Given The Following Single-step Reaction Draw The Curved-arrow Mechanism
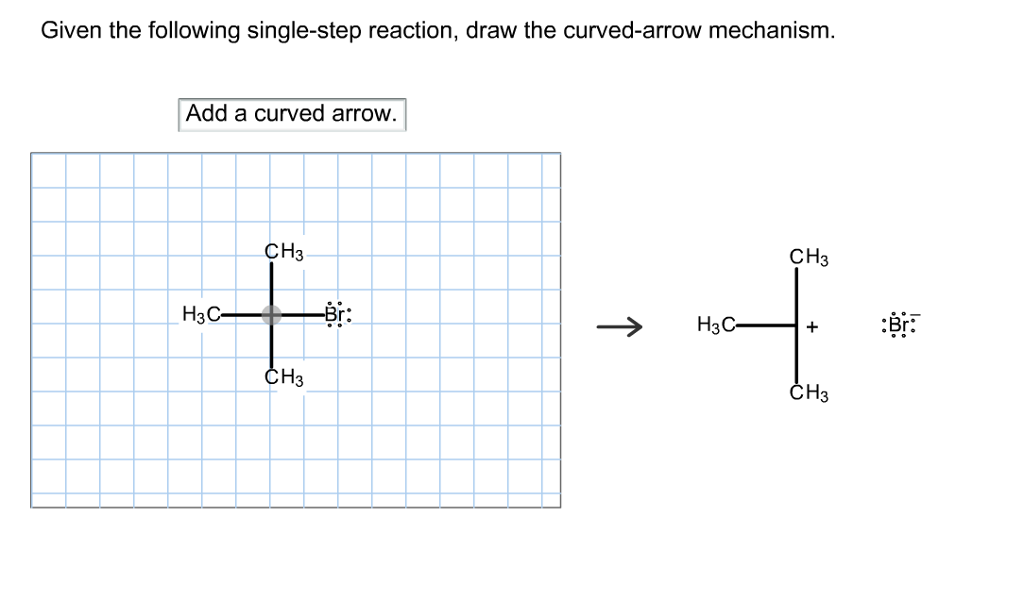
So, you’ve been handed a chemistry problem. It’s a “single-step reaction,” they say. Sounds simple enough, right? Like, one and done. A quick little dance of molecules and poof, you’ve got your product.
And then comes the dreaded instruction: “Draw The Curved-arrow Mechanism.” Cue the dramatic music. Suddenly, that simple one-step feels like a marathon. A very, very wiggly marathon.
My unpopular opinion? This whole “single-step” thing with its fancy arrow-pushing is a bit of a… well, a bit of a ruse. It’s like being told, “Just hop on one foot across this room!” while simultaneously being asked to perform a complex ballet routine mid-air.
We’re presented with this elegant, pristine equation. It looks so neat and tidy. Reactant A merges with Reactant B, and bam, Product C appears, maybe with a little Byproduct D waving goodbye. It’s the chemical equivalent of ordering a pizza and having it appear at your door, no fuss, no muss.
But then, they whip out the:
Given The Following Single-step Reaction Draw The Curved-arrow Mechanism.
And suddenly, the pizza delivery guy is asked to explain exactly how the dough rose, how the cheese melted, and how the pepperoni achieved its perfect curl. All with a series of, you guessed it, curved arrows.
These aren't your garden-variety arrows, mind you. These are magical curved arrows. They don’t just point; they flow. They represent electrons, these tiny, energetic little gremlins that are always on the move.
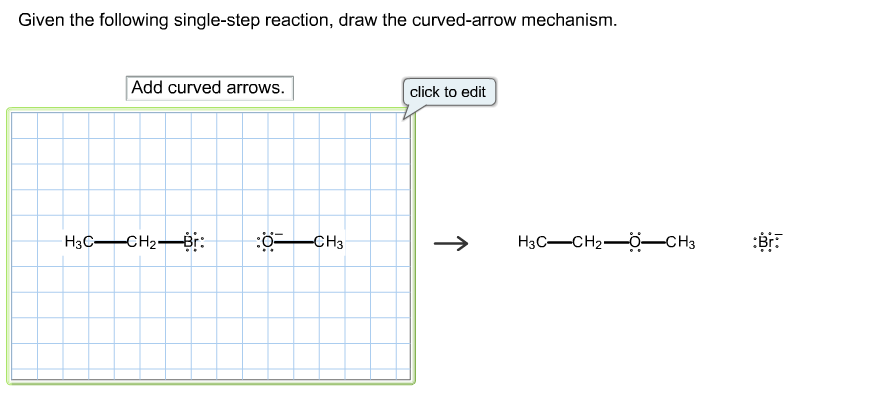
In a single-step reaction, we’re told these electrons make one glorious leap. They travel from one atom to another, or from a bond to an atom, or from an atom to a bond. It’s a grand gesture of electron sharing or electron grabbing.
