Ground State Electron Configuration For Vanadium

Hey there! So, you wanna chat about vanadium? Awesome! It’s one of those elements that’s kinda… well, sneaky. You look at it, and you’re like, “Okay, element number 23, Vanadium. Easy peasy.” But then you dive into its electron configuration, and it’s like, “Whoa there, what’s going on here?” It’s not as straightforward as you might think, and that’s what makes it, dare I say, super interesting.
Think of an atom like a tiny, tiny apartment building. The nucleus is the lobby, right? And then you’ve got all these floors, called energy levels. On each floor, there are different types of rooms, or orbitals. We’ve got the s-rooms, the p-rooms, the d-rooms, and even f-rooms (though we’re not going that deep for vanadium, phew!). Electrons, these little energetic dudes, they love to fill up these rooms, starting from the lowest, cheapest floors, obviously. They’re not exactly ballin’ on a budget, you know?
Now, for Vanadium, element number 23. That means it’s got 23 protons in its nucleus, and in a neutral atom, it’s also got 23 electrons. These electrons are going to zip and zoom around, finding their perfect little orbital homes. Most elements are pretty chill about it, filling up in a predictable order. But vanadium? It’s got a bit of a quirk.
So, if we were just following the “lowest energy first” rule, you’d probably guess something like this: 1s², 2s², 2p⁶, 3s², 3p⁶, 4s², 3d³. Makes sense, right? You’ve filled up all the lower floors, and then you’ve got three little electrons left to hang out in the 3d orbitals. It’s like, “Okay, done with the main apartments, let’s find a spot in the slightly fancier d-suites.”
But here’s the plot twist! This is where vanadium gets its bad rap for being a bit of a rebel. The actual ground state electron configuration for vanadium is 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d³. Wait a minute… that’s exactly what we just said, isn’t it? Yes, it is! But the reason it ends up that way is a little more nuanced than just filling up in order. Sometimes, things get a little… fuzzy.
Okay, let me backtrack slightly. It’s not that the rule is broken, it’s more that we need to consider the stability of the orbitals. Imagine you have a bunch of different room types. Some are super spacious and comfy, others are a bit cramped. Electrons, being smart little things, want to be in the most stable, lowest energy configuration possible. And sometimes, a slightly different arrangement can be surprisingly more stable. It’s like choosing between a slightly longer commute to a super nice apartment or a shorter commute to a decent one. Decisions, decisions!
![[ANSWERED] (1) What is the complete ground state electron configuration](https://media.kunduz.com/media/sug-question/raw/52557527-1659251681.6304457.jpeg?h=512)
The key here is the interplay between the 4s orbital and the 3d orbitals. The 4s orbital is technically at a lower energy level than the 3d orbitals. So, you'd think all the electrons would just pile into the 4s first, right? And for many elements, that's exactly what happens. But as you start adding more and more electrons, things get a little crowded, and the energy levels can get all jumbled up. It's like the apartment manager is trying to squeeze in as many tenants as possible, and suddenly the "cheaper" apartments aren't so cheap anymore!
So, with vanadium, what we see is that the 3d orbitals, even though they’re technically at a slightly higher energy level than the 4s, are more efficient at holding those last few electrons. It’s like they’re saying, “Hey, we might be on a slightly higher floor, but we’ve got more room to spread out, and we’re more stable in the long run.” This is especially true as you move across the transition metals. They're in this delicate balancing act, trying to figure out the most energy-efficient way to arrange their electrons.
Let’s visualize this. We have the 4s orbital, which is shaped like a sphere. It's a nice, simple room. Then we have the 3d orbitals. These are way more complex shapes, like weird little dumbbells or cloverleaves. They have more "space" for electron density in different directions, which can lead to better shielding of the nucleus and a more stable overall configuration for those particular electrons.
