be – a chemical powerhouse that drives reactions forward.

Why Does This Matter to Your Wallet (and Your World)?
This isn't just for chemists in lab coats! The high oxidation states of transition elements are behind some really important stuff:
- Catalysts: Many industrial processes, like making plastics or fuels, rely on transition metals acting as catalysts. Catalysts speed up reactions without being used up themselves. The ability to exist in multiple oxidation states is key to their catalytic function. Think of them as the helpful guides on a hiking trail, showing the way for other molecules to react faster and more efficiently.
- Colors and Pigments: Ever admired the vibrant colors of a painting or the deep blue of some gemstones? Many of these stunning colors come from transition metal compounds, and their colors are often determined by the element's oxidation state. For instance, the intense colors of some ruby reds and sapphire blues are due to the way chromium and iron ions, in specific oxidation states, interact with light. It's like they're wearing different colored outfits depending on their "electron mood"!
- Medicines: Some important medicines, like certain chemotherapy drugs, use transition metals. Their ability to interact with biological molecules is often linked to their oxidation states. It's a delicate chemical dance that can target and fight diseases.
- Batteries: As mentioned with manganese, the ability of transition metals to switch oxidation states is crucial for storing and releasing energy in batteries. Without these electron-shifters, our phones, laptops, and electric cars wouldn't work.
So, the next time you see a bright red stop sign, charge your phone, or marvel at a piece of stainless steel cutlery, give a little nod to the transition elements and their amazing ability to play with their electrons. Their highest oxidation states are like their secret superpowers, enabling them to build, protect, color, and energize our world in ways we often take for granted.
It’s a reminder that even the smallest, most fundamental properties of matter can lead to some of the most significant advancements and everyday conveniences we enjoy. Science is all around us, and sometimes, the most exciting stories are hidden in the very building blocks of the universe!