Think of it like this: you’ve got two friends, Alice and Bob, who want to share a cookie. In a real-life, multi-step world, maybe Alice breaks the cookie in half, then hands a piece to Bob. Or maybe Bob asks Alice nicely, and she gives him a whole piece. There are steps, negotiations, perhaps even a little cookie-crumb drama.
But in the single-step world of curved arrows, Alice and Bob are holding the cookie, and at the exact same nanosecond, a piece of cookie just appears in Bob’s hand. No breaking, no handing, just… electron transfer. It’s efficient, I’ll give it that. Almost too efficient.
So, there you are, staring at your single-step reaction. You’ve got your starting materials, looking all innocent. You’ve got your final product, looking equally innocent. And then you’ve got the prompt, looming like a pop quiz you forgot to study for.
The first thing you do is identify your electron donors and your electron acceptors. Who’s feeling generous with their electrons? Who’s looking a bit electron-deprived? It’s like figuring out who’s bringing the snacks to the party and who’s just going to eat them.
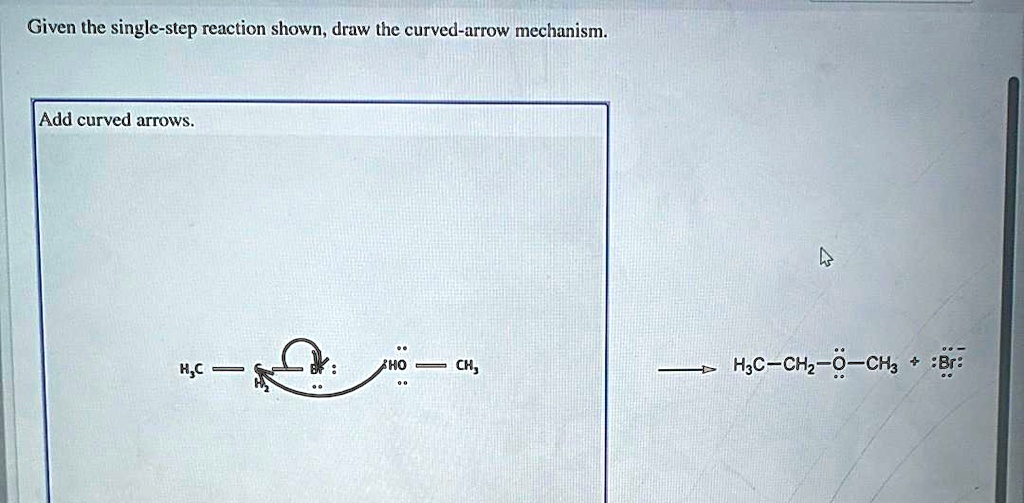
Then, the fun begins. You grab your virtual pen (or your actual pen, if you’re old school like me) and start drawing. You need to show an electron pair, usually from a lone pair or a pi bond, moving to form a new bond. Or, you show a bond breaking, with the electrons going to form a lone pair or stabilize a charge.
The curved arrow is your trusty steed. It originates from the source of the electrons. That’s crucial. It doesn’t just float around aimlessly. It has a starting point, a place of electron abundance.
And where does it go? It points to the atom that’s going to receive those electrons. This is where the magic happens. This is where a new bond might be formed, or an existing species becomes more stable.
In a single-step reaction, there’s only one of these arrow-pushing sequences to draw. Just one. One elegant, swooping gesture that explains the entire transformation. It’s like a perfectly executed pirouette.
