Heat Of Hydrogenation And Stability Of Alkenes

Ever felt like you just needed a good, solid hug? Like, a really firm one? Well, guess what? Molecules get that way too! Today, we're diving into the fascinating world of alkenes and how they react with hydrogen, a process we affectionately call "hydrogenation." Think of it as giving these sometimes-flirty, double-bonded molecules a nice, stable, single-bonded home.
Alkenes, bless their hearts, are those cool kids of the hydrocarbon world. They've got this double bond – like a little party going on, always ready to shake things up. It's a bit like that friend who's always up for spontaneous road trips or trying that questionable street food. Fun, sure, but maybe not the most… settled lifestyle.
Hydrogenation is basically the antidote to this restlessness. We take our alkene, give it a good dose of hydrogen gas (H₂), and with the help of a trusty catalyst (think of it as a really good matchmaker), that double bond breaks apart. Poof! Suddenly, we have a much happier, more stable alkane. It’s like that friend finally settling down, buying a sensible car, and maybe even getting a cat. Admirable, really.
Now, the cool part – and where the "heat" comes in – is that this whole process of an alkene becoming an alkane with hydrogen is like a little exothermic party. Energy is released! It’s like when you finally get that perfectly brewed cup of coffee on a chilly morning – a little burst of warmth and satisfaction. Or when you successfully assemble IKEA furniture without losing your sanity – that feeling of accomplishment often comes with a subtle, internal glow, right?
This "heat of hydrogenation," as the fancy folks call it, isn't just some abstract scientific concept. It’s a way for us to measure just how much that double bond was, shall we say, itching for a change. The more heat released, the more unstable the original alkene was. Think of it like this: a really, really dramatic breakup will often involve a lot more thrown crockery and loud sobbing than a mild disagreement over who left the toilet seat up. The energy released is proportional to the drama, or in our case, the instability.
The Stability Spectrum: From "Meh" to "Finally!"
So, how do we know which alkene is more of a drama queen and which is more of a laid-back chill dude? It all comes down to how many other groups are hanging out with that double bond. These groups can either stabilize or destabilize the double bond.

Imagine the double bond as a slightly wobbly Jenga tower. If you’ve got a bunch of extra little bits and bobs sticking out from around the tower (these are our alkyl groups, by the way), it can actually make the whole structure a bit more stable. It’s like adding little counterweights. Weird, right? More stuff can mean more stability in this particular molecular dance.
Let's break it down:
Monosubstituted Alkenes: The "Just Dipped My Toe In" Guys
These guys have only one alkyl group attached to one of the carbons in the double bond. They're like that person who’s always talking about wanting to travel but never actually books a flight. They've got a bit of potential for adventure, but they're not fully committed. A little bit of heat is released during hydrogenation, not a ton. They're not in a desperate rush to become an alkane.
Think of it like this: you're considering getting bangs. You're thinking about it, you've maybe watched a few YouTube tutorials. There's a small amount of internal energy spent contemplating the change, but it's not a full-blown, life-altering decision yet. The heat released is like the mild anxiety you feel before a slightly daring haircut. Not a bonfire, more like a candle.