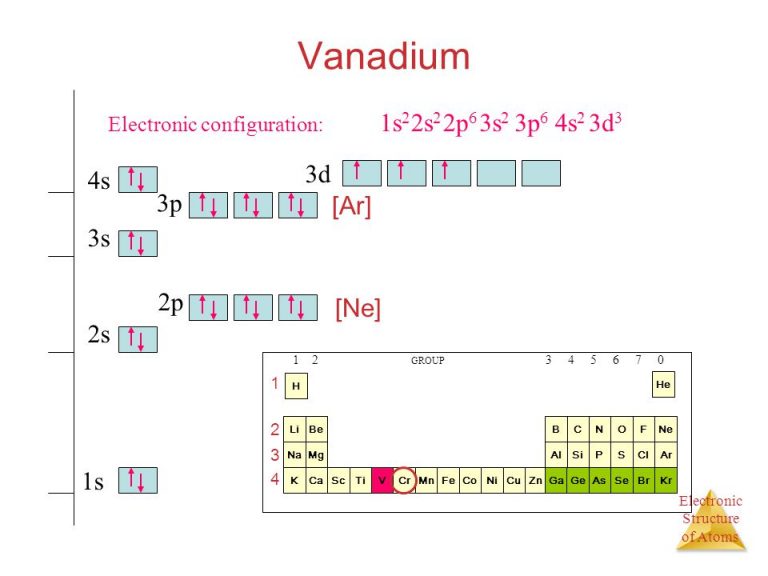
So, when vanadium has its 23 electrons, it fills up everything nicely until it gets to the point where it has electrons for the 4s and the 3d. The general trend is to fill the 4s first. That’s why we see 4s². Two electrons happily settle into the spherical 4s orbital. Think of it as the primary bedroom, nice and accessible.
Then, we have those remaining three electrons. Where do they go? They go into the 3d³ orbitals. There are five 3d orbitals in total, each capable of holding two electrons. So, these three electrons will go into three different 3d orbitals. This follows Hund's rule, by the way, which basically says that electrons will spread out into empty orbitals within a subshell before pairing up. It's like everyone wants their own bedroom before they have to share.
So, you end up with 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d³. It’s a bit of a mouthful, I know. But that 3d³ part is the key. It’s where vanadium starts to show its transitional metal character. This partial filling of the d-orbitals is what gives vanadium so many of its cool properties, like its ability to form multiple oxidation states.
Why is this stability thing such a big deal? Well, imagine you have a perfectly balanced Jenga tower. Everything is just so. Now, if you were to nudge one of the blocks slightly, it might still stand, but it’s a bit more precarious, right? Electrons want that Jenga-tower-perfect stability. Sometimes, filling a subshell almost completely or leaving it almost empty is surprisingly more stable than having a half-filled or just-started subshell. It’s a quantum mechanical dance, really.
For vanadium, the 3d³ configuration, while not a “magic” half-filled (d⁵) or completely filled (d¹⁰) shell, represents a stable arrangement given the number of electrons it has. It’s a compromise, a happy medium. It's not as stable as a full 3d¹⁰, and it's not as stable as a half-filled 3d⁵, but it's more stable than if those three electrons were arranged in a different, less symmetrical way.

Think about it like this: You’re packing for a trip. You have three souvenirs, and you have five designated spots for souvenirs in your suitcase. You wouldn't put all three in one spot, right? You'd spread them out evenly. That's what those three electrons do in the 3d orbitals. They occupy three different orbitals to maximize their separation and minimize repulsion. It’s like they’re social distancing within the atom.
And that 4s²? That’s like the essential luggage. You gotta have those. They’re fundamental to the atom’s structure and its interaction with other atoms. They’re the first things that get packed, in a way, before you even consider those slightly more specialized d-orbital souvenirs.
So, when you see that 3d³, don’t just skip over it. That’s the heart of vanadium’s electron configuration story. It's what allows it to be used in alloys for steel (making it super strong, by the way!), in catalysts, and even in batteries. These d-electrons are the workhorses, the ones involved in the chemical reactions and bonding that make vanadium so useful.
It's kind of like the difference between a perfectly uniform row of soldiers (like a completely filled or half-filled subshell, which is super stable) and a group of three friends who are all hanging out together, but each in their own comfortable spot. They're still a group, and they function as a group, but they're not rigidly packed together. Vanadium's 3d³ is that group of friends.
And this is where the term "ground state" comes in. It just means the atom is in its most stable, lowest energy arrangement. It's not all excited and jumping around to higher energy levels. It's chilling, in its most comfortable configuration. Think of it as the atom in its pajamas, not in its fancy going-out clothes.
So, next time you hear about vanadium, you can nod wisely and say, "Ah yes, the element with the 4s² 3d³ electron configuration!" You’ll be the resident electron whisperer. It’s not always a simple fill-up-the-order situation, and that’s what makes chemistry so darn fun, right? There are always little surprises and exceptions to keep us on our toes.
And honestly, for something so small and invisible, the way electrons arrange themselves has such a massive impact on the macroscopic world. It's like these tiny, invisible decisions are dictating whether your bike frame is super strong or whether that chemical reaction happens. Mind-blowing stuff if you ask me!
So yeah, vanadium. A little bit of a rebel, a little bit of a puzzle, but ultimately, a super important element whose electron configuration is a perfect example of how nature finds its own sweet spot for stability. Pretty neat, huh?