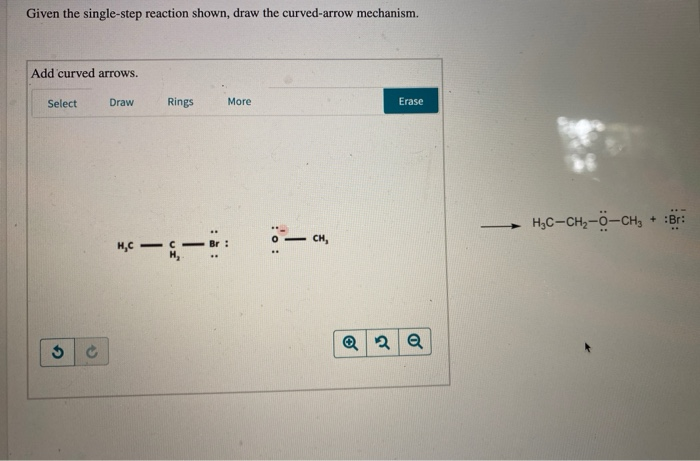
You might have a situation where a nucleophile, with its abundance of electrons (think of it as the "lover of positive things"), attacks an electrophile, which is electron-deficient (the "lover of negative things"). The curved arrow will start from the negative charge or lone pair on the nucleophile and point to the atom in the electrophile that’s hungry for electrons.
Or, you might see a bond breaking. Perhaps a leaving group is about to depart. The curved arrow would then show the electrons from the breaking bond moving onto that leaving group, stabilizing it. It’s a clean break, a dignified exit.
The key is that it all happens simultaneously. There’s no intermediate. No transient species hanging around for a bit. It's a direct transfer, a swift handover. It’s the chemical equivalent of instant messaging, not email.
Sometimes, these single steps can look surprisingly complex, even with just one arrow. You might be drawing an arrow that forms a new bond and simultaneously breaks another. It’s like juggling while doing a handstand. Impressive, but also a little terrifying to watch.
And then, there are those moments of utter self-doubt. Did I draw the arrow from the right place? Is it pointing to the correct atom? Is this really a single step, or am I just desperately trying to make it look like one?
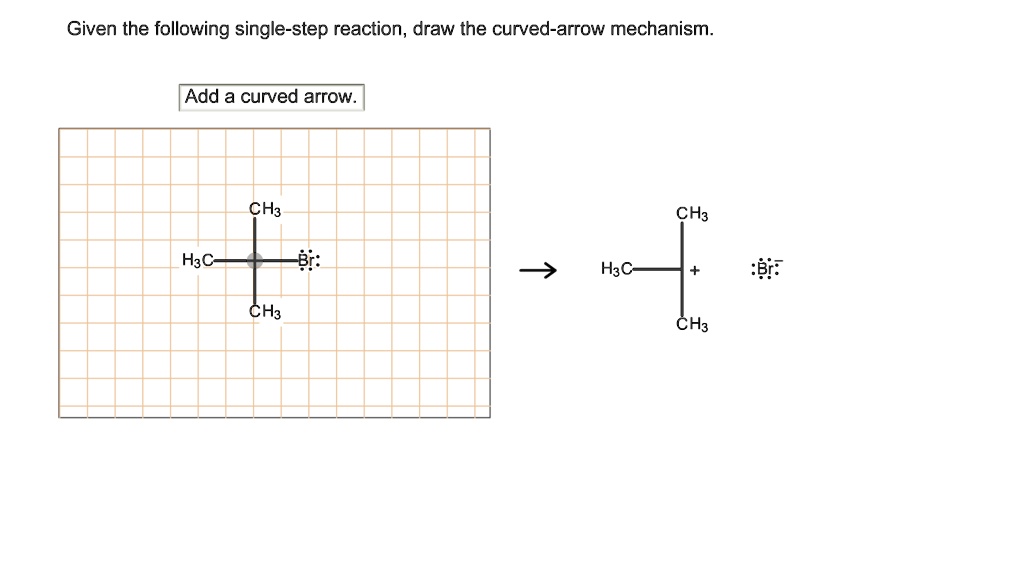
You scrutinize your drawing. You compare it to examples in your textbook, which always seem to have the most pristine, textbook-perfect reactions. Yours might look a little more… artistic. Or perhaps, a little more like a toddler scribbled on it.
But here’s the comforting part. For a single-step reaction, the curved-arrow mechanism is supposed to represent the entire process in one go. It’s the one, definitive dance of electrons. No encores, no intermission. Just a single, powerful performance.
So, the next time you’re faced with a “single-step reaction” and the instruction to “Draw The Curved-arrow Mechanism,” take a deep breath. Embrace the wiggles. Trust your understanding of where those electrons are coming from and where they need to go.
And if all else fails, remember that even the most complicated chemical transformations started with someone, somewhere, drawing a few wiggly arrows. And they probably had a moment of “Wait, does that arrow go there?” too. You’re in good company.
It’s a fun challenge, really. A puzzle with a chemical twist. Just try not to get too lost in the electron-pushing wonderland. And remember, even if it’s a single step, it’s still a step towards understanding the amazing world of chemistry!