Disubstituted Alkenes: The "Tentatively Committed" Crew
These alkenes have two alkyl groups attached. Now we're getting somewhere! They've got a bit more going on. They're like the person who's booked a weekend trip but hasn't packed yet. They're definitely leaning towards change, but they haven't fully committed to the destination. More heat is released during hydrogenation compared to monosubstituted alkenes. They're a bit more eager for that stable, single-bond life.
This is like deciding to redecorate your living room. You've picked out the paint color and bought a new rug. There's a bit more invested energy, a clearer intention. The heat released is like the satisfaction you get from ticking off a couple of big items on your to-do list. It’s a bit warmer, a bit more substantial than just contemplating the idea.
There are two ways to be disubstituted: cis and trans. Think of it like passengers on a bus. In cis, the passengers (alkyl groups) are on the same side of the aisle (the double bond). This can be a bit cramped, a bit awkward, like trying to cram two extra suitcases into the back seat of your car. This cis arrangement is actually less stable than the trans arrangement. All that close proximity leads to some uncomfortable bumping and jostling, hence more energy released when it finally gets to chill out as an alkane.
In trans, the passengers are on opposite sides of the aisle. Much more spacious and polite! It’s like having your luggage neatly arranged in the trunk. This arrangement is more stable. Less internal friction means less heat released when it finally becomes an alkane. It's already pretty comfortable, so the transition isn't as dramatic.
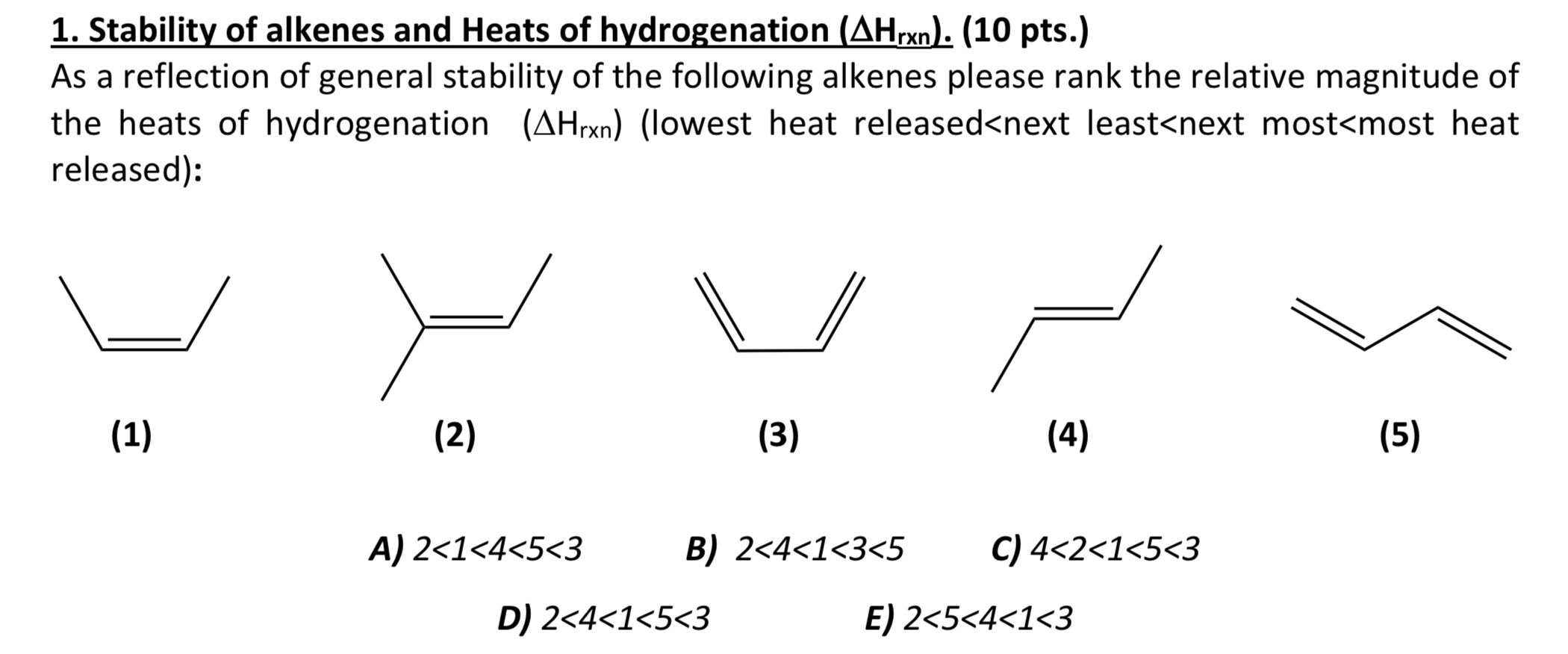
Trisubstituted Alkenes: The "All In!" Enthusiasts
These are the ones with three alkyl groups. They're practically already in the moving truck, just waiting for the keys to their new, stable house. They’re like the person who’s bought the plane tickets, booked the hotel, and is currently scrolling through Instagram for "what to pack" lists. They are so ready. They release even more heat during hydrogenation because they were really keen to ditch that double bond.
This is like starting the actual renovations. You've ripped up the carpet, you're ready for new paint, maybe even knocking down a wall (metaphorically, of course, unless you're really committed). There's significant energy invested, a clear trajectory. The heat released is like the sigh of relief and satisfaction when a major milestone in a big project is completed. It's a warm, comforting glow.
Tetrasubstituted Alkenes: The "Already Moved In" Champions
And then, the ultimate in alkene stability (before they become alkanes, anyway) – tetrasubstituted alkenes! These guys have four alkyl groups. They're basically already in their stable, alkane-like mindset. They release the least amount of heat during hydrogenation because they were already pretty content. They're like the person who's just arrived at their new home, unpacked their toothbrush, and is already planning the housewarming party. They were barely holding onto that double bond life.
This is like having the new furniture in place and the paint drying. It's not just a plan anymore; it's reality. The energy released during hydrogenation is like the quiet hum of contentment that settles over a completed project. It's a gentle warmth, a sign of things being just right.
Why Does This Even Matter? (Besides Making Your Brain Feel Smart)
So, we’ve established that more alkyl groups = more stability = less heat released during hydrogenation. Why should we care about this little bit of chemical trivia?
Well, it’s like understanding why some things in life are a breeze and others are a struggle. This concept helps chemists predict how different molecules will behave. It’s crucial for:
- Drug Design: Sometimes, a slight tweak in the shape or stability of a molecule can make all the difference between a medicine that works and one that doesn't, or even one that causes nasty side effects. It’s like tuning a guitar; a tiny adjustment can make the whole instrument sound better.
- Materials Science: Understanding the stability of molecules helps us create new plastics, fuels, and other materials with specific properties. Think about how some plastics are super brittle and others are really flexible. That difference often comes down to the molecular structure and how stable those bonds are.
- Understanding Natural Processes: Life itself is full of chemical reactions. Understanding the energy involved helps us decode how our bodies work and how nature operates. It’s like understanding the mechanics of a car to appreciate how it moves.
The heat of hydrogenation is essentially a quantifiable measure of the “energy stored” in that double bond. The more energy stored, the more unstable the system, and the more energy is released when it finds a more stable configuration. It’s like a compressed spring – the more you compress it, the more energy it has to release when it snaps back.
So, next time you're enjoying a nice, stable meal (or just a nice, stable couch potato session), you can thank the humble alkene and its journey towards a more settled existence. And remember, a little bit of heat released is just proof that sometimes, change – even for a molecule – can be a very good thing. It’s like finally getting a good night’s sleep after a week of tossing and turning; that feeling of relief is palpable, and in the molecular world, it’s measured in delicious, energetic warmth.
